AP Syllabus focus: ‘Galvanic (voltaic) cells involve a thermodynamically favored reaction, while electrolytic cells involve an unfavored reaction that requires input energy.’
Electrochemical cells connect redox reactions to energy changes. The key distinction is whether the overall reaction is thermodynamically favored and can produce electrical energy, or thermodynamically unfavored and must be driven by an external energy source.
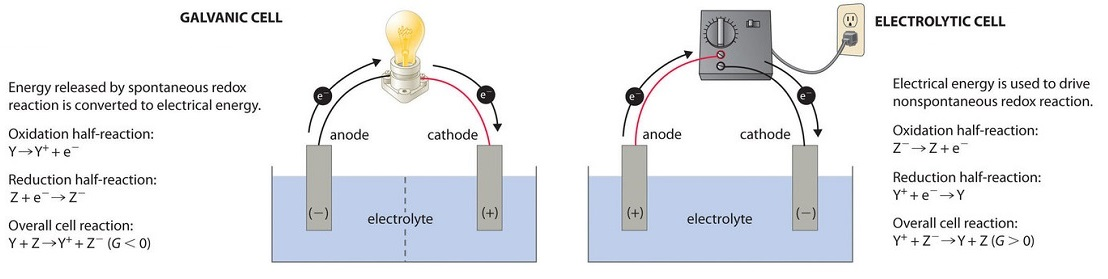
Side-by-side schematic comparing a galvanic cell (spontaneous redox, electrical work output) with an electrolytic cell (nonspontaneous redox driven by an external power source). The diagram emphasizes the direction of electron flow in the circuit and how an applied potential reverses the natural direction to force an otherwise unfavored reaction. Source
Thermodynamic favorability in electrochemical cells
Spontaneous vs nonspontaneous processes
Thermodynamically favored (spontaneous): A process that can proceed without continuous external energy input under the stated conditions.
In cell language, “favored” means the cell reaction can be used as a source of electrical energy, while “unfavored” means electrical energy must be supplied to force the reaction to occur. Thermodynamic favorability is a statement about energy (not speed).
= Gibbs free energy change for the overall cell reaction (J or kJ)
= Gibbs free energy change for the overall cell reaction (J or kJ)
A reaction with can be coupled to useful work (like pushing electrons through a circuit). A reaction with is not “impossible”; it can occur if the surroundings supply enough energy in the correct form.
Galvanic (voltaic) cells: favored reactions that generate energy
Galvanic (voltaic) cell: An electrochemical cell in which a thermodynamically favored redox reaction produces electrical energy.
Core idea: a galvanic cell converts chemical potential energy into electrical energy.
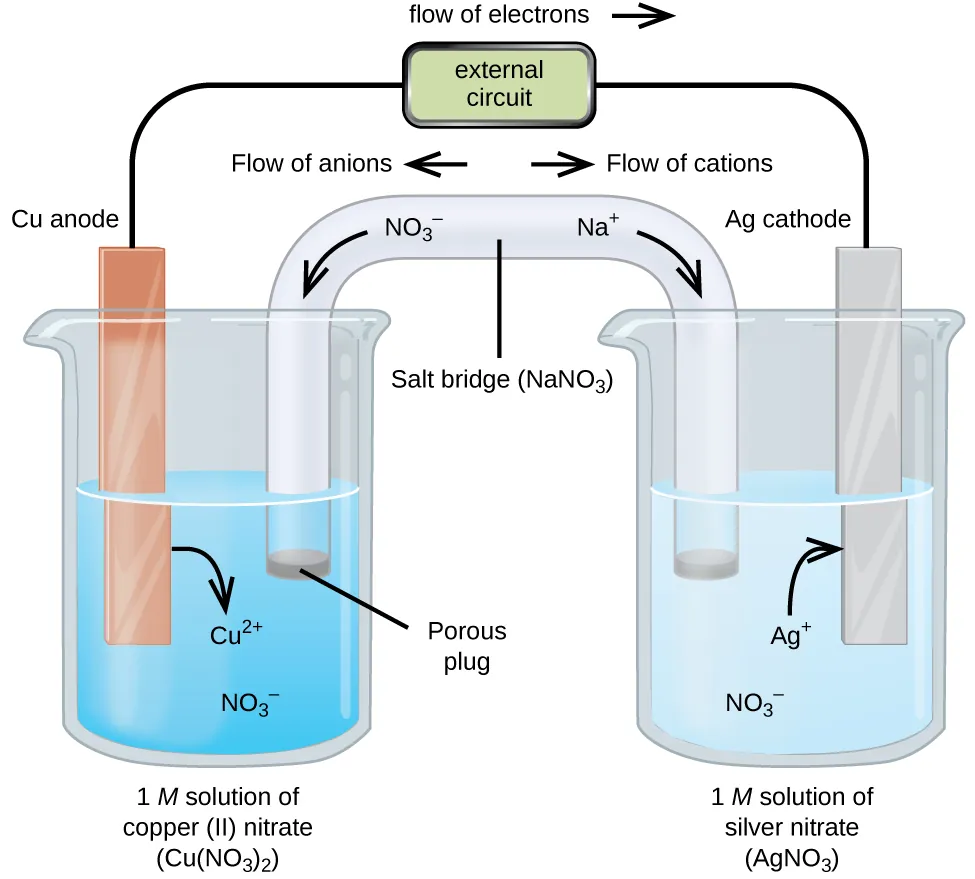
Diagram of a galvanic (voltaic) cell showing separated half-cells connected by an external circuit and a salt bridge. The labels highlight the anode (oxidation), cathode (reduction), electron flow through the wire, and compensating ion migration in the salt bridge that maintains electrical neutrality as the spontaneous reaction proceeds. Source
Key features tied to favorability:
The overall redox reaction is thermodynamically favored under the cell’s conditions.
