AP Syllabus focus: ‘Electrodes, half-cell solutions, a salt bridge, and a measuring device each play specific roles in an electrochemical cell’s function.’
Electrochemical cells convert between chemical and electrical energy by separating oxidation and reduction into connected parts. Knowing what each physical component does helps you predict charge flow, maintain neutrality, and interpret what instruments display.
What an electrochemical cell is made of
An electrochemical cell is built from two coupled half-cells that are connected in two ways: an external electron pathway and an internal ion pathway. Each required component supports one of these connections.
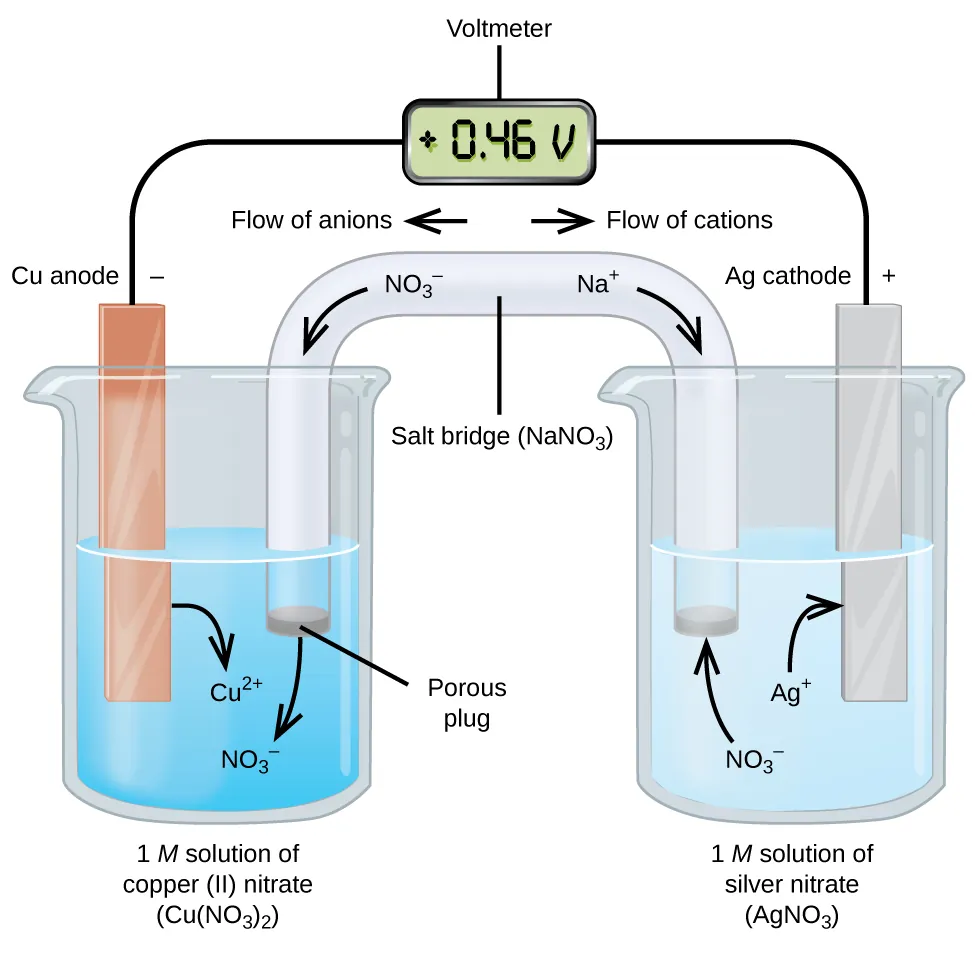
Labeled galvanic-cell schematic showing the two half-cells connected by an external wire (electron pathway) and a salt bridge (ion pathway). The diagram also labels the anode/cathode and illustrates how anions and cations migrate through the bridge to maintain electrical neutrality while a voltmeter reads the potential difference. Source
Electrochemical cell: A system in which a redox reaction is separated into half-reactions so that electron transfer occurs through an external circuit while ions move through an internal pathway to maintain charge balance.
