AP Syllabus focus: ‘Electrochemical cell reactions can be favored (positive voltage) or unfavored (negative voltage requiring an externally applied potential to proceed).’
Electrochemical cells convert chemical change into electrical energy (or the reverse) through redox reactions. Interpreting the sign of the measured voltage is essential because it tells you whether the reaction, as written, is thermodynamically able to run.
What “redox in a cell” means
An electrochemical cell separates two half-reactions so electrons are transferred through an external circuit rather than directly by collision in solution.
Oxidation: loss of electrons by a species (its oxidation number increases).
A species that is oxidised provides electrons; a species that is reduced accepts them.
Reduction: gain of electrons by a species (its oxidation number decreases).
How the cell produces a measurable voltage
Because oxidation and reduction are physically separated, electrons can be forced to move through a wire. That electron motion is driven by a difference in electric potential between the two electrodes, which a voltmeter reports as a cell potential (voltage).
Cell potential (voltage), : the potential difference between two electrodes; it reflects the “push” on electrons to flow through an external circuit.
What the voltage sign means (core AP idea)
The AP Chemistry interpretation is:
Positive voltage: the overall cell reaction is favored in the direction written (it can proceed without an external power source).
Negative voltage: the overall cell reaction is unfavored in the direction written (it will not proceed as written unless driven by an externally applied potential).
This is exactly what the syllabus statement captures: favored cell reactions correspond to positive voltage, while unfavored ones correspond to negative voltage that requires outside electrical energy to proceed.
“As written” is the key phrase
The sign is tied to how the overall reaction is written and how the measurement leads are connected.
If you reverse the chemical equation, you reverse the direction of electron transfer and the voltage changes sign.
If you keep the chemistry the same but swap the voltmeter leads, the reading changes sign because the meter reports the potential of one terminal relative to the other.
So, a negative reading often indicates not that “nothing can happen,” but that the spontaneous direction is the reverse of the direction you are testing/measuring.
Connecting voltage sign to electron flow (qualitative)
In a favored cell reaction (positive voltage), electrons flow spontaneously through the external wire from the site where oxidation occurs toward the site where reduction occurs. You can identify oxidation/reduction by electron bookkeeping:
In the oxidation half-reaction, electrons appear on the product side.
In the reduction half-reaction, electrons appear on the reactant side.
If you conceptually force electrons to flow the opposite way (for the same chemical pair), you are describing an unfavored direction; the cell potential for that direction is negative, and sustaining that flow requires an external power supply.
What “externally applied potential” implies
When a cell reaction is unfavored (negative voltage):
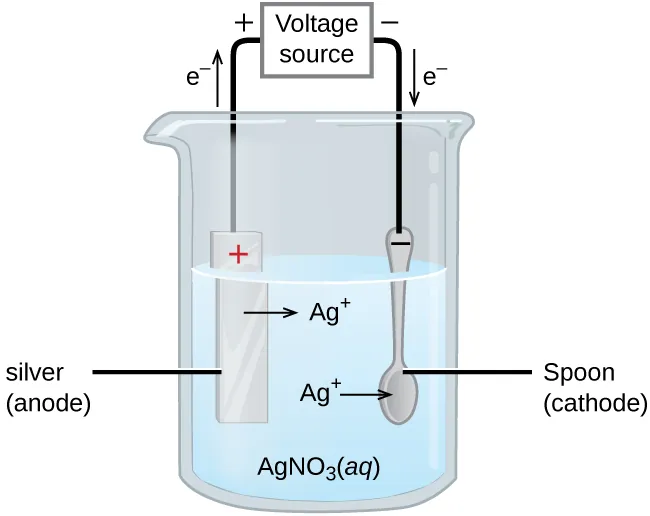
This electrolytic-cell schematic (silver electroplating) highlights that a nonspontaneous redox process is driven by an external voltage source. The diagram’s polarity markings and electron-flow arrows connect the idea of an “externally applied potential” to forced electron motion and the resulting oxidation at the anode and reduction at the cathode. Source
You must apply a voltage of sufficient magnitude (from a battery or power supply) to drive electrons “uphill.”
The applied voltage must oppose the spontaneous direction and enforce the nonspontaneous one.
The system is then consuming electrical energy to make the chemical change occur.
This is the operational meaning of “negative voltage requiring an externally applied potential to proceed.”
Interpreting macroscopic observations using voltage sign
The voltage sign is a thermodynamic indicator of direction, which helps you interpret what you would observe when the circuit is completed:
Positive voltage (favored): sustained electron flow is possible; chemical species are consumed/produced in the direction written.
Negative voltage (unfavored): no sustained electron flow occurs in that direction unless a power source is connected; if the system is allowed to operate freely, it tends to proceed in the reverse direction.
Importantly, a measured voltage close to zero indicates little driving force for net electron flow under the stated conditions, while a large positive or negative value indicates a strong driving force (in the forward or reverse direction, respectively).
Practical sign conventions students must handle
Students commonly lose points by mixing up where the “positive” sign comes from. Keep these conventions straight:
A voltmeter reports . Swapping leads flips the sign.
The sign refers to the overall reaction direction being tested. Reverse the reaction, reverse the sign.
“Favorable” in this context means capable of producing a positive measured cell voltage without needing an external power source; “unfavorable” means the opposite.
FAQ
Most commonly, the voltmeter leads are reversed.
Less commonly, you may have inadvertently set up the half-cells opposite to the intended “as written” direction, so the spontaneous direction is the reverse of what you assumed.
Not necessarily.
It means the reaction is unfavoured in the stated direction without added electrical energy. The reverse reaction may be favoured, and some negligible/initial charge movement can occur before the system settles.
Voltage is a relative measurement: it depends on which side you label as higher potential.
If you redefine the “forward” reaction (or swap measurement terminals), you redefine the sign convention, so $E$ changes sign even though the physical cell is unchanged.
“Positive voltage” refers to electric potential difference, not the sign of the moving charge carriers.
In metal wires, electrons move; conventional current is defined opposite to electron flow. A positive cell voltage corresponds to a spontaneous driving force for electrons in one direction (and conventional current the other way).
It implies a weak driving force for net electron flow under those conditions.
Small changes in setup or conditions can flip the sign of the measured voltage, making the preferred direction difficult to observe without careful control of the measurement convention.
Practice Questions
(2 marks) A student connects an electrochemical cell to a voltmeter and obtains a reading of . What does the negative sign indicate about the reaction as written, and what must be done for the reaction to proceed in that direction?
States that a negative voltage means the cell reaction is thermodynamically unfavoured/non-spontaneous as written (1).
States that an externally applied potential/voltage (e.g. power supply) is required to drive it in that direction (1).
(5 marks) Consider the overall reaction written for a cell: A voltmeter gives a positive reading when the cell operates with this reaction direction. (a) Explain what the positive voltage indicates about the reaction direction (1 mark). (b) State which species is oxidised and which is reduced, justifying your choices using electron/oxidation-number reasoning (3 marks). (c) If the voltmeter leads are swapped while the cell chemistry is unchanged, state what happens to the reading (1 mark).
(a) Positive voltage indicates the reaction is favoured/spontaneous in the direction written (1).
(b) is oxidised: oxidation number increases from 0 to / it loses to form (1).
(b) is reduced: oxidation number decreases from to 0 / it gains to form (1).
(b) Provides correct electron-transfer justification consistent with both changes (e.g. transferred overall) (1).
(c) Swapping leads reverses the sign of the voltmeter reading (same magnitude, opposite sign) (1).

