AP Syllabus focus: ‘Determine ΔG°rxn from formation values: ΔG°rxn = ΣΔGf°(products) − ΣΔGf°(reactants).’
Standard free energies of formation let you compute a reaction’s standard Gibbs free energy change without measuring the reaction directly. The key is careful use of stoichiometric coefficients, phases, and sign conventions.
Core idea: build reactions from formation data
Standard Gibbs free energy of formation,
Tabulated standard free energies of formation provide a reference free energy for making 1 mole of a substance from its elements under standard conditions, with the substance in its standard state at that temperature.
Standard Gibbs free energy of formation (): the Gibbs free energy change for forming 1 mol of a compound from its constituent elements in their standard states.
A reaction’s overall can be found by combining these formation processes algebraically, which leads to the products-minus-reactants relationship.
The equation you must use (with stoichiometry)
= standard Gibbs free energy change of reaction (typically of reaction as written)
= stoichiometric coefficient from the balanced equation (unitless)
= standard free energy of formation for a species in its stated phase/state (typically )
Use this only after the chemical equation is correctly balanced, because coefficients directly scale how much each species contributes to the total free energy change.
How to apply formation values correctly
Required workflow
Balance the overall reaction first (atoms and charge as appropriate).
Identify every species whose is needed, including correct phase labels (e.g., (s), (l), (g), (aq)).
Multiply each tabulated by its stoichiometric coefficient.
Add contributions for products, add contributions for reactants, then subtract: products minus reactants.
Report with an appropriate sign and units consistent with the data source.
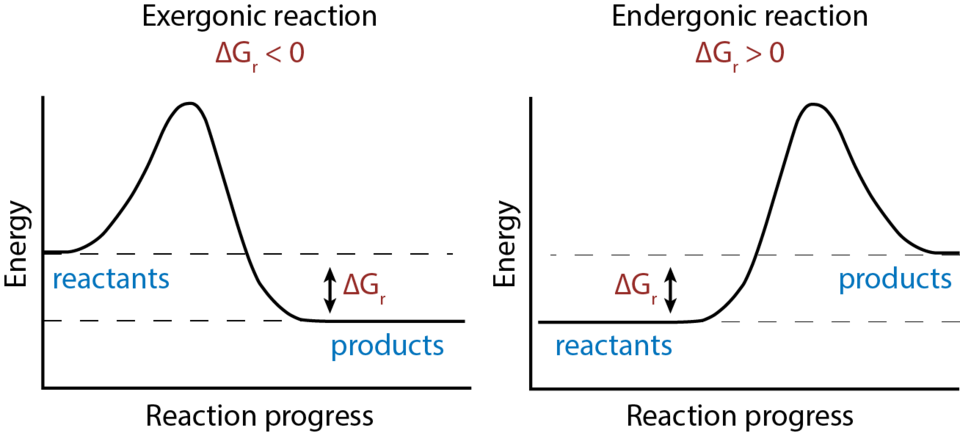
This reaction-coordinate diagram contrasts exergonic and endergonic reactions by plotting free energy versus reaction progress. The vertical difference between reactants and products corresponds to , making it clear why negative indicates products are thermodynamically favored and positive indicates reactants are favored. Source
What has (and why it matters)
Any element in its standard state has by definition (e.g., , , ).
This is a major simplification: such elements contribute nothing to the sum, even if they appear as reactants or products.
Be careful: an element not in its standard state is not zero (e.g., , , at 298 K).
State specificity (don’t mix phases)
depends on the species and its state:
and have different values.
and are different chemical descriptions, so they require different formation data.
Common pitfalls that change the sign or magnitude
Forgetting to multiply by coefficients (especially when coefficients are greater than 1).
Using the right compound but the wrong phase from a data table.
Dropping species that “seem small” (every term matters because the calculation is a sum).
Confusing (per mole of species formed) with (per mole of reaction as written).
FAQ
Values can vary due to updated reference data, different temperature assumptions (often 298 K), rounding, and consistency choices across thermodynamic datasets.
They rely on conventions (not direct measurement of isolated ions). A common convention sets $\Delta G_f^\circ[\text{H}^+(aq)] = 0$ at the reference temperature.
Only if the table explicitly matches the temperature. Otherwise you need additional temperature-dependent data; assuming constant values can introduce significant error.
You must obtain the value from another reliable thermodynamic source or use alternative thermodynamic pathways; you cannot infer it from the reaction equation alone.
Tabulated $\Delta G_f^\circ$ for (aq) species assumes a standard-state reference (idealised, typically 1 mol dm$^{-3}$ activity). Real solutions may deviate, especially at higher ionic strength.
Practice Questions
Given: , , . Calculate for .
Uses and (1)
Correct substitution with coefficients (1)
Correct result: (1)
For , given .
(a) Determine . (b) State which reactants have and justify.
(a) Applies products minus reactants with coefficients (1)
(a) Uses for products (1)
(a) Uses and (1)
(a) Correct result: (1)
(b) Justification: elements in their standard states have by definition (1)

