AP Syllabus focus: ‘In a coupled system, summing the individual reactions yields an overall reaction that achieves the desired outcome and has ΔG° < 0.’
Coupled reactions let chemistry “pay” for an unfavored change by combining it with a favored one.
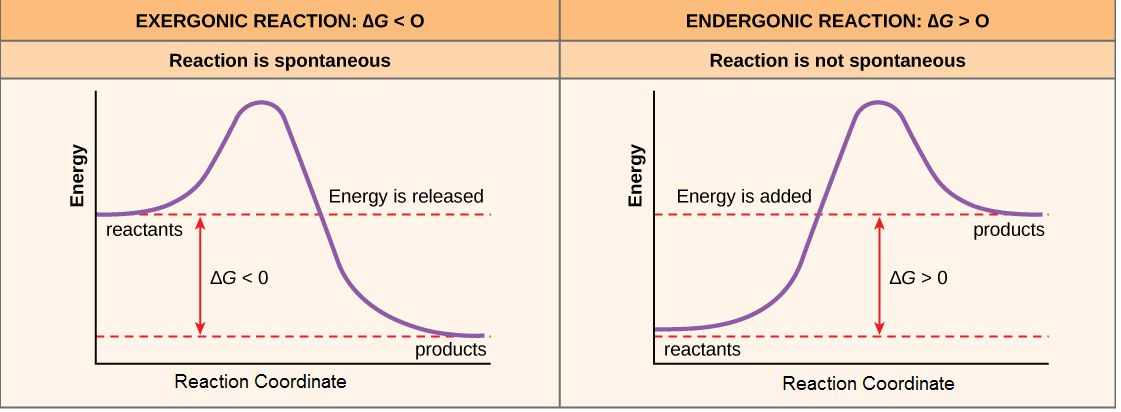
Reaction-coordinate diagrams for exergonic and endergonic reactions, showing how Gibbs free energy changes from reactants to products. The sign of is determined by the relative energies of reactants and products (downhill for exergonic, uphill for endergonic). This is the energetic picture that coupling exploits: a sufficiently downhill step can offset an uphill step in the net sum. Source
This page focuses on how adding reactions makes a net process thermodynamically favored by ensuring an overall negative standard free energy change.
Core idea: net favorability comes from adding reactions
In reaction coupling, you combine multiple chemical equations so that unwanted steps are offset by strongly favorable steps. Thermodynamic favorability under standard conditions is judged by the sign of ΔG° for the overall reaction.
Overall reaction: the net chemical equation obtained by algebraically adding component reactions and canceling any species that appear on both sides.
To get a desired product, you choose component reactions that share species (often intermediates) so they cancel when summed, leaving the target transformation.
The criterion you are trying to achieve
The syllabus requirement can be stated operationally: when you add the individual reactions, you must also add their ΔG° values. If the result is negative, the coupled net reaction is thermodynamically favored under standard conditions.
= standard Gibbs free energy change for the net (summed) reaction, in
= standard Gibbs free energy change for the th component reaction, in
This additivity is why coupling works: a sufficiently negative driving reaction can outweigh a positive nonspontaneous (unfavored) one.
How to correctly “add” reactions and their ΔG°
Adding reactions is algebra with chemical meaning.
The overall equation must be balanced, and the free energies must be adjusted in exactly the same way you adjust the equations.
Allowed operations (and what happens to ΔG°)
Add reactions as written
Add left sides together and right sides together
Then cancel identical species appearing on both sides
Add the corresponding ΔG° values
Reverse a reaction
Swap reactants and products
Change the sign of ΔG° (a favored direction becomes unfavored, and vice versa)
Multiply a reaction by a factor
Multiply all coefficients by that factor
Multiply ΔG° by the same factor (because free energy is extensive with respect to reaction amount)
Why intermediates matter in coupling
Coupled systems typically share one or more intermediates so that:
The intermediate is produced in one step and consumed in another
It cancels from the overall reaction
The net equation shows only the overall chemical change you care about, while the thermodynamics reflect the sum of the steps
What “overall ΔG° < 0” means (and what it doesn’t)
When ΔG°overall < 0, the combined process is thermodynamically favored under standard-state conditions for the overall reaction you wrote.
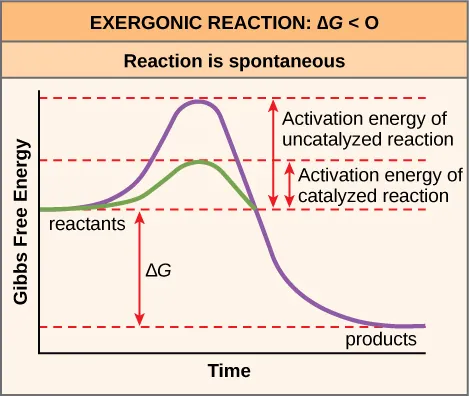
Free-energy profile comparing an uncatalyzed pathway (higher activation barrier) to a catalyzed pathway (lower barrier) while keeping the overall free-energy change the same. The vertical difference between reactants and products represents , whereas the peak height above reactants represents the activation energy. This reinforces that predicts thermodynamic favorability, not reaction speed. Source
In practice, effective coupling requires:
The equations are combined consistently (same direction conventions)
The driving reaction is sufficiently negative in ΔG° magnitude
Species cancel properly so the net reaction matches the desired outcome
Common pitfalls that break net-favorability reasoning
Forgetting to flip the sign of ΔG° when reversing a component reaction
Scaling coefficients but not scaling ΔG° by the same factor
Cancelling species incorrectly (especially when coefficients differ)
Treating reactions as “added” even when they cannot share/cancel species to form the intended net equation
FAQ
Because $\Delta G^\circ$ is a state function: it depends only on initial and final states. Adding stepwise pathways gives the same net state change, so the free-energy changes sum.
No. “Adding” is thermodynamic bookkeeping. Physical coupling usually requires shared intermediates so that one step supplies reactants consumed by another.
Nothing extra is done to $\Delta G^\circ$. Cancellation is already accounted for by adding the steps’ $\Delta G^\circ$ values after correctly orienting and scaling the component equations.
Not in a chemically meaningful way for driving a target change, because there is no mechanism to transfer the “drive” between them; the equations won’t combine to eliminate an intermediate and yield the desired net reaction.
It guarantees thermodynamic favourability for the written net reaction under standard conditions, but real systems may require effective coupling (shared intermediates and pathways) for the drive to be realised.
Practice Questions
Q1 (2 marks): Two steps have and . If added to form an overall reaction, determine and whether the overall reaction is thermodynamically favoured under standard conditions.
(1)
Overall reaction thermodynamically favoured because (1)
Q2 (5 marks): An unfavoured step A has . A proposed driving step B has as written. You may multiply any reaction by an integer and/or reverse it. Decide the smallest integer multiple of B needed to couple with A to give , and state the resulting .
Recognises multiplying B by gives (1)
Sets condition (1)
Solves so smallest integer (1)
Calculates (1)
States overall reaction is thermodynamically favoured because negative (1)

