AP Syllabus focus: ‘Determine mass deposited on or removed from an electrode (as in electroplating) using Faraday’s laws and electron accounting.’
Electrolysis can change electrode mass because electrons drive reduction at one electrode and oxidation at the other. Faraday’s laws connect electrical charge to moles of electrons, letting you predict plating or dissolution quantitatively.
Mass changes at electrodes: the core idea
When a species is reduced onto an electrode, that electrode gains mass; when an electrode material is oxidised into solution, that electrode loses mass. In both cases, the link between electricity and chemical change is electron counting.
Electroplating vs dissolution
Electrode mass changes depend on which half-reaction occurs at that surface.
Electroplating: the process of depositing a solid metal onto an electrode surface by reducing metal ions from solution (or melt) to metal atoms at the cathode.
Electroplating typically involves a metal cation (like , , ) gaining electrons and becoming a metal solid, increasing the electrode’s mass.
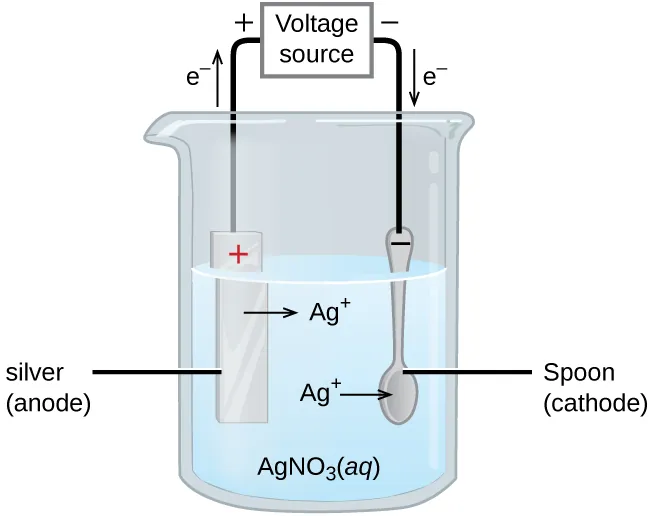
Electroplating cell diagram showing a silver anode dissolving to form in solution while is reduced and deposited as Ag(s) onto the spoon cathode. The labeled electron flow through the external circuit highlights that reduction occurs at the cathode (mass gain) and oxidation occurs at the anode (mass loss). Source
Dissolution is the reverse: a metal solid becomes ions, decreasing the electrode’s mass.
Using Faraday’s laws to connect charge to mass
Faraday’s laws state that the amount of chemical change is proportional to the total charge passed. The essential workflow is: current and time → charge → moles of electrons → moles of substance → mass change.
A key point is that the balanced half-reaction tells you how many electrons per mole of metal (or other species) are transferred.
= charge passed, C
= current, A ()
= time, s
= moles of electrons transferred, mol
= Faraday constant,
= mass deposited or dissolved, g
= molar mass of deposited/dissolved species,
= electrons transferred per formula unit (from the half-reaction), mol /mol substance
This calculation only works when you correctly identify from the relevant half-reaction at that electrode.
