AP Syllabus focus: ‘Use current, charge, and time relationships (I = q/t) to analyze charge flow in electrochemical cells.’
Electrolysis problems begin with tracking how much charge moves through a circuit. Faraday’s law connects measurable electrical quantities—current and time—to total charge, which represents the extent of electron flow in the cell.
Core Electrical Quantities in Electrochemistry
Current and charge in a cell
In an electrochemical setup, oxidation and reduction are forced or allowed to occur by moving electrons through an external wire.
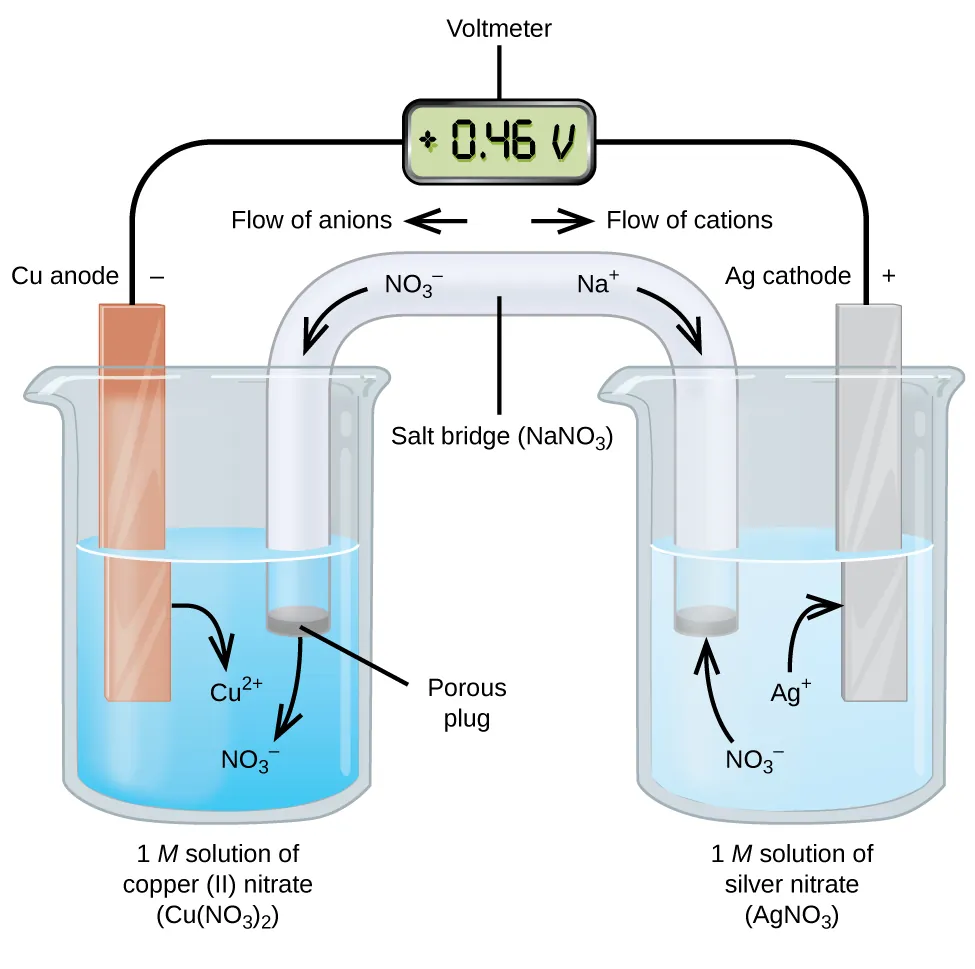
Diagram of a standard galvanic cell showing separated half-cells connected by an external wire and a salt bridge. The labels identify anode vs. cathode and explicitly indicate the direction of electron flow through the wire and ion migration through the salt bridge, reinforcing what “current in a cell” physically represents. Source
What you can measure directly is the current passing through the circuit over some time, which determines the total charge transferred.
A key idea for AP Chemistry is that charge transfer is cumulative: the longer the current runs (at a given value), the more charge passes through the cell.
Current (I): the rate at which electric charge flows past a point in a circuit; measured in amperes (A).
Current describes “how fast” charge is moving, not the amount of reaction by itself.
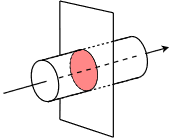
Schematic defining electric current as charge passing a cross-section of a wire per unit time. The figure visually separates the ideas of “amount of charge transferred” () and “how quickly it passes” (current, ), which is exactly the conceptual distinction needed before applying in electrolysis problems. Source
Charge (q): the total quantity of electricity transferred; measured in coulombs (C).
Time and unit consistency
Electrochemistry calculations require consistent SI units.
Time (t) must be in seconds (s) when using amperes and coulombs.
Convert common lab times:
minutes to seconds by multiplying by 60
hours to seconds by multiplying by 3600
Convert current units when needed:
mA to A by dividing by 1000
Faraday’s Law Basics: Linking I, q, and t
The fundamental relationship
The AP Chemistry relationship you use to analyse charge flow is:
= current, in
= charge transferred, in
= time, in
Rearranging this relationship lets you find any one quantity from the other two (for example, finding charge from a measured current over a known time).
Interpreting what the equation means in a cell
Using in electrochemical cells supports the following reasoning:
At fixed current, charge is proportional to time: doubling the time doubles the charge passed.
For a shorter time, achieving the same charge requires a larger current.
Charge depends on current and time, not directly on the identity of the electrolyte; the chemical details determine what that charge accomplishes chemically, but the electrical bookkeeping starts with , , and .
Practical Notes for AP-Style Problem Setups
What’s usually assumed
Most AP-style electrolysis prompts imply:
a steady (constant) current during the interval stated
the circuit is operating normally so current is actually flowing for the full time
Common pitfalls to avoid
Mixing units (e.g., using minutes with amperes without converting to seconds)
Dropping units: correct reporting includes A, s, and C where appropriate
Confusing “current” with “charge”:
current is a rate
charge is a total amount accumulated over time
What you are measuring conceptually
In an electrochemical cell, electrons are the mobile charge carriers in the wire. Treating the measured current as electron flow allows you to quantify total charge transfer during electrolysis, which is the starting point for later stoichiometric connections.
FAQ
Voltage affects how hard the circuit “pushes” charge and what reactions can occur, but $I = q/t$ is purely a definition linking rate of charge flow to time.
An ammeter must be placed in series so the same current passes through it. Placing it in parallel can give incorrect readings or damage the meter.
Charge is quantised. The number of electrons corresponding to a charge $q$ is $N = q/e$, where $e \approx 1.602\times10^{-19}\ \mathrm{C}$ per electron.
Typically it quickly stops, because charge builds up in the half-cells. The salt bridge allows ionic movement to maintain electroneutrality so sustained current is possible.
In chemistry problem setups, $q$ is usually treated as a positive amount of charge passed. Sign conventions (electron charge being negative) matter mainly when analysing direction, not total magnitude.
Practice Questions
(2 marks) A constant current of passes through an electrolytic cell for minutes. Calculate the total charge transferred.
(1) Converts time to seconds and uses
(1) Correct value with unit:
(5 marks) A student runs an electrolysis experiment at for , then at for a further minutes.
(a) Calculate the charge transferred in each interval. (3 marks)
(b) Calculate the total charge transferred. (2 marks)
(1) Uses for interval 1:
(1) Converts minutes to
(1) Uses for interval 2:
(1) Sums charges:
(1) Correct units shown for charges (C)

