AP Syllabus focus: ‘Repeated collisions lead to thermal equilibrium, where average kinetic energies and therefore temperatures of both bodies are the same.’
Thermal equilibrium links what you measure (temperature) to what particles do (random motion). In AP Chemistry, it explains why heat transfer stops and why equal temperatures signal equal average kinetic energies.
What Thermal Equilibrium Means
When two objects (or regions of matter) are in thermal contact, energy can be exchanged through particle collisions. Over time, this exchange reduces the temperature difference until there is no net heat transfer.
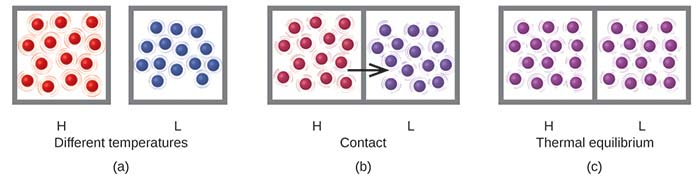
Three-panel particulate model showing two samples initially at different temperatures, then placed in thermal contact, and finally reaching thermal equilibrium. The visual progression emphasizes that energy redistribution occurs through collisions until both regions share the same temperature (equal average kinetic energy), after which there is no net heat flow. Source
Thermal equilibrium: The state in which two bodies in thermal contact have the same temperature, so there is no net heat transfer between them.
A key AP idea is that temperature is tied to particle motion: a higher temperature corresponds to a higher average kinetic energy of particles. Therefore, when temperatures become equal, the average kinetic energies become equal as well (even though individual particles still have a wide range of energies).
Microscopic Picture: Why Collisions Drive Equality
At the particle level, thermal contact enables collisions across the boundary between the two bodies.
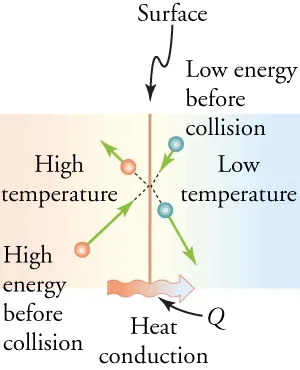
Particle-level diagram of conduction at an interface between a high-temperature region and a low-temperature region. The velocity vectors indicate higher kinetic energy on the hot side, and the labeled heat flow shows net energy transfer from hot to cold during repeated collisions. Source
In the initially warmer body, particles have a higher average kinetic energy.
In the initially cooler body, particles have a lower average kinetic energy.
Collisions transfer kinetic energy from faster-moving particles to slower-moving particles.
Repeated collisions gradually shift the average kinetic energies toward the same value in both bodies.
This process does not require chemical change; it is a redistribution of energy among particles. Importantly, collisions continue even at equilibrium, but energy transfers in opposite directions balance out, producing zero net transfer.
Conditions at Equilibrium (Macroscopic Signs)
At the macroscopic level, thermal equilibrium is identified by stable temperatures that have become equal (assuming the bodies remain in contact and conditions are steady).
