AP Syllabus focus: ‘Include ionic charge and multi-electron half-reactions when converting between moles of electrons, charge passed, and chemical change.’
Electrolysis calculations hinge on treating electrons as stoichiometric reactants. The main challenge is correctly accounting for ion charge and multi-electron half-reactions so charge passed, moles of electrons, and chemical amounts remain consistent.
Core idea: electrons link electricity to chemical change
In any electrolytic process, the measured electrical quantity is charge delivered to the cell, but the chemical bookkeeping must be done in moles of electrons and then in moles of species using a balanced half-reaction. The key step in difficult problems is identifying how many electrons correspond to producing (or consuming) one mole of a substance.
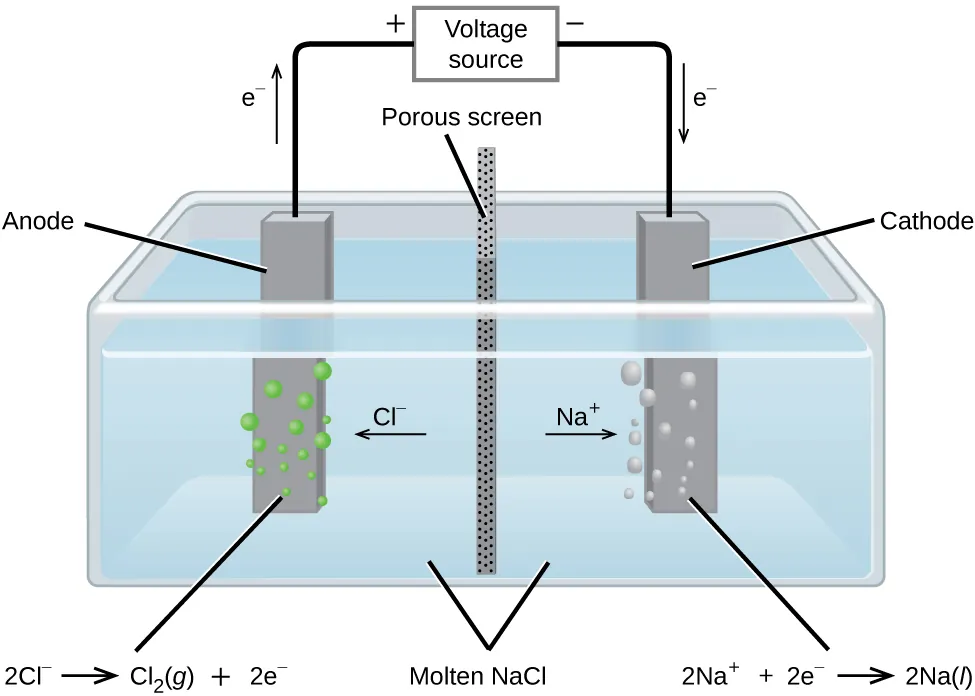
Schematic of an electrolytic cell for molten showing ion migration, electrode polarity, and electron flow in the external circuit. The diagram pairs each electrode with its balanced half-reaction, making the electron coefficients (and therefore electron-to-chemical mole ratios) visually explicit. This helps connect measured charge to and then to moles of products formed at each electrode. Source
Faraday constant and charge–electron conversion
Faraday constant (F): The charge carried by 1 mole of electrons, approximately .
Because electrolysis problems may involve ions with charges like or , the “1 mole of electrons” step must be followed by a stoichiometry step that reflects the ion’s charge and the balanced half-reaction.
= total charge passed (coulombs, C)
= moles of electrons transferred (mol )
= Faraday constant ()
This equation gives moles of electrons, not moles of ions or product. The chemical amount comes from the electron coefficients in the relevant half-reaction.
Accounting for ion charge: electrons per ion
For many simple electrode processes, the ion’s charge directly indicates the minimum number of electrons needed per ion (after proper balancing).
Cations reduced to metals (electron gain equals ionic charge magnitude)
A metal ion with charge typically reduces to the metal by gaining electrons.
General pattern:
Interpretation: 1 mol requires mol for complete reduction.
Examples of electron counts (conceptual):
requires 2 e− per Cu deposited
requires 3 e− per Al deposited
This “electrons per ion” factor is what converts between (from charge) and moles of metal deposited.
Anions oxidised to neutral species (electrons produced depend on diatomic formation)
For anions that form neutral elements at the anode, you must balance both charge and atoms, which often introduces diatomic products and changes the electron count per mole of product.
Common pattern for halides:
Meaning:
2 mol e− correspond to 1 mol formed
1 mol e− corresponds to 1 mol consumed
Oxygen formation from oxide/hydroxide similarly requires careful balancing, so you should rely on the balanced half-reaction rather than guessing from charge alone.
Multiple-electron half-reactions: using stoichiometric coefficients correctly
Many electrode reactions transfer more than 1 electron per mole of chemical species. The correct approach is always:
Write the relevant half-reaction for the electrode process
Ensure it is balanced for atoms and charge
Use the electron coefficient as the conversion factor between moles of electrons and moles of substance produced/consumed
The electron stoichiometric factor (what “n” really means in practice)
In AP Chemistry electrolysis contexts, the critical number is the moles of electrons per mole of target species, taken directly from the balanced half-reaction.
