AP Syllabus focus: ‘Visual representations help identify where oxidation and reduction occur and the direction of current flow in galvanic and electrolytic cells.’
Electrochemical cell diagrams compactly encode what is oxidised, what is reduced, and how charge moves. Learning to “read” the notation lets you locate each half-reaction and infer electron and current directions without relying on a picture.
What a Cell Diagram Encodes
A cell diagram is a shorthand representation of two half-cells connected so electrons can flow through an external circuit while ions move through an internal connection.
Cell diagram: A symbolic line notation that shows the phases, key species, and physical separation between the oxidation half-cell and reduction half-cell in an electrochemical cell.
Core Notation and Separators
Single vertical line, |: a phase boundary (for example, solid electrode in contact with aqueous ions).
Double vertical line, ||: the salt bridge or porous barrier (ions can pass; solutions are kept largely separate).
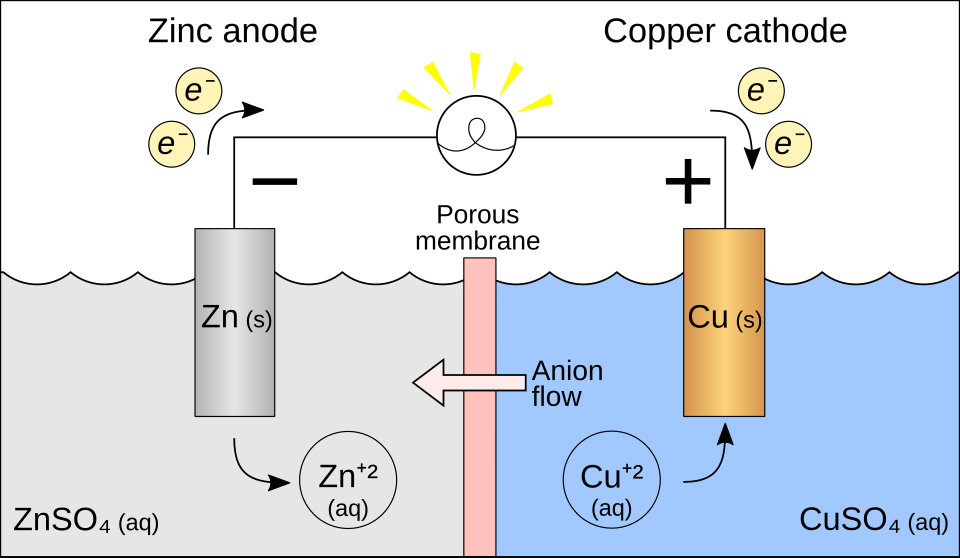
Schematic of a Daniell (Zn–Cu) galvanic cell showing the two half-cells separated by a barrier and connected by an external wire. The diagram explicitly labels the anode/cathode and uses arrows to show electron flow through the wire and compensating ion migration to maintain charge balance. Use it to connect the meaning of the diagram’s physical separator to the role of the salt bridge/porous barrier in completing the circuit. Source
Species written closest to the separator are typically those directly involved at that interface.
Standard Layout (How to Read Left-to-Right)
By convention, many diagrams are written as:
Anode (oxidation) half-cell on the left
Cathode (reduction) half-cell on the right
This convention helps you quickly locate where oxidation and reduction occur from the diagram alone, matching the syllabus emphasis on using visual representations to identify half-reactions and current direction.
Locating the Half-Reactions from the Diagram
Step 1: Identify the Half-Cells
Split the diagram at ||:
Left side = one half-cell
Right side = the other half-cell
Then interpret each half-cell by reading phase boundaries |:
A common pattern is metal(s) | metal ion(aq) (a metal electrode in its ion solution)
Or inert electrode(s) | aqueous species when no solid conducting reactant/product is present
Step 2: Determine Oxidation vs Reduction Sites
Use the diagram’s left/right placement to assign:
Left half-cell: oxidation half-reaction (electrons produced)
Right half-cell: reduction half-reaction (electrons consumed)
Do not depend on electrode “+” or “−” labels; instead, use oxidation/reduction definitions and diagram placement.
Step 3: Write Each Half-Reaction
For a metal/metal-ion half-cell:
If it is the anode side, write it as oxidation: metal atoms become ions + electrons.
If it is the cathode side, write it as reduction: ions gain electrons to form metal.
For non-metal systems (common with inert electrodes), identify which aqueous species changes oxidation state at that electrode surface.
Step 4: Combine Half-Reactions Correctly
Ensure electrons cancel when adding half-reactions.
Keep phases consistent with the diagram (s, aq, g).
Do not alter species that are only present to conduct charge (spectators) unless the diagram shows they change chemically.
Determining Electron Flow and Conventional Current
Electron Flow in the External Circuit
Once anode and cathode are located:
Electrons flow from anode to cathode through the wire (from oxidation to reduction).
Conventional current: The direction positive charge would flow in a circuit; it is opposite to electron flow in metal wires.
Conventional Current Direction
Conventional current in the external circuit runs from cathode to anode (opposite the electrons).
Many prompts say “current direction”; if unclear, state explicitly whether you mean electron flow or conventional current.
Using the Diagram to Infer Ion Movement (Internal Charge Balance)
Cell diagrams also imply how the solutions remain electrically neutral as redox occurs.
Oxidation at the anode typically produces cations (or consumes anions), so anions migrate into the anode compartment through the bridge/barrier.
Reduction at the cathode typically consumes cations (or produces anions), so cations migrate into the cathode compartment.
These migrations maintain charge balance; they do not replace electron flow in the wire.
