AP Syllabus focus: ‘Use Faraday’s laws to connect current and time elapsed to the amount of material produced or consumed in a redox process.’
Electrolysis links electrical measurements to chemical change. By tracking electrons transferred in a balanced half-reaction, you can convert between moles of product formed, total charge passed, and the current or time required.
Core idea: chemical amount ↔ electrons ↔ charge ↔ current/time
In electrolysis, the amount of substance produced or consumed is determined by how many electrons are forced through the cell.
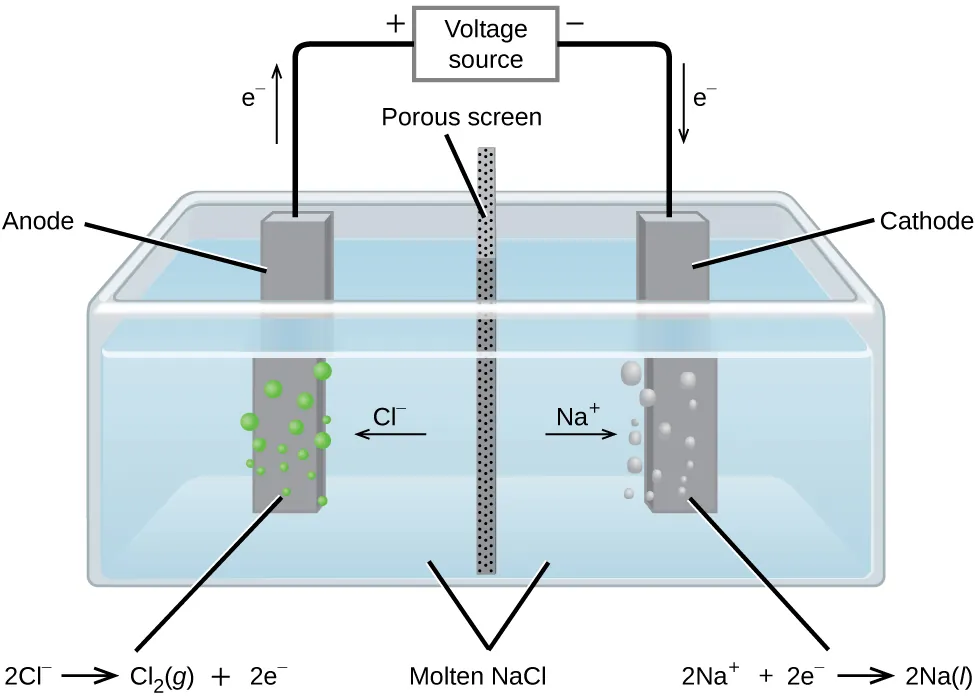
Simplified electrolytic cell diagram for molten NaCl electrolysis, labeling the anode/cathode, the voltage source, electron flow in the wire, and ion migration in the melt. It reinforces the core idea that the external circuit controls how many electrons are delivered to the redox process at each electrode. Source
The key is to treat electrons as a stoichiometric “reactant” that connects the circuit to the redox chemistry.
Key electrical quantities
Current (I): The rate of charge flow through a circuit, measured in amperes (A), where .
Current tells you how fast charge is moving. Time tells you how long it moves. Together they determine total charge delivered to the electrochemical reaction.
Faraday constant (F): The magnitude of charge carried by 1 mole of electrons, .
Faraday’s constant converts between moles of electrons and coulombs of charge, which is what you measure electrically.
Essential relationships (Faraday’s law tools)
= total charge passed (C)
= current (A)
= time (s)
= moles of electrons transferred (mol )
= Faraday constant ()
These two relationships are typically chained: chemistry , then , then or .
The stoichiometry bridge: from substance to electrons
To connect amount of substance to charge, you must use a balanced redox equation (often a half-reaction at an electrode).