If the half-reaction is :
2 mol e− transfer per 1 mol B formed
If the half-reaction is :
2 mol e− transfer per 1 mol formed
equivalently, 1 mol e− per 1 mol consumed
This is where students often lose accuracy: they convert charge to moles of electrons correctly, but then assume “1 mol e− makes 1 mol product” even when the half-reaction shows otherwise.
Consistency checks and common pitfalls (specific to charge and multi-electron transfer)
Use these quick checks to avoid sign and factor errors:
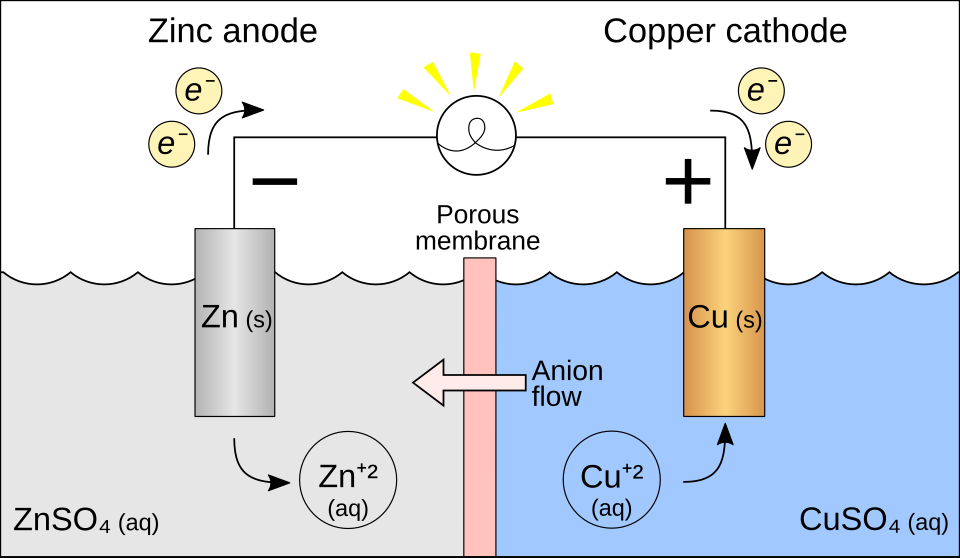
Labeled schematic of a copper–zinc electrochemical cell highlighting electron flow through the wire and ion movement that maintains electrical neutrality. Even though the figure is drawn for a galvanic setup, the same definitions still apply in electrolysis: oxidation occurs at the anode and reduction occurs at the cathode. Seeing the separated charge-transport paths helps students distinguish “electrons balance charge in half-reactions” from “ions move in solution to prevent charge buildup.” Source
Charge conservation check: electrons are not mass; they only balance charge. If the left side of a half-reaction has net positive charge, electrons should appear on the left (reduction). If net negative, electrons should appear on the right (oxidation).
Magnitude check for ions: higher-charge cations (e.g., ) must involve more electrons per ion than cations if they form neutral metals.
Diatomic products: when elements form as , the electron count per mole of product is usually double what you might expect from a single ion.
Coefficient discipline: the coefficients in the balanced half-reaction are the only valid mole ratios linking electrons to chemical change; never “simplify away” coefficients if it changes the species amount you are asked about.
FAQ
Electron transfer must exactly neutralise the ionic charge.
A $3+$ charge means the species is missing three electrons relative to the neutral atom, so reduction to the element requires gain of three electrons per ion.
Use the balanced half-equation as the conversion.
For chloride oxidation, $2\text{Cl}^- \rightarrow \text{Cl}_2 + 2\text{e}^-$ shows that 2 mol e− correspond to 1 mol $\text{Cl}_2$, not 2 mol.
No. It often is for simple metal-ion deposition, but not universally.
Polyatomic ions, oxygen-containing species, and cases involving diatomic formation require full half-equation balancing to determine the electron coefficient.
Any scaled version is acceptable, but your mole ratios must match the version you use.
If you double every coefficient, you must also double the electron coefficient; the conversion from electrons to product must stay consistent.
Look for species that naturally occur as pairs (e.g., $\text{H}_2$, $\text{Cl}_2$, $\text{O}_2$).
If the product is diatomic and you treated it as monatomic in stoichiometry, your calculated moles of product will typically be off by a factor of two.
Practice Questions
Question 1 (2 marks) Molten is electrolysed. Write the cathode half-equation and state how many moles of electrons are required to produce 1 mol of Mg.
Cathode half-equation: (1 mark)
2 mol required per 1 mol Mg (1 mark)
Question 2 (6 marks) A solution containing ions is electrolysed to plate aluminium metal. A total charge of passes through the circuit. (a) Write the cathode half-equation. (1 mark) (b) Determine the moles of electrons transferred. (2 marks) (c) Determine the moles of aluminium deposited, clearly showing how the ion charge affects the stoichiometry. (3 marks)
(a)
(1 mark)
(b)
Uses (1 mark)
Correct substitution and value: (1 mark)
(c)
States stoichiometric link: 3 mol per 1 mol Al from the half-equation (1 mark)
Correct setup: (1 mark)
Correct value: (1 mark)