Because the half-reactions are physically separated, the cell must provide routes for both electrons and ions, or the reaction quickly stops due to charge buildup.
Electrodes: solid conductors where half-reactions occur
Electrodes are the solid surfaces that allow electron transfer between the chemical species in solution and the external circuit.
Electrode: A conductive solid (metal or inert conductor) that provides a surface for oxidation or reduction and a pathway for electrons to enter or leave the half-cell.
Key roles of electrodes:
Provide a conducting path so electrons can move through the wire rather than directly between species in solution.
Provide a reaction interface where dissolved species can gain or lose electrons.
Sometimes participate chemically (e.g., a metal electrode that can dissolve to ions or be plated with metal), or be inert (conductive but not consumed) when only solution species react.
A practical cell uses well-connected electrodes (good contact with the wire and solution) so the measured electrical effect corresponds to the ongoing redox process.
Half-cell solutions: the chemical environments that supply reactants
Each electrode sits in a half-cell solution that contains the relevant oxidised and/or reduced forms of a redox couple. These solutions are the reservoirs of reactants and products for each half-reaction.
Important functions of half-cell solutions:
Supply mobile ions that can approach the electrode surface to react.
Establish the chemical conditions (identity of ions, their concentrations, and solvent) that enable a particular half-reaction to occur.
Provide ionic conductivity within each compartment so charge can move locally as electrons enter/leave via the electrode.
In many laboratory setups, the half-cell solutions are kept physically separated (for example, in separate beakers) to prevent direct mixing that would bypass the electrical pathway.
Salt bridge (or porous barrier): ion pathway that prevents charge buildup
A salt bridge connects the two half-cells internally and completes the circuit for ion flow.
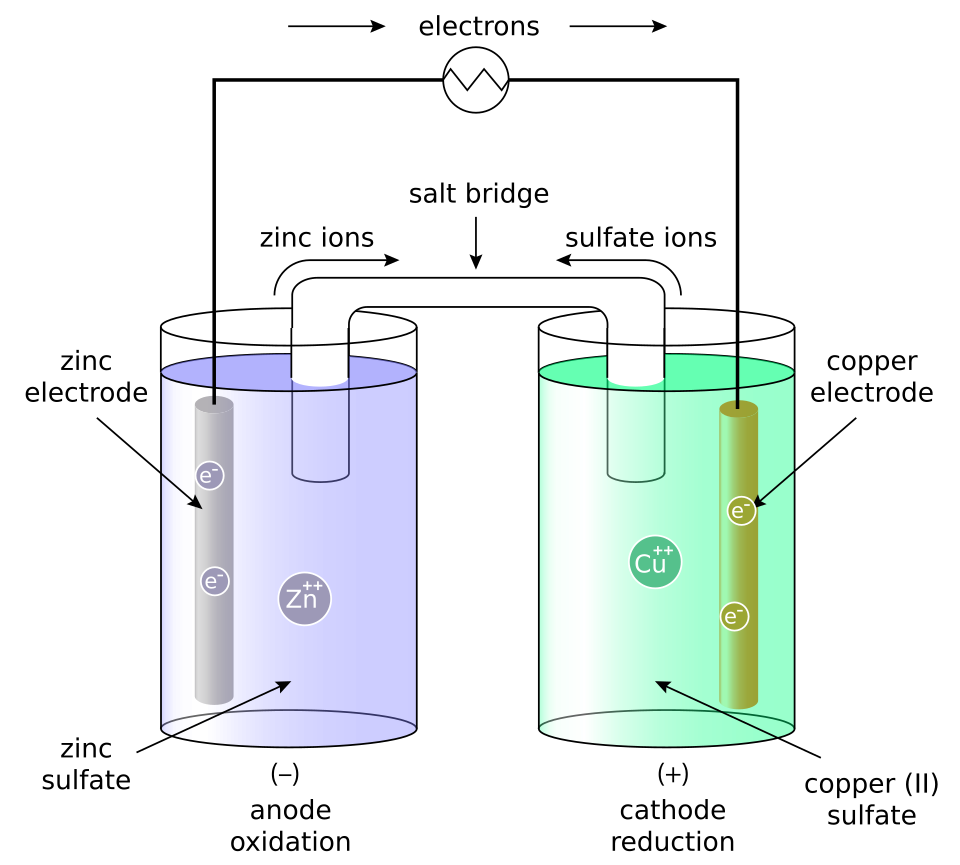
Labeled Daniell-cell diagram (Zn/Zn and Cu/Cu) emphasizing how the salt bridge completes the internal circuit and prevents charge buildup in each half-cell. The figure visually ties ion migration in the bridge to continued electron flow in the external circuit, clarifying why the redox process stalls if the ion pathway is removed. Source
Salt bridge: A connection (often a tube filled with an inert electrolyte gel) that allows ions to migrate between half-cells to maintain electrical neutrality without extensively mixing the half-cell solutions.
