AP Syllabus focus: ‘Many observable processes are reversible, such as evaporation/condensation, gas adsorption/desorption, and salt dissolution/precipitation; key chemical examples include proton transfer (acid–base) and electron transfer (redox).’
Reversibility is the foundation of equilibrium: many systems can proceed in both forward and reverse directions. Recognising what makes a process reversible helps you predict when mixtures of reactants and products can persist.
What “reversible” means in chemistry
A process is reversible when products can reform reactants under the same overall conditions, so the system can move in either direction depending on what is present.
Reversible process: A physical or chemical change that can proceed in both forward and reverse directions, so reactants and products can interconvert within the same system.
Reversibility is easiest to discuss for a closed system where matter does not escape; otherwise, one direction may dominate simply because a species leaves the system (for example, a gas venting).
= Stoichiometric coefficients (unitless)
The double arrow emphasises that both directions are possible; it does not imply equal amounts or equal speeds at all times.
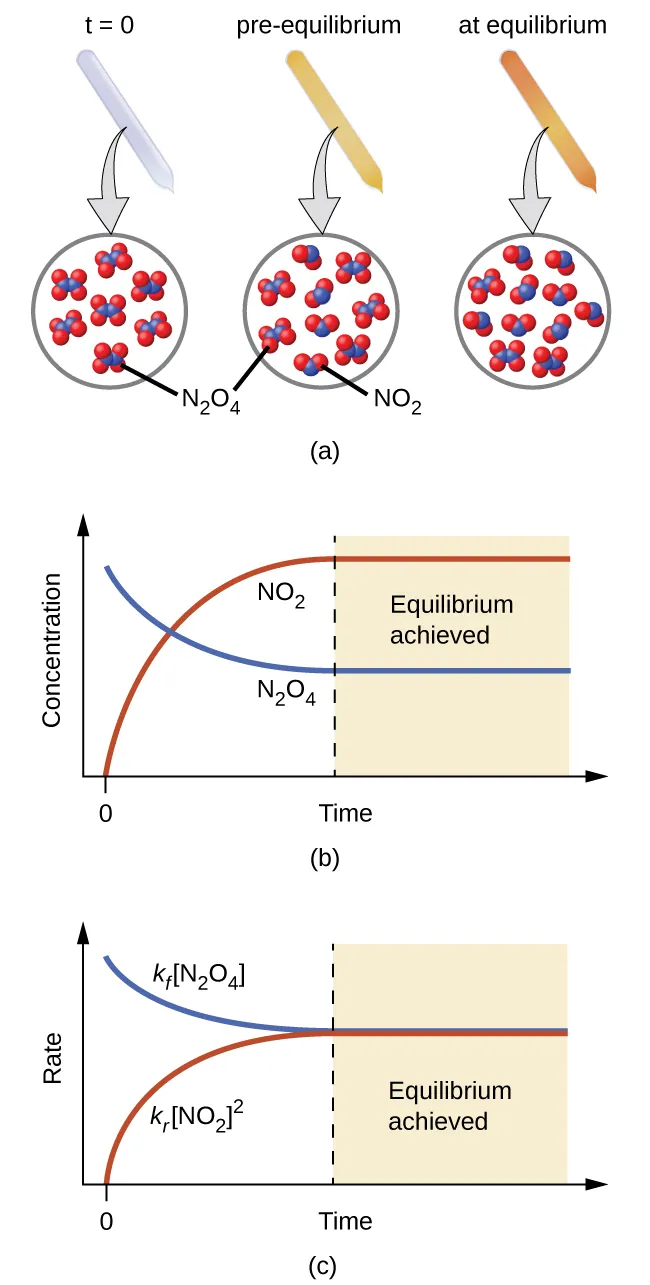
A reversible gas-phase system () is shown moving toward equilibrium, with particle diagrams tied to concentration–time and rate–time graphs. The plots highlight that equilibrium occurs when forward and reverse reaction rates become equal, even though particles continue interconverting. This is a concrete visual example of why the equilibrium arrow indicates bidirectionality, not equal amounts at every moment. Source
Reversible physical processes (observable examples)
Physical changes are often reversible because particles can move between phases or locations without changing identity. The syllabus highlights these common cases.
Evaporation and condensation
Evaporation: liquid particles escape into the gas phase.
Condensation: gas particles return to the liquid phase.
In a sealed container, both can occur because:
Some liquid molecules always have enough kinetic energy to escape.
Some gas molecules collide with the liquid surface and are captured.
The reverse process becomes more important as more vapour accumulates, making the overall change potentially reversible.
