AP Syllabus focus: ‘When reactions are added to obtain an overall reaction, their enthalpy changes are added to obtain the net enthalpy change.’
Adding chemical equations is a bookkeeping skill that lets you combine measured or given reaction steps into one net process. The key idea is that the energy changes for the steps combine in the same way.
Core principle: add equations, add enthalpies
When two or more reactions are combined to produce a net (overall) reaction, the net enthalpy change is found by combining the individual enthalpy changes in the same mathematical way you combine the equations: cancellation in the equation corresponds to cancellation in the chemistry, but the enthalpies still sum.
Intermediate: a species produced in one step and consumed in a later step, so it does not appear in the overall reaction.
Intermediates are the “internal” species that cancel when you add steps; only reactants and products that remain after cancellation belong in the overall equation.
How to add reactions to get the overall reaction
1) Write the target as “steps that add up”
To add reactions successfully, you must ensure the set of steps can be summed so that unwanted species cancel. Practically, this means:
List each step reaction as a full chemical equation (include phases if given).
Decide which species must cancel (often intermediates) so the remaining species match the desired overall reactants/products.
2) Add the equations like algebra
When adding reaction equations:
Add all reactants from all steps on the left.
Add all products from all steps on the right.
Cancel any species that appear on both sides (same formula and phase).
If a species appears on both sides with different coefficients, cancel the smaller amount and keep the leftover coefficient on the appropriate side.
3) Add the enthalpy changes with the same combination
The overall enthalpy change is the algebraic sum of the step enthalpy changes that correspond to the equations you added.
= net enthalpy change for the overall reaction, in kJ (or kJ/mol as stated)
= enthalpy change for step , with its stated units (commonly kJ for the written reaction, or kJ/mol as defined)
This equation is only meaningful if the set of step equations truly adds to the overall equation you report; the chemistry (cancellation and remaining species) and the arithmetic (sum of values) must be consistent.
What must match between the chemistry and the energy
Coefficients and “per reaction as written”
Enthalpy changes are tied to the reaction as written. When you report , it must correspond to the final, combined equation you obtained (with its specific stoichiometric coefficients).
Units and interpretation
Be attentive to how the enthalpy is expressed:
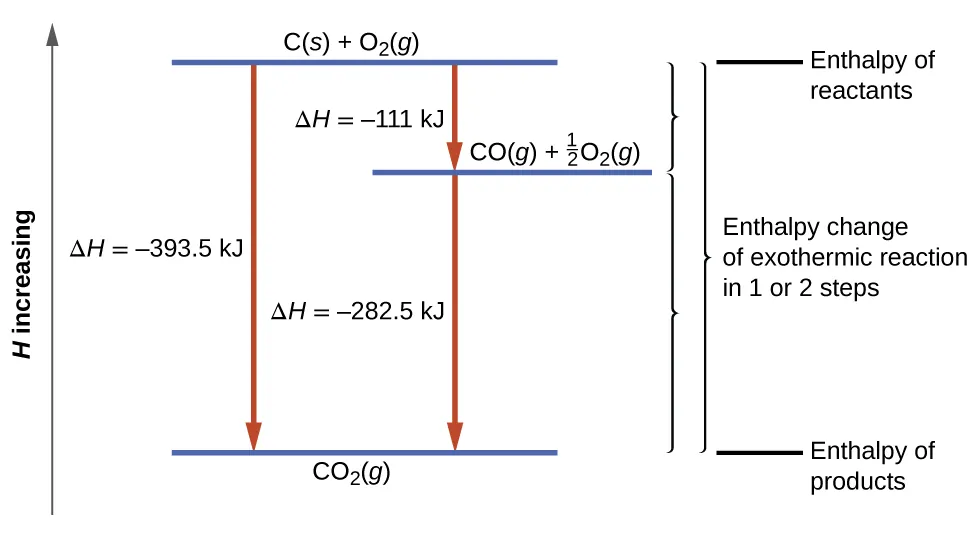
This enthalpy-level diagram for forming shows both a direct route and a two-step route through the intermediate . The vertical spacing represents enthalpy levels, and the labeled arrows show that the two step enthalpies sum to the same overall change. It provides a visual check that the “reaction as written” and the reported must stay consistent across steps. Source
If each step lists in kJ “for the reaction,” then summing gives kJ for the overall written reaction.
If steps list in kJ/mol, confirm what “per mole” refers to (often per mole of reaction, or per mole of a specified substance). Your combined result must keep that meaning consistent.
Self-checks that prevent common mistakes
Use these quick checks before finalising:
Atom check: total atoms of each element match on both sides of your overall equation.
Cancellation check: any intermediate you intended to eliminate truly cancels completely.
Sign check: if your summed is negative, the overall process is exothermic; if positive, it is endothermic.
Consistency check: you should be able to trace every term in the overall equation back to a reactant/product that survives after adding the steps.
Common pitfalls (and how to avoid them)
Cancelling species that are not identical (e.g., cancelling with is not valid).
Forgetting to cancel coefficients properly when the same species appears with different amounts on both sides.
Adding equations that do not actually produce the intended overall reaction (the algebra must reflect the chemical pathway you are constructing).
Mixing enthalpy units or meanings (kJ vs kJ/mol, or “per mole of substance X” vs “per reaction”).
FAQ
The overall reaction represents only what is consumed from outside the mechanism and what is produced at the end.
Intermediates are created and then used up, so their net change is zero; including them would misrepresent what is actually required overall.
Treat it like algebraic cancellation. Subtract the smaller coefficient from the larger and keep the remainder on the side where it is larger.
Only after this cancellation should you finalise the overall equation and interpret the net change.
No. Different states correspond to different enthalpy content and different chemical/physical situations.
Only cancel species that are chemically identical and in the same phase as written.
You may add enthalpy changes only when they refer to compatible “amounts of reaction.”
Check whether $\text{kJ mol}^{-1}$ is per mole of reaction as written or per mole of a named substance; keep that reference consistent across steps before adding.
Keep the sign explicit: negative for exothermic, positive for endothermic.
For significant figures, follow the least precise decimal place among the step enthalpies when you add/subtract, and always include units (typically kJ for the overall written equation).
Practice Questions
(2 marks) Two reactions are added: Reaction 1: , Reaction 2: , Write the overall reaction and calculate .
Overall reaction: (1)
(1)
(5 marks) Consider: (i) , (ii) , (iii) , Add the reactions to obtain an overall equation with no , , or present. Determine .
Correct addition showing cancels (1)
Correct addition showing cancels (1)
Correct addition showing cancels (1)
Overall equation: (1)
(1)

