AP Syllabus focus: ‘Graphs of concentration, partial pressure, or reaction rate versus time can be used to understand how a system reaches chemical equilibrium.’
Equilibrium graphs translate an invisible molecular process into visible trends. By reading shapes, slopes, and plateaus on concentration-, partial pressure-, and rate-versus-time plots, you can identify when equilibrium is reached and what is happening throughout the approach.
What equilibrium graphs show
Concentration or partial pressure vs time
A concentration-time or partial-pressure-time graph tracks how amounts of reactants and products change as a reaction proceeds toward equilibrium.
Reactant curves typically decrease from their initial values and then level off
Product curves typically increase from their initial values and then level off
The “flat” region (constant value) indicates no net change over time at the macroscopic level
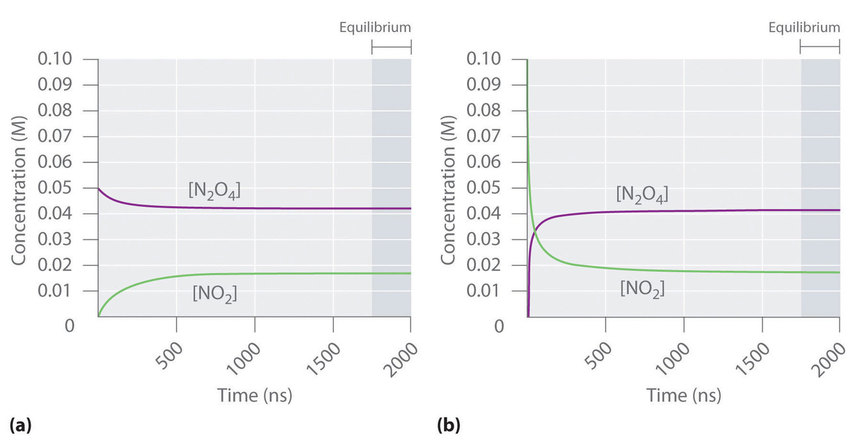
Concentration–time plots for the reversible �� system, shown for two different starting conditions. In each case, the curves approach constant plateau values, emphasizing that equilibrium is identified by concentrations becoming time-invariant (horizontal), not by reactant and product concentrations becoming equal. Source
Reaction rate vs time
A rate-time graph shows how quickly the forward and reverse processes occur as the system evolves.
The forward rate usually starts larger (if mostly reactants are present) and decreases
The reverse rate usually starts smaller and increases as products accumulate
Equilibrium is indicated when the two rates become equal and constant
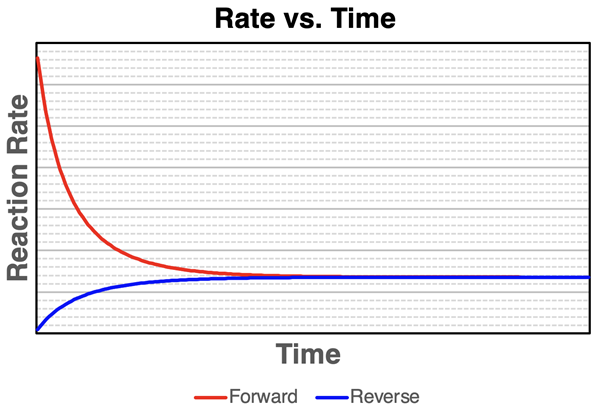
Rate–time curves for a reversible reaction, showing the forward and reverse reaction rates evolving until they become equal. The intersection and subsequent overlap at a constant value indicates dynamic equilibrium: reactions continue microscopically, but there is no net macroscopic change because the opposing rates match. Source
Key graph-reading ideas
Using slope to infer direction
On a concentration or partial pressure graph, the slope tells you whether a species is being formed or consumed at that moment.
Slope (on a concentration/pressure vs time graph): the rate of change of a plotted quantity with time; a positive slope means increasing, and a negative slope means decreasing.
A steep slope indicates rapid change (far from equilibrium), while a shallow slope indicates slower change (closer to equilibrium).
Recognising the equilibrium time
Equilibrium is reached at the time when:
Concentration/partial pressure curves become horizontal (constant)
Rate curves (forward and reverse) become equal and remain equal
Do not assume equilibrium means equal concentrations of reactants and products; graphs commonly show different plateau heights.
Connecting rate and concentration graphs
Rate trends are consistent with concentration trends:
As reactant concentration/partial pressure drops, forward collisions become less frequent, so the forward rate falls
As product concentration/partial pressure rises, reverse collisions become more frequent, so the reverse rate rises
When the forward and reverse rates match, concentrations stop changing (plateau)
= change in concentration, in
= change in time, in s
This expression links the visual steepness of a concentration-time curve to “how fast” the system is changing at that time.
Common graph features and how to interpret them
Plateaus and what they mean
A plateau means the plotted quantity is constant, which supports the equilibrium idea that macroscopic properties are steady. However:
Both reactants and products should still be present at the plateau
The plateau indicates stability over time, not that the reaction has stopped
Curves that level off at different values
Different plateau levels reflect the equilibrium mixture composition. A higher plateau for products suggests a larger equilibrium amount of product (but the graph alone does not quantify an equilibrium constant).
Multiple species on one set of axes
When several curves are shown together:
Compare when each curve becomes flat to identify a single equilibrium time for the system
Compare how quickly each curve changes early on to infer which species changes fastest (often tied to stoichiometry and starting amounts)
FAQ
If the product curve starts high and decreases while the reactant curve increases, the net change is towards reactants initially.
Rate–time plots would show the reverse rate initially greater than the forward rate.
Near equilibrium, changes are small, so measurement uncertainty can be comparable to the real change.
Sampling, instrument resolution, and slight temperature drift can all create scatter around an otherwise flat plateau.
Yes, but the solid’s “concentration” is not typically plotted because it is effectively constant.
You would look for plateaus in dissolved species concentrations or gas partial pressures instead.
This can indicate an external disturbance during the run (e.g. brief temperature fluctuation or mixing issue).
The system temporarily leaves equilibrium and later re-establishes equal forward and reverse rates.
A compressed scale can make significant changes appear flat, while an expanded scale can exaggerate minor drift.
Always check axis units and increments before deciding whether a region is truly constant.
Practice Questions
Question 1 (2 marks) A rate-versus-time graph for a reversible reaction shows the forward rate decreasing and the reverse rate increasing until they intersect and then remain equal. What does the intersection point represent, and what is true about concentrations after this time?
Intersection represents the time when forward and reverse rates are equal (equilibrium established). (1)
After this time, concentrations remain constant (no net change), though reactions continue. (1)
Question 2 (5 marks) For , a concentration–time graph shows decreasing from to and increasing from to , with both becoming constant after . Explain what the graph indicates about (i) when equilibrium is reached, (ii) whether at equilibrium, and (iii) how the forward and reverse rates compare before and after .
Equilibrium reached when both concentrations become constant; here after . (1)
At equilibrium, concentrations need not be equal; here (0.40 vs 0.60). (1)
Before , net change occurs (concentrations changing), implying forward and reverse rates are unequal. (1)
Since decreases while increases, forward rate reverse rate overall before . (1)
After , forward rate reverse rate (dynamic equilibrium) with no net concentration change. (1)