Roles of the salt bridge:
Prevents charge accumulation: if one half-cell becomes too positive or too negative, electron flow through the wire would be hindered.
Maintains overall neutrality by letting ions migrate in response to changing charge in each half-cell.
Minimises direct chemical mixing of the half-cell solutions, helping keep the half-reactions separated.
Uses an electrolyte whose ions are typically spectator ions (they should not react with the redox species or form precipitates).
If the salt bridge is missing, blocked, or contains reactive ions, the cell potential can become unstable or the cell can stop quickly.
Measuring device: reading or controlling the electrical output
A measuring device is attached to the electrodes through the external circuit to observe (or impose) an electrical effect.
Measuring device: An external instrument connected to the electrodes (such as a voltmeter or ammeter) that measures the cell’s electrical behaviour, or a power supply that controls it.
Typical roles:
A voltmeter measures the potential difference between electrodes while ideally drawing negligible current.
An ammeter measures current when placed in series, indicating the rate of charge flow in the external circuit.
In some setups, an external power source may be connected to enforce a particular direction of electron flow; regardless, the instrument’s role is to interface the cell with the external circuit in a measurable way.
How the components work together (core idea)
Electrodes + wire: provide the external path for electrons.
Half-cell solutions: provide the reacting species and local ionic conductivity.
Salt bridge: provides the internal path for ions to maintain charge balance.
Measuring device: provides observation (and sometimes control) of the electrical output created by the separated half-reactions.
FAQ
“Inert” ions do not oxidise/reduce with cell species and do not form precipitates.
Reactive bridge ions can consume reactants or clog the junction, altering readings.
Yes; it still permits ion migration.
It may allow more mixing than a gel bridge, which can slowly change half-cell composition.
If no solid reactant/product can act as the conductor, an inert electrode supplies a surface for electron transfer without being consumed.
Different ion mobilities can create a small charge separation at interfaces.
This can slightly shift the measured potential.
Use high internal resistance (draws minimal current) and secure, clean electrical contacts.
Avoid contaminated or drying salt bridges.
Practice Questions
Q1 (2 marks): State the role of (i) an electrode and (ii) a salt bridge in an electrochemical cell.
Electrode: provides a conductive surface/pathway for electron transfer where a half-reaction occurs. (1)
Salt bridge: allows ion migration to maintain electrical neutrality/complete the circuit without major mixing. (1)
Q2 (5 marks): A student builds a cell using two half-cells connected by a salt bridge and wires to a voltmeter. Explain, in terms of the core components, why (a) electrons travel through the wire rather than the solution, (b) ions must travel through the salt bridge, and (c) the half-cell solutions are kept separate.
Electrons travel through the wire because the electrodes are conductors that provide the external electron pathway between half-reactions. (1)
The solution does not serve as the external electron path; charge in solution is carried by ions rather than free electrons. (1)
Ions move through the salt bridge to prevent charge build-up in either half-cell and keep each solution electrically neutral. (1)
The salt bridge completes the internal circuit by providing an ionic connection between half-cells. (1)
Keeping half-cell solutions separate prevents direct reaction/mixing that would bypass the external circuit and reduce measurable electrical output. (1)