Gas adsorption and desorption
Adsorption: gas particles attach to a solid surface (often via intermolecular attractions).
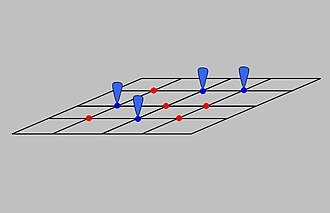
This schematic depicts a surface with equivalent adsorption sites, distinguishing occupied sites (adsorbed particles) from unoccupied sites (available sites). The diagram supports the kinetic picture of reversibility: adsorption populates surface sites while desorption vacates them, allowing a dynamic balance to be established under suitable temperature and pressure conditions. It also connects naturally to the idea of surface coverage used in the Langmuir model. Source
Desorption: adsorbed particles leave the surface and re-enter the gas phase.
Reversibility is promoted when:
The surface is not permanently changed (no strong bonding that locks particles in place).
Temperature and pressure allow both attachment and escape to occur.
Salt dissolution and precipitation
Dissolution: ions leave an ionic solid and become solvated in water.
Precipitation: dissolved ions re-form the ionic solid.
In contact with water, a slightly soluble salt can show both directions:
Ions continually detach from the crystal surface.
Dissolved ions can collide with the crystal and reattach.
This is why saturated solutions can contain undissolved solid while still containing ions in solution.
Reversible chemical processes (key reaction types)
Chemical reversibility requires that products can undergo a reaction pathway back to reactants under the same set of conditions.
Proton transfer (acid–base)
Many acid–base reactions are reversible because proton transfer can occur in either direction depending on relative acid/base strengths.
When an acid donates to a base, the products are a new acid and base that may transfer the proton back.
Reversibility is common in aqueous solution where frequent collisions and solvation stabilise ions and molecules, allowing both forward and reverse proton transfers.
Electron transfer (redox)
Some redox processes are reversible because electrons can be transferred in either direction between two species.
If both oxidised and reduced forms are present, collisions (or an external circuit in electrochemical contexts) can enable electron flow either way.
Whether redox is effectively reversible depends on how strongly conditions favour one direction (for example, if one product is removed, the reverse becomes less likely).
What controls whether reversibility is noticeable
Even if a reverse pathway exists, reversibility may be hard to observe when:
One direction is overwhelmingly favoured by conditions (e.g., products escaping, strong driving forces).
Reactants or products are physically separated, preventing collisions.
A product is removed (gas released, precipitate filtered off), suppressing the reverse process.
In AP Chemistry, treat reversibility as the key prerequisite for equilibrium: if both directions can occur within the same system, mixtures of reactants and products can persist rather than going “to completion.”
FAQ
If the system is open, stirring and large volumes of solvent can disperse ions and physically remove them from the solid surface.
Also, pouring off solution removes ions, preventing reattachment and making precipitation less likely.
Adsorption is surface attachment; absorption is uptake into the bulk material.
The syllabus example is adsorption/desorption, which is often reversible because particles can leave the surface without changing identity.
Yes, but removing a product (gas escaping, precipitate filtered off) can make the reverse direction negligible.
In a sealed system where products remain present, reversibility is more likely to be observed.
Higher temperature generally increases particle kinetic energy, which can increase evaporation and desorption and can help dissolved ions escape solvation.
Lower temperature often favours condensation and adsorption by reducing escape tendencies.
Protons are transferred through collisions and solvent-mediated interactions.
Because the conjugate acid–base pair remains present in solution, back-transfer can occur readily when collisions and relative strengths allow it.
Practice Questions
Question 1 (2 marks) State two observable processes that are reversible and, for each, name the forward and reverse change.
1 mark: Correct reversible process named (e.g., evaporation/condensation, adsorption/desorption, dissolution/precipitation).
1 mark: Correctly states matching forward and reverse directions for one additional process.
Question 2 (5 marks) A student claims that “once a salt dissolves, it cannot reform the solid unless the water evaporates.” Using the idea of reversibility, explain why this claim is not always correct. Your answer must refer to dissolution/precipitation and to at least one other reversible process from the syllabus.
1 mark: States dissolution and precipitation are opposite directions of a reversible process.
1 mark: Explains that in a saturated system, ions can reattach to the solid while others dissolve (both directions possible).
1 mark: Identifies a condition enabling reversibility (e.g., closed system; presence of undissolved solid; collisions at the crystal surface).
1 mark: Cites one other reversible example (evaporation/condensation OR adsorption/desorption) correctly.
1 mark: Links the second example to the idea that particles can move in both directions under the same conditions (not requiring a permanent one-way change).