Writing the half-reaction to get the electron ratio
Mass changes are tied to the electrode reaction:
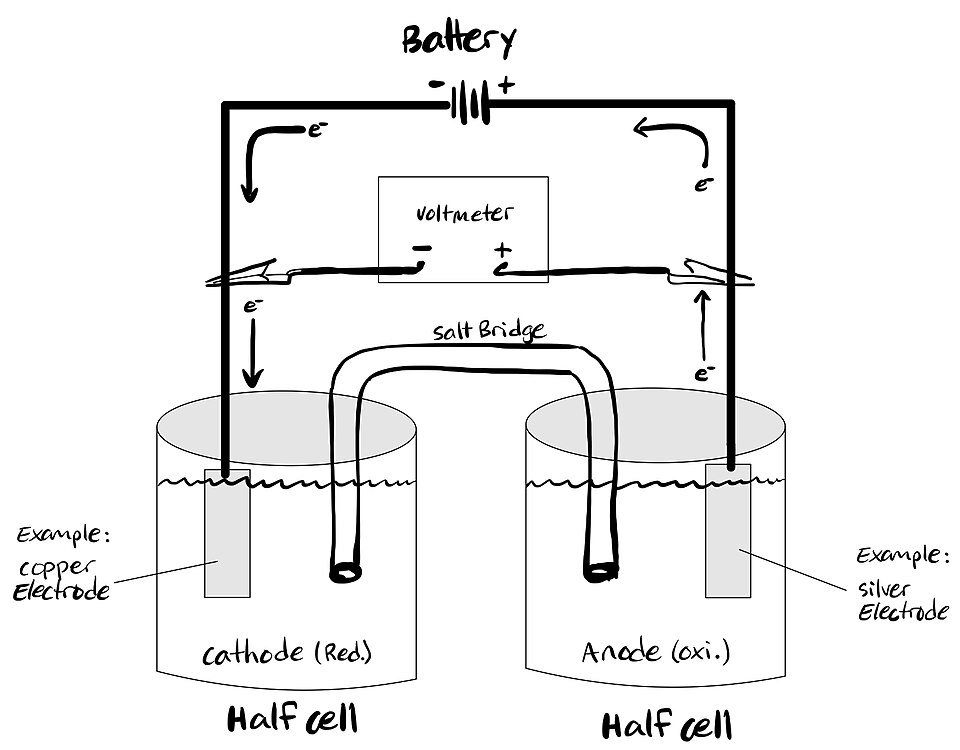
Diagram of an electrolytic cell with labeled half-reactions, showing oxidation at the anode and reduction at the cathode driven by an external power source. This kind of schematic is useful for mapping each electrode’s half-reaction to the electron count , which is the key link between charge passed and predicted mass change. Source
Metal deposition at the cathode (mass increases)
General form:
The electrode gains mass because solid metal forms on the surface.
The moles of metal deposited equal .
Metal dissolution at the anode (mass decreases)
General form:
The electrode loses mass because metal atoms leave the solid as ions.
The moles of metal dissolved equal .
What the balance of electrons implies for both electrodes
In an electrolytic setup, the same current passes through both electrodes, so the same total moles of electrons are transferred at the anode and cathode. However, mass changes can differ if:
different substances react at the two electrodes, or
differs (e.g., needs 1 , but needs 2 ).
Common pitfalls that change the predicted mass
Using the overall (molecular) equation instead of the electrode half-reaction to find .
Forgetting to convert time to seconds when using .
Confusing the sign of mass change:
cathode (reduction): typically mass increases for metal-ion plating
anode (oxidation): typically mass decreases for metal dissolution
Ignoring that the deposited/dissolved species must match the half-reaction (use the correct molar mass ).
FAQ
If side reactions also consume electrons (e.g., hydrogen evolution), not all charge produces metal.
You can account for this by multiplying the theoretical mass by the efficiency (as a decimal).
Deposited metal can be powdery and fall off, or the surface may not adhere well.
Mechanical loss reduces measured mass without changing the charge passed.
Use the actual cathode half-reaction and the molar mass of the solid formed.
If multiple solids can form, you need additional information (conditions or observations) to decide which reaction dominates.
The calculated mass assumes a single metal with a single oxidation state.
Real dissolution may preferentially remove one component, so the effective $M$ and $z$ can differ from a pure-metal assumption.
Yes. Gas bubbles can cling to the electrode, making the apparent mass higher until they detach.
Careful rinsing and drying procedures are needed to obtain a reliable mass change.
Practice Questions
Q1 (3 marks) A nickel object is electroplated in a solution using a current of for . Calculate the mass of Ni deposited. (, )
Uses with to find (1)
Finds (1)
Uses so and (1)
Q2 (6 marks) Copper is purified by electrolysis using copper electrodes in . A current of passes for . (a) Calculate the mass change of the cathode.
(b) Calculate the mass change of the anode.
(c) Write the half-equations at both electrodes.
(, )
Converts time: and calculates (1)
Finds (1)
Uses for copper and finds (1)
Cathode mass change: (allow 1.32–1.34 g) (1)
Anode mass change: (allow 1.32–1.34 g) (1)
Half-equations: cathode ; anode (1)