The cell can operate as a power source (it can run a device) without an external battery forcing it.
As the reaction proceeds, the system releases free energy; that energy can appear as electrical work.
Common student pitfalls (about favorability):
Confusing “favored” with “fast”: a favored reaction may still proceed slowly if kinetically hindered.
Thinking any redox reaction automatically makes a working battery: only those with a favored overall direction under the conditions will generate electrical energy.
Electrolytic cells: unfavored reactions that require input energy
Electrolytic cell: An electrochemical cell in which an external energy source forces a thermodynamically unfavored redox reaction to occur.
Core idea: an electrolytic cell converts electrical energy into chemical change.
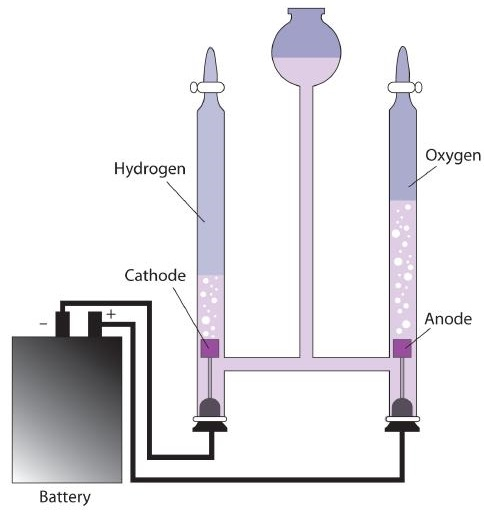
Diagram of an electrolytic cell illustrating how an external power source drives electron flow to force a nonspontaneous redox reaction. The labels connect the battery to the electrodes, making it clear that electrical energy input is what sustains the chemical change during electrolysis. Source
Key features tied to unfavorability:
The overall redox reaction is thermodynamically unfavored under the conditions in the cell.
The process requires input energy, typically from an external power supply, to drive electron transfer in the nonspontaneous direction.
The electrical energy supplied is stored as chemical potential energy in the products (e.g., plating a metal, generating a reactive substance).
Important clarifications:
“Requires input energy” means continuous energy input while the reaction is running; removing the power source stops (or greatly reduces) the driven reaction.
Electrolytic processes are often used to make substances that are not product-favored in the desired direction under those conditions.
Comparing favored vs unfavored: what AP Chemistry expects you to say
When distinguishing cell types, focus on the overall thermodynamic direction:
Galvanic (voltaic): overall reaction is favored; cell produces electrical energy.
Electrolytic: overall reaction is unfavored; cell consumes electrical energy to make reaction proceed.
How to interpret typical prompts without drifting into unnecessary detail:
If a description says the cell “acts as a battery” or “powers a device,” it implies a galvanic cell (favored reaction).
If a description says a “power supply is connected” or the process is “driven by an applied voltage/current,” it implies an electrolytic cell (unfavored reaction).
FAQ
Yes. During discharge it operates as a galvanic cell (producing electrical energy). During charging, an external power source drives the reverse, nonspontaneous direction, so it operates electrolytically.
Nonspontaneous means the process is not thermodynamically favoured without energy input. Supplying electrical energy can make the overall coupled process proceed in the desired direction.
Beyond thermodynamics, practical electrolysis often needs extra voltage due to:
electrode overpotentials,
internal resistance,
side reactions and inefficiencies.
Yes. Changing conditions (especially concentrations) can change whether the overall reaction is favoured. If conditions make the desired direction unfavoured, the same redox pair would require external energy to run that direction.
A favoured reaction may still deliver poor power if current is limited by factors such as high internal resistance, sluggish interfacial charge transfer, or mass-transport limitations.
Practice Questions
(2 marks) A redox process is set up in a cell and is observed to run a small motor without any external power supply connected. State whether the cell is galvanic (voltaic) or electrolytic, and state whether the overall reaction is thermodynamically favoured or unfavoured.
Galvanic/voltaic (1)
Thermodynamically favoured / spontaneous / (1)
(5 marks) A student claims: “If a reaction is nonspontaneous, it cannot be made to occur in an electrochemical cell.” Using the ideas of galvanic versus electrolytic cells, assess this claim. Your answer must:
identify which type of cell corresponds to spontaneous reactions and which to nonspontaneous reactions,
explain the role of external energy input,
describe the direction of energy conversion in each type of cell.
Galvanic cell corresponds to thermodynamically favoured/spontaneous reaction (1)
Electrolytic cell corresponds to thermodynamically unfavoured/nonspontaneous reaction (1)
External energy input (power supply) is required to force the unfavoured reaction in an electrolytic cell (1)
Energy conversion: chemical energy to electrical energy in a galvanic cell (1)
Energy conversion: electrical energy to chemical change/chemical potential energy in an electrolytic cell (1)

