AP Syllabus focus: ‘Predict when ΔG° < 0 using the signs of ΔH° and ΔS°: favored at all, none, high, or low temperatures depending on the sign combination.’
Thermodynamic favorability depends on how enthalpy and entropy compete as temperature changes. By tracking the signs of ΔH° and ΔS°, you can predict whether a process is favored at all temperatures, none, or only high/low temperatures.
Core idea: how temperature changes favorability
A process is thermodynamically favored under the stated conditions when its Gibbs free energy change is negative.
Thermodynamically favored: a process with ΔG° < 0 under the specified conditions, meaning it is energetically “downhill” in free energy.
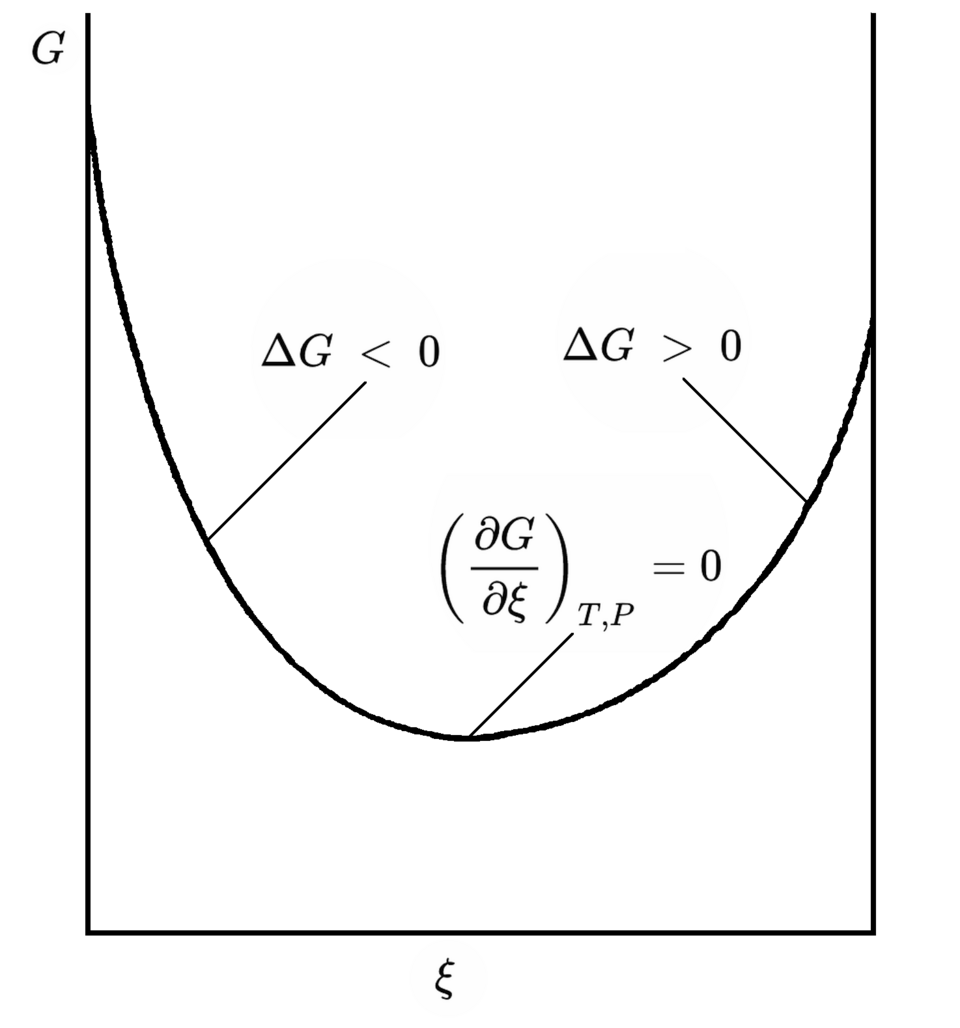
Gibbs free energy is plotted versus reaction progress (extent of reaction), showing that systems spontaneously move in the direction that decreases until equilibrium is reached. The minimum corresponds to equilibrium, where the driving force vanishes (the slope is zero). This visual supports the idea that negative free-energy change corresponds to an energetically “downhill” direction under the stated conditions. Source
Temperature enters through the entropy term, so increasing increases the magnitude of the contribution.
= standard Gibbs free energy change (typically )
= standard enthalpy change (typically )
= absolute temperature (K)
= standard entropy change (typically after unit conversion)
This sign-analysis method is qualitative: it tells you the temperature region(s) where ΔG° will be negative, without requiring numerical data.
