AP Syllabus focus: ‘If ΔH° and ΔS° are known at a temperature, calculate ΔG° directly using ΔG° = ΔH° − TΔS°.’
Gibbs free energy links enthalpy, entropy, and temperature into one criterion for thermodynamic driving force under standard conditions. This page focuses on using at a specified temperature.
When and why to use
You use this relationship when you are given (or can determine) standard enthalpy change and standard entropy change for a process and need the standard Gibbs free energy change at a particular temperature.
This approach is especially useful because:
Enthalpy captures heat/bond-energy effects.
Entropy captures energy dispersal and matter/energy distribution effects.
Temperature controls how strongly the entropy term contributes.
A key idea is that the entropy contribution is scaled by temperature, so the same matters more at higher .
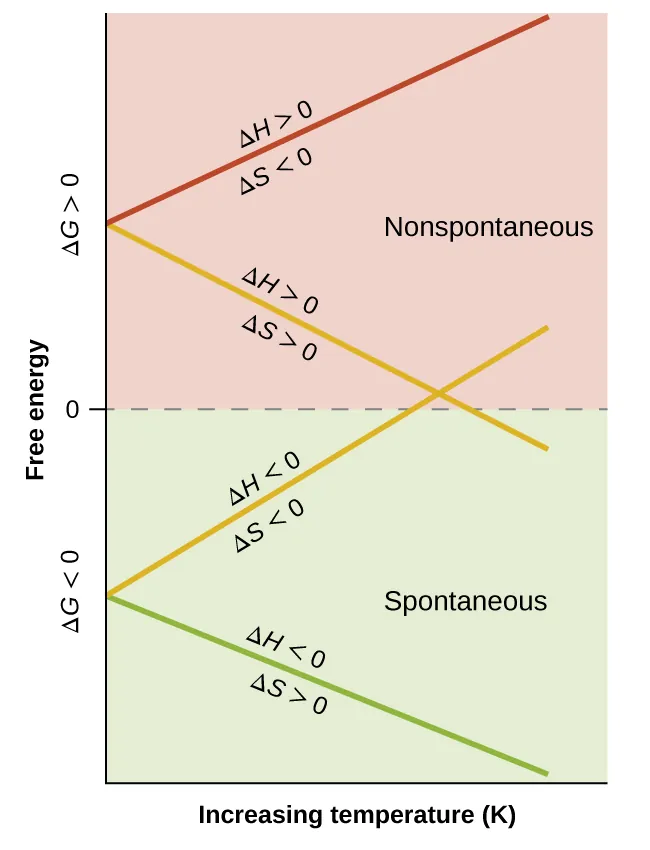
These lines show how varies with temperature for different sign combinations of and . The vertical position relative to indicates whether the process is thermodynamically favored, and the temperature where a line crosses zero corresponds to the point where (equilibrium for that process). Source
Key quantities (standard-state context)
Standard Gibbs free energy change, : The change in Gibbs free energy for a reaction/process when reactants and products are in their standard states; it combines enthalpy and entropy effects at a specified temperature.
Using is only meaningful when your and values refer to the same reaction as written (including physical states) and the same standard-state basis. In typical AP Chemistry usage, standard-state thermodynamic data are tabulated for a stated reference pressure and specified concentrations for solutes; the crucial point here is internal consistency among the values you use.
The equation and what each term means
= standard Gibbs free energy change (energy per mole of reaction as written), typically in or
= standard enthalpy change (energy per mole of reaction as written), typically in or
= absolute temperature in
= standard entropy change (energy per mole per kelvin), typically in
This equation is a bookkeeping tool: it combines an enthalpy “push” () with an entropy “push” () to determine the net free-energy change under standard conditions at that temperature.
Unit discipline (the most common source of mistakes)
Because and must be subtracted, they must be in the same energy units.
Practical rules:
Always use Kelvin for temperature: .
If is given in and is in , convert one so both match.
Commonly: convert to by dividing by 1000.
Keep track of the “per mole of reaction” basis:
If you scale the balanced equation, , , and therefore all scale by the same factor.
Interpreting the result at the given temperature (without overgeneralising)
After calculating at the stated temperature:
A negative indicates the reaction/process is thermodynamically favoured under standard conditions at that temperature.
A positive indicates it is not thermodynamically favoured under standard conditions at that temperature.
A value near zero suggests little driving force under standard conditions at that temperature.
This interpretation is tied to the specific temperature used in the calculation; changing changes the magnitude of the entropy contribution (), which can shift even when and are treated as constant over the temperature range.
