AP Syllabus focus: ‘For all electrochemical cells, oxidation occurs at the anode and reduction occurs at the cathode. (Electrode ‘+’/‘−’ labels are not assessed.)’
Electrochemistry becomes much easier when you anchor every description to one rule: identify where electrons are produced and where they are consumed. That immediately tells you the anode, the cathode, and the oxidation/reduction processes.
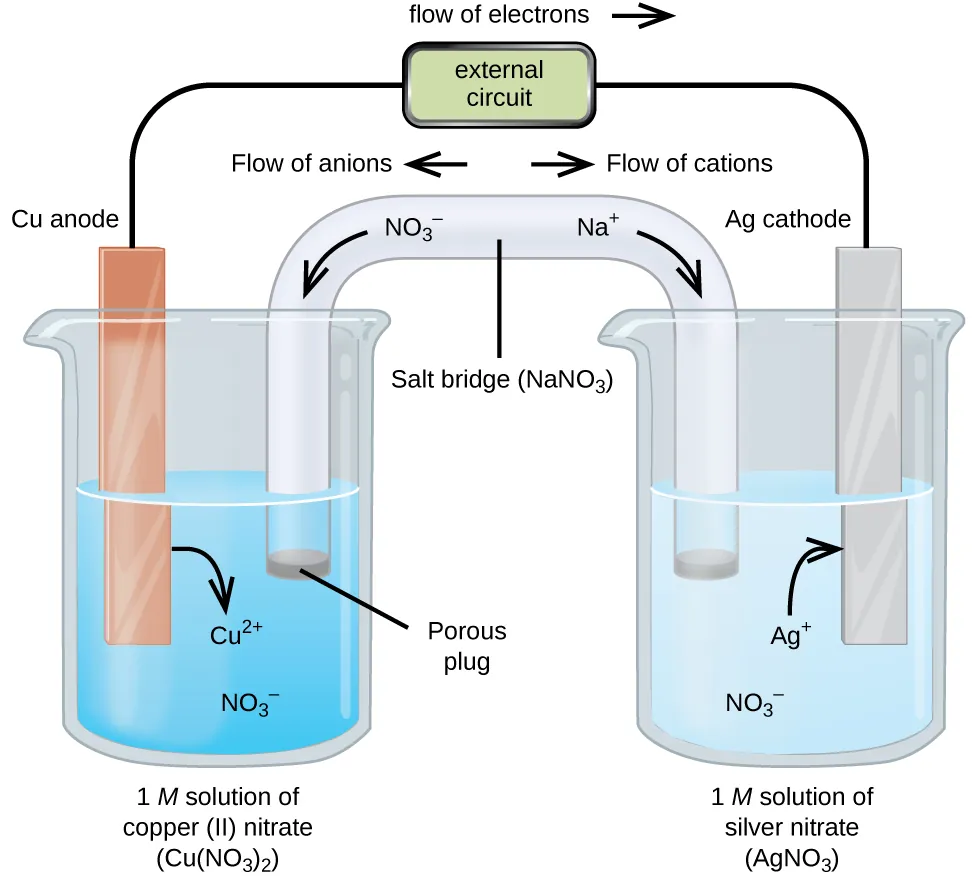
A labeled galvanic (voltaic) cell schematic showing oxidation at the anode and reduction at the cathode, with electrons traveling through the external circuit from anode to cathode. It also highlights charge balance via salt-bridge ion migration (anions toward the anode compartment and cations toward the cathode compartment). Source
The non-negotiable rule
In every electrochemical cell (regardless of whether it is producing electrical energy or requiring it), the labels are assigned by the chemistry happening at each electrode:
Anode: site of oxidation
Cathode: site of reduction
Because this rule is universal, it is more reliable than memorising electrode signs.
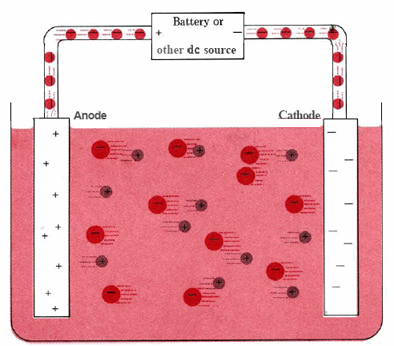
An electrolytic-cell diagram emphasizing that oxidation still occurs at the anode and reduction at the cathode, even though the electrode signs are reversed relative to a galvanic cell. The figure shows a battery driving electron flow so the anode is electron-poor (positive) and the cathode is electron-rich (negative), while ions in solution migrate to maintain charge neutrality. Source
Electrode “+” and “−” depend on the type of cell and are not the core assessed idea here.