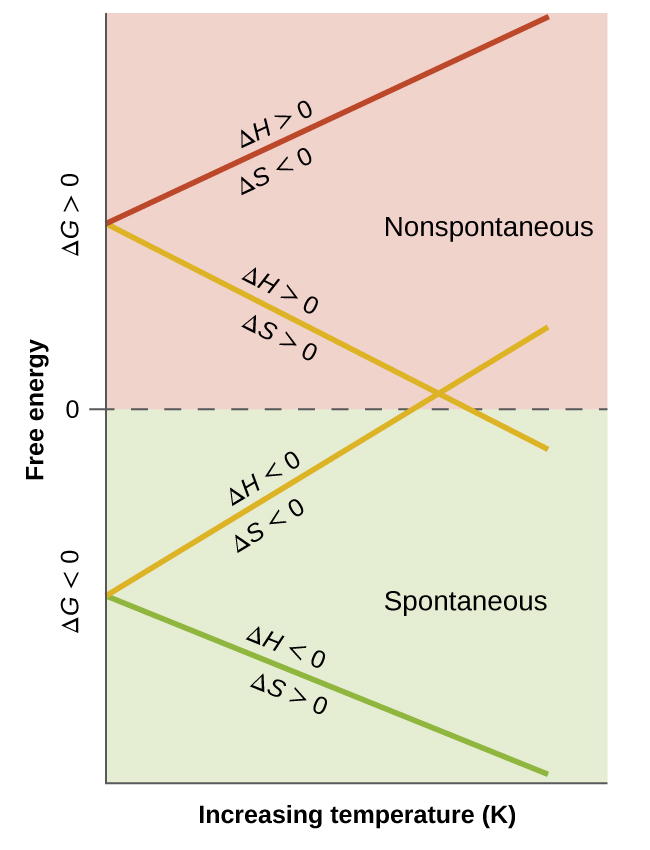
This graph summarizes how varies with temperature for the four possible sign combinations of and . The y-intercept reflects , while the slope is controlled by , so changing the sign of flips whether increasing makes more negative or more positive. The crossings of visually mark the temperature thresholds separating “spontaneous” () from “nonspontaneous” () behavior. Source
Sign logic you must be able to apply
How to think about each term
ΔH° term: “enthalpy push”
ΔH° < 0 helps make ΔG° negative (favors the process).
ΔH° > 0 opposes making ΔG° negative.
term: “entropy push scaled by temperature”
If ΔS° > 0, then is negative, and it becomes more negative as increases (increasingly favorable).
If ΔS° < 0, then is positive, and it becomes more positive as increases (increasingly unfavorable).
The four required cases (sign combinations)
Case 1: ΔH° < 0 and ΔS° > 0
Both terms favor ΔG° < 0:
ΔH° is negative.
is negative for any .
Prediction: favored at all temperatures (within the range where ΔH° and ΔS° signs remain valid).
Case 2: ΔH° > 0 and ΔS° < 0
Both terms oppose ΔG° < 0:
ΔH° is positive.
is positive and grows with .
Prediction: favored at no temperatures (again, within the applicable temperature range).
Case 3: ΔH° < 0 and ΔS° < 0
Enthalpy favors; entropy opposes:
ΔH° negative helps.
positive hurts more at higher .
Temperature dependence:
At low , the entropy penalty is small, so ΔH° can dominate.
At high , the entropy penalty grows and can overwhelm ΔH°.
Prediction: favored at low temperatures only.
Case 4: ΔH° > 0 and ΔS° > 0
Enthalpy opposes; entropy favors:
ΔH° positive hurts.
negative helps more at higher .
Temperature dependence:
At low , the entropy benefit is small, so ΔH° dominates (unfavorable).
At high , the entropy term can outweigh ΔH°.
Prediction: favored at high temperatures only.
Common reasoning errors to avoid
Always use Kelvin for any temperature reasoning involving .
Don’t forget the minus sign in ; it flips how ΔS° affects ΔG°.
“Favored at high temperature” does not mean “fast”; favorability is thermodynamic, not kinetic.
FAQ
If $\Delta H^\circ$ and $\Delta S^\circ$ are treated as roughly constant, set $\Delta G^\circ = 0$ in $\Delta G^\circ = \Delta H^\circ - T\Delta S^\circ$.
This gives $T \approx \dfrac{\Delta H^\circ}{\Delta S^\circ}$ (with consistent units), which approximates the boundary between $\Delta G^\circ>0$ and $\Delta G^\circ<0$.
They can vary because heat capacities differ between reactants and products.
Over modest temperature ranges, sign-based predictions usually hold, but near phase changes or very wide ranges, $\Delta H^\circ$ and $\Delta S^\circ$ may shift enough to move (or remove) the crossover.
Not necessarily. $\Delta S^\circ<0$ means the system’s entropy decreases.
That can arise from:
fewer accessible microstates,
fewer gas particles,
stronger constraints on motion (e.g. association/condensation), but “order” is a simplified interpretation and may mislead in complex systems.
A larger $|\Delta S^\circ|$ makes the $T\Delta S^\circ$ term grow more rapidly with temperature.
So reactions with large positive $\Delta S^\circ$ become favourable at lower “high temperatures,” while reactions with large negative $\Delta S^\circ$ become unfavourable more quickly as $T$ rises.
Then $T\Delta S^\circ$ contributes little even at high $T$, so $\Delta G^\circ$ is dominated by $\Delta H^\circ$.
In that limit, temperature has minimal effect on favourability, and the process behaves almost like “enthalpy-controlled” thermodynamically.
Practice Questions
(2 marks) A reaction has and . Predict whether it is thermodynamically favoured at high temperatures, low temperatures, all temperatures, or no temperatures.
Identifies “favoured at low temperatures” (1)
Correct reasoning that increasing makes more positive when , opposing favourability (1)
(5 marks) For a process with and : (a) State how the sign of changes with increasing temperature. (1) (b) Use to explain whether the process is favoured at low temperatures. (2) (c) Predict the temperature conditions under which the process becomes favoured. (2)
(a)
States is negative and becomes more negative as increases when (1)
(b)
Notes is positive (1)
Concludes at low , the magnitude of is small so tends to remain positive/unfavoured (1)
(c)
Predicts favoured at high temperatures (1)
Explains high increases the magnitude of the negative term so it can outweigh positive giving (1)

