AP Syllabus focus: ‘ΔG° applies when all reactants and products are in standard states: pure substances, 1.0 M solutions, or gases at 1.0 atm (or 1.0 bar).’
Thermodynamic quantities are only comparable when measured under clearly defined reference conditions. This page explains what standard states mean in AP Chemistry and how to interpret ΔG° as a standard-condition free energy change.
What ΔG° Means (and What It Does Not)
ΔG° is the Gibbs free energy change for a reaction as written when every reactant and product is in its standard state. It is a property of the reaction under those reference conditions, not a guarantee about any real mixture you might have in a beaker.
Standard state: the reference condition of a substance used to define standard thermodynamic quantities; for AP Chemistry this means a pure substance, a 1.0 M solution, or a gas at 1.0 atm (or 1.0 bar).
Because standard states are reference conditions, ΔG° is best interpreted as: “How thermodynamically favorable is the reaction if everything starts (and is compared) in standard states?”
Standard State vs. Standard Conditions
Students often blend these ideas, but they are not identical.
Standard state specifies the reference state for each species (pure, 1.0 M, or 1 atm/1 bar).
Standard conditions informally means “measured using standard states” (and often, but not always, at 298 K). The key point for ΔG° is the standard state requirement.
Standard States You Must Recognise
AP Chemistry uses the following standard states (as in the syllabus statement).
Pure Substances (Elements and Compounds)
For a pure solid or pure liquid, the standard state is the pure substance in its most stable form at the stated conditions.
Examples of what counts as standard state:
pure liquid water, H₂O(l)
solid sodium chloride, NaCl(s)
“Pure” matters: mixtures (like ethanol in water) are not pure-substance standard states.
Solutions at 1.0 M
For a dissolved species, the AP standard state is 1.0 M.
This is a reference point for solutes (e.g., H⁺(aq), Cl⁻(aq), Fe²⁺(aq)).
When concentrations differ from 1.0 M, the situation is nonstandard, so ΔG is not necessarily equal to ΔG°.
Gases at 1.0 atm (or 1.0 bar)
For gases, the standard state is a pressure of 1.0 atm (or 1.0 bar, depending on convention).
This is why you may see standard thermodynamic tables and problems reference either 1 atm or 1 bar.
Real gases can deviate from ideality, but AP interpretation typically treats the listed standard state as the reference without extra corrections.
Reaction Definition and the “As Written” Idea
ΔG° depends on the balanced chemical equation and the physical states listed. If you change coefficients, phases, or the way the reaction is written, you change ΔG° because you are defining a different process.
A compact way to express what ΔG° represents is:
= standard Gibbs free energy change for the reaction as written (typically kJ/mol reaction)
= sum of standard Gibbs free energies of the products in their standard states (kJ/mol reaction)
= sum of standard Gibbs free energies of the reactants in their standard states (kJ/mol reaction)
This equation highlights the core meaning: ΔG° compares standard-state reactants to standard-state products.
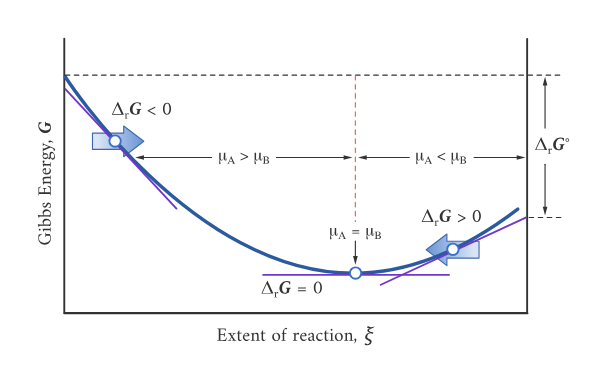
A graph of Gibbs energy versus extent of reaction showing that the equilibrium composition corresponds to the minimum in . The labeled regions connect sign to direction: when the slope is negative the forward reaction lowers (), and at the minimum () the system is at equilibrium. Source
How to Use ΔG° Correctly in Interpretation
To stay aligned with what ΔG° is actually telling you, keep these rules in mind:
ΔG° applies only when all species are in their standard states (pure substances, 1.0 M, 1.0 atm/1.0 bar).
ΔG° is tied to the reaction as written; changing the chemical equation changes ΔG°.
ΔG° is a thermodynamic reference value; it describes a standard-state comparison, not the necessarily the free energy change under arbitrary lab concentrations and pressures.
FAQ
No. Standard state specifies reference pressure/concentration/purity; it can be defined at a stated temperature.
In many courses, data tables are at 298 K, which is why 298 K is commonly associated with “standard”.
1 bar is the modern IUPAC standard pressure; older conventions often use 1 atm.
AP Chemistry typically accepts either, as long as you are consistent with the data provided.
If water is a reactant/product as a pure liquid, its standard state is pure H$_2$O(l).
If water is acting as the solvent, it is often treated as a pure liquid reference in thermodynamic conventions, even though it is in a mixture.
Strictly, thermodynamics uses “activity” rather than raw concentration for real solutions.
AP problems usually approximate activity with concentration, so 1.0 M serves as the practical reference state.
It provides a simple, reproducible reference point for comparing dissolved species.
Historically, alternative standards (like 1 molal) exist, but AP uses 1.0 M for consistency with common data tables.
Practice Questions
(2 marks) State the standard state used in AP Chemistry for: (a) an aqueous solute, and (b) a gas.
(a) 1.0 M solution / concentration of 1.0 mol dm (1)
(b) 1.0 atm (or 1.0 bar) gas pressure (1)
(5 marks) A student claims that for a reaction applies whenever the temperature is 298 K, even if reactant concentrations are not 1.0 M and gas pressures are not 1 atm. Assess this claim using the meaning of standard state and .
Identifies that refers to a reaction when all species are in their standard states (1)
States standard state for solutions is 1.0 M (1)
States standard state for gases is 1.0 atm (or 1.0 bar) (1)
States standard state for pure solids/liquids is the pure substance (1)
Concludes the claim is incorrect because 298 K alone does not ensure standard states; nonstandard concentrations/pressures mean in general (1)