What “oxidation” and “reduction” mean (electron accounting)
Electron transfer is the defining feature of redox in cells. Use electron gain/loss rather than vague ideas like “adding oxygen.”
Oxidation: Loss of electrons by a species (electrons appear as products in the half-reaction).
A useful check is that an oxidised species typically shows an increase in oxidation number, consistent with losing negative charge (electrons).
Reduction: Gain of electrons by a species (electrons appear as reactants in the half-reaction).
A reduced species typically shows a decrease in oxidation number, consistent with gaining negative charge.
What “anode” and “cathode” mean (location-based labels)
Electrodes are named for the process occurring on their surface, not for their material or their sign.
Anode: The electrode at which oxidation occurs; electrons are produced at this electrode by the oxidation half-reaction.
This means the anode is the “source” of electrons within the cell: oxidation creates electrons that must be consumed elsewhere.
Cathode: The electrode at which reduction occurs; electrons are consumed at this electrode by the reduction half-reaction.
This means the cathode is the “sink” for electrons: reduction requires electrons, so it must receive them from the anode through the external circuit.
Recognising anode vs cathode from half-reactions
To identify anode and cathode quickly, focus on where appears in each half-reaction.
= electrons released (produced), unitless count tied to stoichiometry
= electrons gained (consumed), unitless count tied to stoichiometry
Once you have the two half-reactions, the mapping is automatic:
Half-reaction with on the product side oxidation anode
Half-reaction with on the reactant side reduction cathode
Electron flow implication (tied to the rule)
The anode/cathode rule also fixes the direction of electron movement in the wire:
Electrons are generated at the anode (by oxidation)
Electrons are used up at the cathode (by reduction)
Therefore, electrons travel externally from anode to cathode
This direction comes directly from conservation of charge: electrons produced by oxidation cannot accumulate indefinitely, so they must be consumed by reduction.
Why “+ / − electrode” is not a safe shortcut
Students often try to identify electrodes using signs first, but that creates errors because sign conventions vary with the overall situation. For this subtopic, what matters is:
Do not define anode/cathode by “+” or “−.”
Do define anode/cathode by oxidation/reduction.
If you ever feel unsure, return to electron bookkeeping: the location producing is the anode; the location consuming is the cathode.
Fast checks that prevent common mistakes
Use these consistency checks when labelling electrodes:
If you label an electrode as the anode, its half-reaction must show electrons as products.
If you label an electrode as the cathode, its half-reaction must show electrons as reactants.
In a correctly balanced overall redox equation, electrons cancel; this only works if oxidation and reduction are assigned to different sites (anode vs cathode) so production and consumption match.
If a species is being oxidised, it must be at the anode; if a species is being reduced, it must be at the cathode—even when the electrode is inert and not chemically changing.
FAQ
Because the same material can serve as an anode in one setup and a cathode in another, depending on which direction the redox change is driven.
The process-based definition (oxidation vs reduction) works universally, including with inert electrodes such as Pt or graphite.
The label refers to what happens at the surface, not whether the electrode itself is consumed.
At an inert anode, oxidation occurs for a species in solution; at an inert cathode, reduction occurs for a species in solution.
Surface catalysis and adsorption can make one electrode kinetically better at a given half-reaction, influencing which reaction actually occurs there.
However, once the half-reaction locations are established, the naming rule still holds: oxidation site = anode; reduction site = cathode.
Yes, in local corrosion or heterogeneous surfaces, microscopic regions can act as tiny anodes and cathodes simultaneously.
You would still label each region by the local process: oxidation at anodic spots, reduction at cathodic spots.
Those phrases come from typical charge-balance situations in certain cells, but they are not defining criteria for anode/cathode.
The only definition you should rely on is electron accounting: where $e^-$ are produced (anode) and where $e^-$ are consumed (cathode).
Practice Questions
Question 1 (1–3 marks) Given the half-reaction , state whether it occurs at the anode or the cathode, and whether it is oxidation or reduction.
1 mark: Identifies the process as oxidation (electrons on product side / loss of electrons).
1 mark: Identifies the electrode as the anode.
1 mark: Correct linkage statement (oxidation occurs at the anode).
Question 2 (4–6 marks) A cell operates with the following half-reactions:
(a) Identify which half-reaction occurs at the anode and which occurs at the cathode. (b) State the direction of electron flow in the external circuit using the electrode names. (c) State which species is oxidised and which species is reduced.
(a) 1 mark: Half-reaction (2) is anode (oxidation).
(a) 1 mark: Half-reaction (1) is cathode (reduction).
(b) 1 mark: Electrons flow from anode to cathode.
(b) 1 mark: Applies to this case: from Cu electrode (anode) to Ag electrode (cathode).
(c) 1 mark: Cu(s) is oxidised to .
(c) 1 mark: is reduced to Ag(s).