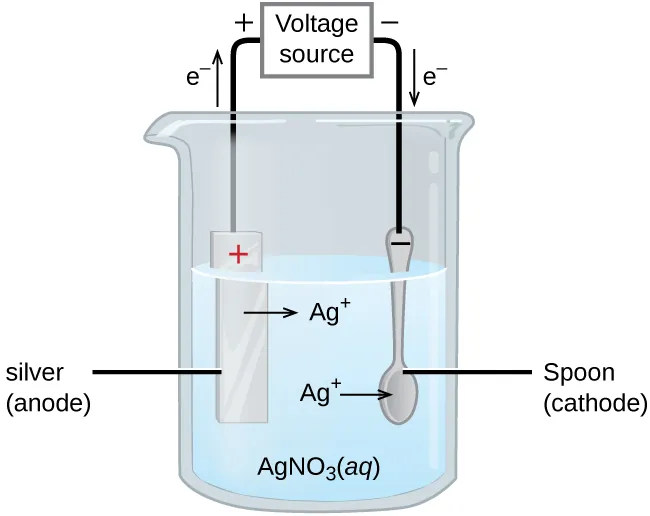
Electroplating cell diagram showing a metal anode, an object cathode, and a voltage source driving electrons so that metal ions deposit on the cathode. This visual directly supports writing correct half-reactions (oxidation at the anode, reduction at the cathode) before converting or . Source
The coefficient on sets the electron-per-mole ratio.
How to set up the conversion (no calculation steps)
Identify the species whose amount is given or requested (mass, moles, or sometimes volume for a gas).
Write the relevant balanced half-reaction showing electrons explicitly.
Use the half-reaction coefficients to relate:
moles of species moles of
Convert moles of to charge with .
Use to solve for the unknown:
solve for time if current is known
solve for current if time is known
Common “amount” formats you may need to convert first
If given mass, convert to moles using molar mass before using the half-reaction ratio.
If given moles directly, go straight to the electron ratio.
If given a gas amount (less common here), ensure the problem provides a way to get moles, then proceed through electrons.
Units and operational checkpoints
Keeping units consistent prevents most errors.
Use seconds for time when applying (convert minutes/hours to seconds).
Current in amperes already equals .
Charge comes out in coulombs.
must be in so that comes out in moles.
Chemical reasonableness checks
More electrons per mole (larger coefficient) means more charge is required for the same amount of product.
For a fixed current, needing more charge implies a longer time.
For a fixed time, needing more charge implies a larger current.
Where students most often go wrong
Not including the electron coefficient from the balanced half-reaction.
Using the wrong half-reaction (mixing anode and cathode chemistry for the species of interest).
Forgetting to convert time units to seconds before using .
Treating as of ions rather than per mole of electrons.
FAQ
Use a half-reaction when the problem specifies what happens at a particular electrode or names a product formed at the anode/cathode.
A full equation is fine only if it still shows the same electron-to-species ratio for the substance you are tracking.
Rewrite the appropriate half-reaction so that electrons appear explicitly.
Then read $n$ directly as the coefficient on $e^-$ per mole of the species formed/consumed.
Scaling a balanced half-reaction by a factor scales both moles of species and moles of electrons by the same factor.
Charge depends on the electron-to-species ratio, which remains consistent when scaled correctly.
Not directly. You would need the total charge passed, often found from a current–time graph (area under the curve).
Once $q$ is known, you can still use $q=n_{e^-}F$ to connect to chemical amount.
It means all measured charge goes into the intended redox reaction (no side reactions).
If efficiency is less than 100%, you would multiply the theoretical charge (or product) by the efficiency fraction.
Practice Questions
Question 1 (1–3 marks) A constant current of runs for . Calculate the total charge passed, in coulombs.
Converts time to seconds: (1)
Uses (1)
Correct charge: (1)
Question 2 (4–6 marks) In an electrolytic process, of is produced at the cathode via . The current is . Calculate the time required.
Uses half-reaction to find moles of electrons: (2)
Uses with (1)
Calculates charge: (1)
Uses (1)
Calculates time: (1)