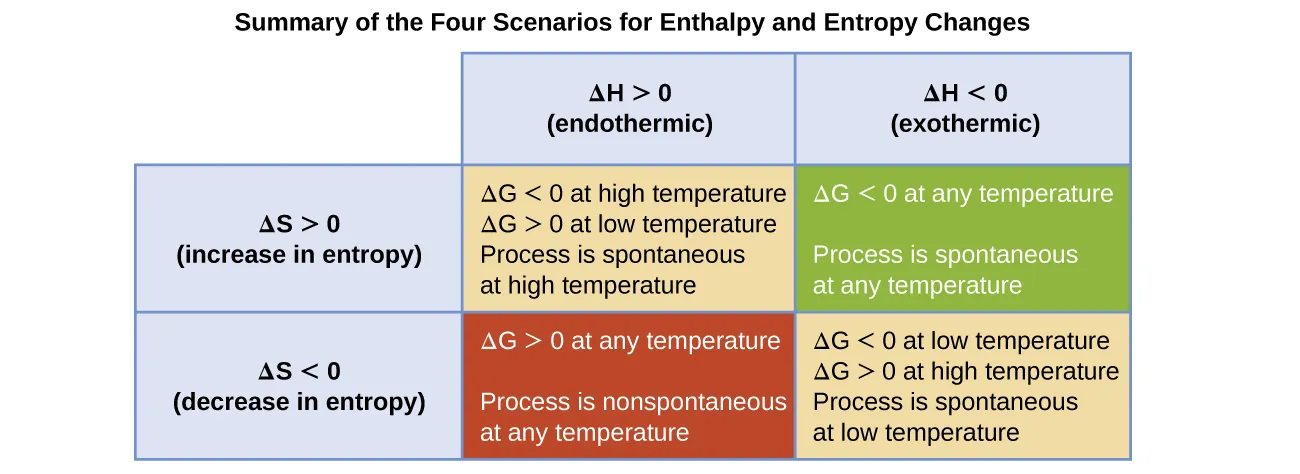
This table summarizes the four possible sign-combinations of and and the resulting temperature dependence of . It makes the role of the term explicit: increasing strengthens the entropy contribution and can flip the sign of when and have the same sign. Source
Assumptions and limitations students should state or recognise
Using cleanly at a “given temperature” rests on assumptions that are usually acceptable in AP-level problems but still worth knowing:
and are treated as temperature-independent over the range considered (often around 298 K unless stated otherwise).
The computed value is standard free energy, not the free energy change for arbitrary (nonstandard) concentrations/pressures.
Physical states matter: , , and depend on whether substances are (s), (l), (g), or (aq).
What “at a given temperature” means operationally
To apply the syllabus skill precisely:
Use the temperature provided (not automatically 298 K).
Convert that temperature to K.
Compute the entropy contribution as with consistent units.
Subtract that contribution from to obtain for that temperature under standard conditions.
FAQ
Because both $ \Delta H^\circ $ and $ \Delta S^\circ $ can vary with temperature via heat capacities.
For large temperature changes, treating them as constants can noticeably shift $T\Delta S^\circ$ and therefore $ \Delta G^\circ $.
It can be viewed as the temperature-scaled energy dispersal contribution.
In many treatments, it corresponds to the portion of energy change associated with entropy that affects how much energy is available to drive change under standard conditions.
Yes. The equation requires absolute temperature because entropy has units per kelvin.
Using Celsius would shift $T$ by 273.15 and give an incorrect magnitude for $T\Delta S^\circ$ and thus $ \Delta G^\circ $.
A negative $ \Delta S^\circ $ makes $T\Delta S^\circ$ negative, so subtracting it becomes addition: $ \Delta G^\circ = \Delta H^\circ + |T\Delta S^\circ| $.
This means entropy can oppose the process more strongly as temperature rises.
Match the precision of your inputs after unit conversions.
If $T$ is given as an integer kelvin and $ \Delta S^\circ $ has three significant figures, keep intermediate values (like $T\Delta S^\circ$) unrounded, then round $ \Delta G^\circ $ at the end.
Practice Questions
Q1 (3 marks) A reaction has and at . Calculate at .
Converts to (or converts to J): 1 mark
Calculates (or equivalent in J): 1 mark
Calculates (with correct sign and units): 1 mark
Q2 (6 marks) For a process, and . (a) Calculate at . (3 marks)
(b) Calculate at . (2 marks)
(c) State, based on your values, which temperature gives the more thermodynamically favoured standard free-energy change. (1 mark)
Unit conversion for entropy: (or consistent alternative): 1 mark
at 298 K: : 1 mark
, correct sign/units: 1 mark (b)
at 350 K: : 1 mark
, correct sign/units: 1 mark (c)
Identifies as more favoured because is more negative (negative vs positive): 1 mark

