AP Syllabus focus: ‘Equilibrium is dynamic: forward and reverse processes continue simultaneously at equal rates, so there is no net change that can be observed in the system.’
Chemical equilibrium can look static in the lab, but it is maintained by continuous molecular-level change. Understanding dynamic equilibrium and equal forward/reverse rates prevents common errors about what “equilibrium” really means.
Core idea: equilibrium is dynamic
In a reversible reaction, reactants form products while products can also reform reactants. Over time, these two opposing processes can balance.
Dynamic equilibrium: A state in a closed system where the forward and reverse processes continue simultaneously, but produce no net macroscopic change because they occur at equal rates.
“Dynamic” means particles are still reacting and interconverting; “equilibrium” means the overall amounts do not change measurably with time.
What “no net change” means (and what it does not mean)
At equilibrium, you can often measure that concentrations (solutions) or partial pressures (gases) stay constant.
This constancy happens even though individual molecules are still reacting.
Does mean: the mixture’s composition is stable over time.
Does not mean: reactants and products have the same concentration.
Does not mean: the reaction has stopped.
Forward and reverse rates become equal
As a system approaches equilibrium, the forward rate and reverse rate change until they match.
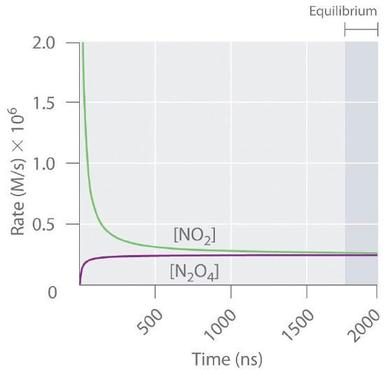
Forward and reverse reaction rates vs. time for a reversible system approaching equilibrium. The curves illustrate that the rates are generally different early on but converge until the kinetic condition is reached. After that point, both rates remain equal and nonzero, consistent with dynamic equilibrium. Source
Forward rate / Reverse rate: The speed at which reactants form products (forward) and products form reactants (reverse), typically expressed as change in concentration per unit time.
If you start with mostly reactants, the forward reaction has many opportunities (collisions, encounters) to occur, so its rate is initially high, while the reverse rate is initially low because little product is present. As products accumulate, the reverse rate increases.
= forward reaction rate (e.g., )
= reverse reaction rate (e.g., )
This equality of rates is the defining kinetic condition for equilibrium. Once achieved, the system’s macroscopic properties remain constant because any formation of products is balanced by product consumption at the same pace.
Why the rates change over time
Rates depend on how often reacting particles meet in the correct way. Qualitatively:
As reactants are used up, reactant concentration decreases, so the forward rate tends to decrease.
As products build up, product concentration increases, so the reverse rate tends to increase.
Eventually, the two rates become equal, locking in a constant overall composition.
Microscopic picture at equilibrium
At equilibrium, both directions continue:
Reactant molecules still convert into product molecules.
Product molecules still convert back into reactant molecules.
Individual molecules do not “know” equilibrium has been reached; equilibrium is a system-level balance.
A useful mental model is a busy two-way hallway: people continue moving in both directions, but if the flow rates match, the number of people on either side stays the same.
Common misconceptions to avoid
“Equilibrium means equal amounts.” False. Equilibrium requires equal rates, not equal concentrations; one side may be strongly favoured while still meeting .
“Equilibrium means nothing is happening.” False. The forward and reverse processes are both active.
“Once at equilibrium, the system can’t change.” The system’s composition can change if conditions are altered; the key point here is that, at equilibrium under fixed conditions, the net change is zero because the opposing rates match.
FAQ
Use an isotopic tracer (e.g., label an atom in the reactant).
After equilibrium is established, the label appears in product molecules over time, showing ongoing interconversion even though bulk concentrations stay constant.
Rate depends on pathway and conditions, not just amounts.
Different rate laws and molecular mechanisms can allow equal rates at very different reactant-to-product ratios.
A catalyst increases the rates of both forward and reverse reactions by lowering activation energy for each pathway.
Equilibrium is still defined by $r_f=r_r$, but the system reaches that point faster.
Only if there is no net gain/loss of the reacting species.
If gases escape or enter, the composition can drift, preventing a stable state where opposing rates balance for the defined reaction system.
No.
Individual molecules continue to convert back and forth; equilibrium describes constant overall amounts, not fixed identities of particular molecules.
Practice Questions
(2 marks) State what is meant by dynamic equilibrium in a reversible reaction, and state the relationship between the forward and reverse rates at equilibrium.
Equilibrium is dynamic: forward and reverse reactions continue simultaneously (1).
At equilibrium the forward and reverse rates are equal, e.g. (1).
(6 marks) A reversible reaction occurs in a sealed container at constant temperature. Initially, only is present. Describe how the forward and reverse reaction rates change with time until equilibrium is reached, and explain why the measured concentrations become constant even though reactions continue.
Forward rate initially high because is high (1).
Reverse rate initially ~0/very low because is ~0/very low (1).
As is converted to , decreases so forward rate decreases (1).
As forms, increases so reverse rate increases (1).
Equilibrium reached when (1).
Concentrations become constant because formation and consumption of each species balance (no net change), though interconversion continues (1).

