AP Syllabus focus: ‘The standard Gibbs free energy change measures thermodynamic favorability: processes with ΔG° < 0 are favored under standard conditions.’
Thermodynamic predictions answer whether a reaction is energetically “downhill,” not whether it is fast.
In AP Chemistry, the sign of ΔG° is the key indicator of whether a process is favored under standard conditions.
Thermodynamic favorability and what ΔG° represents
The idea of “favored” vs “unfavored”
A process is thermodynamically favored under standard conditions when it has a natural tendency to proceed in the written direction without requiring continuous external energy input. A process is thermodynamically unfavored when it does not have that tendency under standard conditions.
Standard Gibbs free energy change (ΔG°): the change in Gibbs free energy for a reaction as written when all species are in their standard states; it is a thermodynamic criterion for favorability under standard conditions.
Because ΔG° is tied to the reaction “as written,” changing the balanced equation (for example, reversing it or multiplying coefficients) changes the reported ΔG° accordingly.
How to read the sign (and what it means physically)
The sign of ΔG° encodes whether the products are at lower or higher Gibbs free energy than the reactants under standard conditions:
Negative ΔG°: moving from reactants to products lowers Gibbs free energy; the reaction is favored under standard conditions.
Positive ΔG°: moving from reactants to products raises Gibbs free energy; the reaction is unfavored under standard conditions.
ΔG° = 0: the system is at a boundary condition under standard conditions; there is no thermodynamic “push” in either direction.
A useful physical interpretation is that Gibbs free energy tracks energy available to do non-expansion work while accounting for the entropy/enthalpy balance implicitly.
A favored process corresponds to a decrease in G for the system at standard conditions.
The sign rules you must apply correctly
= standard Gibbs free energy change (commonly reported in )
= depends on the balanced reaction as written (including phase labels)
These inequalities are not “rate statements.”
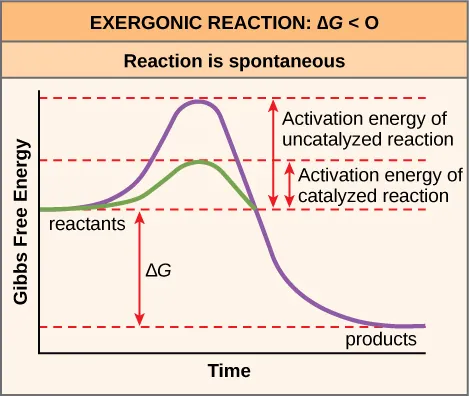
Free-energy diagram for an exergonic reaction comparing uncatalyzed and catalyzed pathways: the catalyst lowers the activation energy barrier while leaving the overall (reactants-to-products difference) unchanged. This reinforces that thermodynamic favorability (sign of ) and reaction rate are separate ideas. Source
They do not say how quickly the reaction occurs, only whether the products are thermodynamically preferred under standard conditions.
Reaction direction, scaling, and sign changes
Handling ΔG° algebraically is a frequent source of mistakes:
Reversing a reaction flips the sign: if the forward reaction has ΔG° < 0, the reverse has ΔG° > 0.
Multiplying a reaction by a factor multiplies ΔG° by the same factor, because ΔG° is extensive with respect to the amount of reaction as written.
Adding reactions adds their ΔG° values only if you also add the balanced equations correctly (species cancel consistently). This preserves the directionality of “favored” vs “unfavored” for the overall process under standard conditions.
Standard-conditions emphasis (what the statement does and doesn’t claim)
Why “under standard conditions” matters
The syllabus statement explicitly limits the interpretation to standard conditions. That means:
ΔG° tells you favorability for a specific reference setup.
A reaction with ΔG° < 0 is favored in that reference setup, but changing conditions (concentrations/pressures) can change the actual driving force.
Common interpretation pitfalls (sign-focused)
Confusing ΔG° (a standard-state criterion) with “the reaction always happens.”
Forgetting that the sign applies to the reaction as written (direction matters).
Dropping phase labels conceptually: changing a species from (g) to (aq), for instance, changes the thermodynamic situation and can change ΔG°.
FAQ
In AP usage, yes: “favoured under standard conditions” corresponds to spontaneous tendency under those conditions.
The word describes direction of change in free energy, not the speed.
Because $\Delta G^\circ$ is tied to the balanced equation as a whole.
Changing coefficients changes the stoichiometric “reaction unit,” so the per-reaction basis stays consistent.
Yes. If one equation is a scaled version of another, $\Delta G^\circ$ scales the same way.
If the equations are not actually equivalent overall, they describe different thermodynamic changes.
Typically via measured equilibrium data or electrochemical measurements, then converted to $\Delta G^\circ$ using established thermodynamic relationships.
Direct calorimetry alone does not generally provide $\Delta G^\circ$.
A larger magnitude indicates a stronger thermodynamic driving force under standard conditions.
However, magnitude still does not provide a timescale or mechanism for the process.
Practice Questions
(2 marks) Under standard conditions, Reaction A has and Reaction B has . State which reaction is thermodynamically favoured and which is unfavoured.
A favoured because . (1)
B unfavoured because . (1)
(5 marks) Explain what the sign of indicates about thermodynamic favourability under standard conditions. Include the meaning of , and describe what happens to if the reaction is reversed and if the equation is multiplied by 2.
indicates thermodynamically favoured under standard conditions. (1)
indicates thermodynamically unfavoured under standard conditions. (1)
indicates no net thermodynamic driving force under standard conditions. (1)
Reversing the reaction changes the sign of . (1)
Multiplying the equation by 2 doubles (extensive with reaction amount). (1)