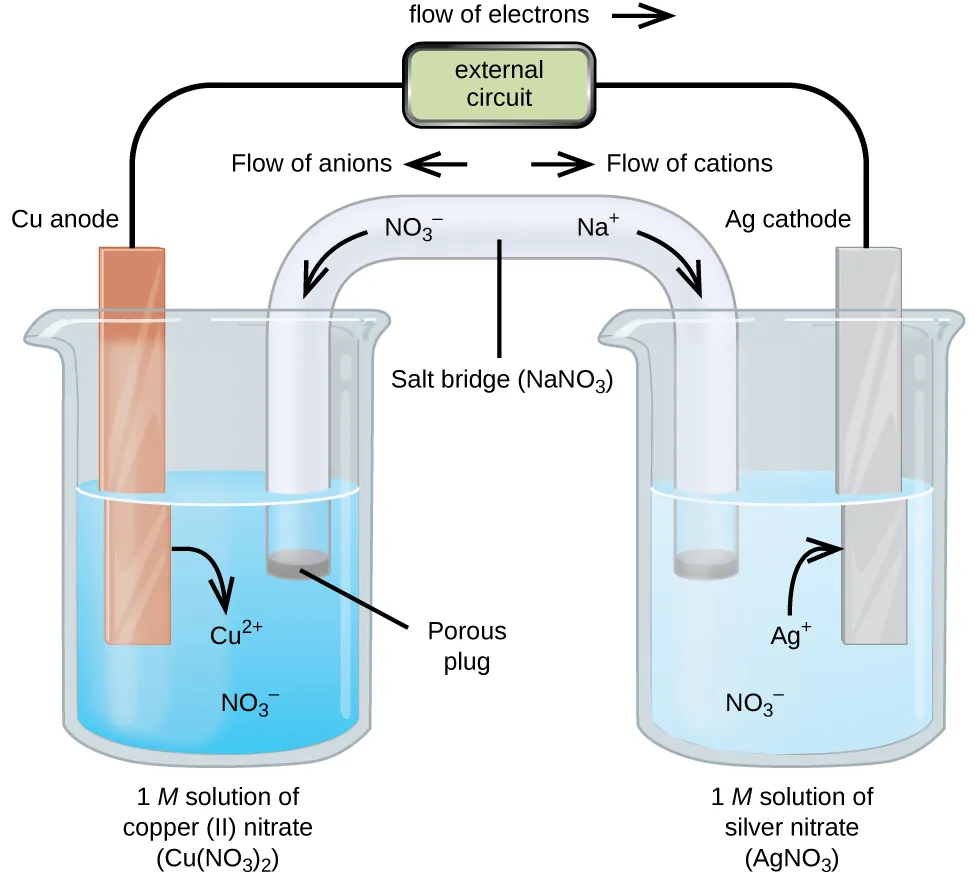
Diagram of a galvanic cell highlighting two different kinds of charge motion: electrons moving through the external circuit and ions migrating through the salt bridge to preserve electroneutrality. The labels make it easy to distinguish oxidation-side solution changes from reduction-side solution changes while tracking how the circuit is completed. This is the visual counterpart to reading a cell diagram as a compact encoding of both half-reactions and charge flow. Source
Special Cases in Cell Diagrams
Inert Electrodes (Pt or C)
If no conducting solid is produced/consumed (for example, only gases and aqueous ions participate), the diagram includes an inert conductor:
Pt(s) or C(s) provides a surface for electron transfer.
The inert electrode is written at the appropriate side of the half-cell, adjacent to the species it contacts (often a gas or aqueous redox couple).
Gas Electrodes
Gas species are written with phase boundaries indicating contact:
A common pattern is Pt(s) | gas(g) | ions(aq)
The placement shows the gas is involved at that electrode interface.
Multiple Aqueous Species in a Half-Cell
When both oxidised and reduced forms are aqueous, they may be listed together in that half-cell:
The diagram indicates the redox couple present at that electrode.
Identify which species is oxidised/reduced by considering electron production/consumption at the anode/cathode positions.
FAQ
It is a convention to place oxidation on the left and reduction on the right.
If a diagram is written differently, you must rely on explicit labels provided or accompanying context, not the left/right assumption.
It indicates multiple aqueous species are present in the same half-cell at one electrode interface.
It does not imply they are separate phases; both participate as a redox couple.
If a species is not part of the redox change at the electrode, it typically maintains charge balance or conductivity.
Only include it in half-reactions if its oxidation state changes.
State both directions explicitly:
Electron flow: anode $\rightarrow$ cathode
Conventional current: cathode $\rightarrow$ anode
It means neither half-cell has a solid conducting reactant/product electrode.
Platinum serves only as an inert conductor for electron transfer at each half-cell interface.
Practice Questions
(2 marks) A cell is represented as: Zn(s) | Zn(aq) || Cu(aq) | Cu(s).
State (i) which side is oxidation and (ii) the direction of electron flow in the external circuit.
Left-hand half-cell is oxidation / anode (1)
Electrons flow from Zn electrode to Cu electrode (left to right) (1)
(5 marks) A cell diagram is: Pt(s) | H(g) | H(aq) || Fe(aq), Fe(aq) | Pt(s).
(a) Identify the anode and cathode half-cells.
(b) Write the oxidation half-reaction and the reduction half-reaction.
(c) State the direction of conventional current in the external circuit.
(a) Left is anode (oxidation), right is cathode (reduction) (1)
(b) Oxidation: H(g) 2H(aq) + 2e (1)
(b) Reduction: 2Fe(aq) + 2e 2Fe(aq) (1)
Electrons cancel when combining (implicit by matching 2e) (1)
(c) Conventional current flows from cathode to anode (right to left) (1)