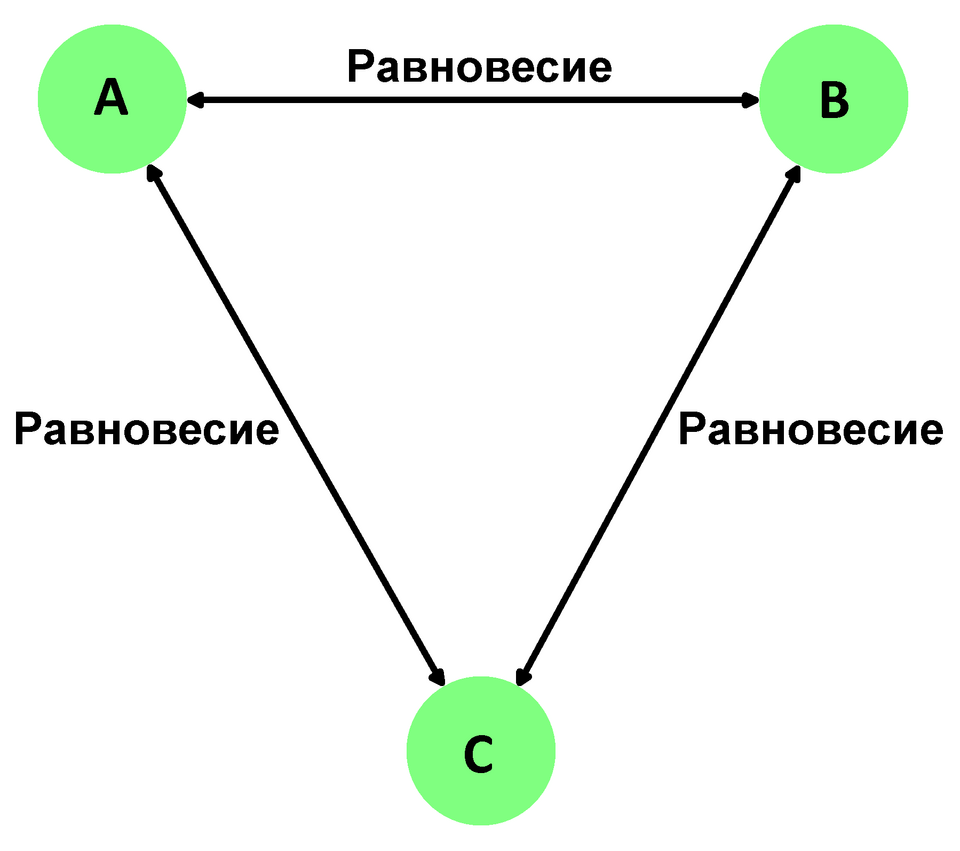
Diagram illustrating the zeroth-law idea that thermal equilibrium is transitive: if A is in thermal equilibrium with B and B with C, then A is in thermal equilibrium with C. This is the logical basis for using a thermometer as a “third system” to compare temperatures consistently. Source
= temperature of body A at equilibrium (K or °C)
= temperature of body B at equilibrium (K or °C)
= net heat transferred between A and B (J)
The first line states the temperature criterion. The second line expresses the heat-transfer consequence: at equilibrium, there is no overall (net) heat exchange between the bodies, even though microscopic collisions persist.
Important Clarifications (Common AP Misconceptions)
Equal temperature does not mean equal thermal energy. Two samples can share a temperature but contain different total energy because total energy depends on how much matter there is and how energy is stored in that substance.
Equilibrium is a state, not a speed. How fast equilibrium is reached depends on factors that change collision effectiveness, such as surface contact, mixing/stirring, and the material’s ability to conduct energy internally.
Thermal equilibrium is specific to heat exchange. Two objects can have the same temperature yet still not be in thermal equilibrium with their environment if external energy transfer continues (for example, if one is being heated while in contact).
FAQ
It does not require direct contact.
Thermal equilibrium can be reached by energy transfer via radiation (and, in fluids, convection), provided exchange continues until $q_{\text{net}}=0$ and temperatures equalise.
The thermometer must reach thermal equilibrium with what it measures.
If it is still exchanging energy, its temperature (and reading) is still changing, so it lags behind the object’s true temperature.
Equilibrium is slower when energy transfer is less effective, for example:
small contact area
poor thermal conductivity
little mixing (in liquids)
large masses requiring more energy redistribution
At equilibrium, transfers in both directions balance.
Over time, as temperatures match, the average energy exchanged from A to B equals that from B to A, so the net is zero.
Yes, in practical situations.
If one object is simultaneously gaining/losing energy to another source (e.g., ongoing heating), equal temperatures can be momentary rather than a true equilibrium state between the two objects.
Practice Questions
(2 marks) Define thermal equilibrium and explain what must be true about the particles’ average kinetic energies when it is reached.
Correct definition: same temperature and no net heat transfer (1)
States average kinetic energies are equal when temperatures are equal (1)
(5 marks) Two solids, A (hotter) and B (cooler), are placed in thermal contact and isolated from the surroundings. Describe, in terms of particle collisions and energy transfer, how the system reaches thermal equilibrium and what characterises the final state.
Energy is transferred via collisions at the interface (1)
Net transfer is from hotter A to cooler B initially (1)
Average kinetic energy of A decreases; of B increases (1)
Temperatures become equal at equilibrium () (1)
At equilibrium, collisions continue but there is no net heat transfer () (1)

