AP Syllabus focus: ‘Particles in a warmer body have a greater average kinetic energy than particles in a cooler body.’
Temperature is not “how much heat” something contains; it is a molecular-level indicator of motion. The kinetic molecular view links what a thermometer reads to how fast particles are moving on average.
Core kinetic molecular idea
In the kinetic molecular theory, particles in matter are in constant random motion. That motion is described by kinetic energy, and temperature tracks the typical (average) amount of that energy.
A warmer sample has particles moving faster, on average.
A cooler sample has particles moving slower, on average.
“Faster” refers to the magnitude of particle velocities in random directions, not motion in a single direction.
Temperature as an average, not a total
A crucial AP Chemistry distinction is that temperature relates to an average per particle, not the total energy of the whole sample.
Temperature: A measure proportional to the average kinetic energy of particles in a substance.
Because temperature is an average, a small hot object can have a higher temperature than a large cool object even if the large object contains more total thermal energy.
Average kinetic energy and the Kelvin scale
For an ideal monatomic gas, the link between temperature and average translational kinetic energy is direct and quantitative. This supports the statement that “warmer means faster particles.”
= average kinetic energy per particle (J)
= Boltzmann constant ()
= absolute temperature (K)
This relationship requires Kelvin because Kelvin is proportional to thermal motion and has a true zero point: at 0 K, the idealised extrapolated average translational kinetic energy approaches zero.
What “average kinetic energy” implies for speed
Kinetic energy depends on both mass and speed, so “higher temperature” does not mean every particle has the same speed. Instead:
Increasing increases the average kinetic energy.
A higher average kinetic energy corresponds to a higher average particle speed.
Individual particles still have a wide range of speeds at any fixed .
Distributions: not all particles move equally fast
Particle speeds in a gas follow a Maxwell–Boltzmann distribution (a spread of speeds).
What happens to the distribution when temperature increases
When temperature increases:
The peak of the distribution shifts to higher speed (typical particles move faster).
The distribution becomes broader (a wider range of speeds occurs).
The fraction of very fast particles increases noticeably.
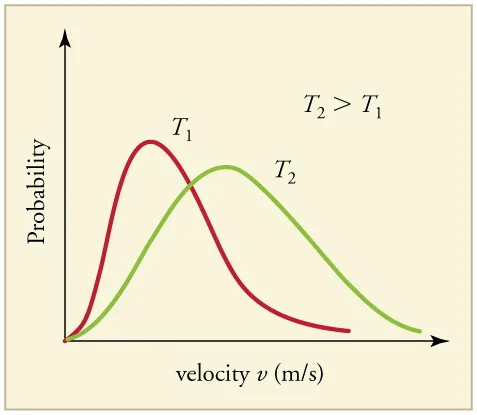
Maxwell–Boltzmann speed distributions for the same gas at two different temperatures. The higher-temperature curve is shifted rightward (higher typical speeds) and is broader, illustrating that increasing raises the average kinetic energy while preserving a spread of molecular speeds. Source
This distribution view is the microscopic meaning of “particles in a warmer body have a greater average kinetic energy”: the entire speed profile shifts so the average rises, even though some particles remain relatively slow.
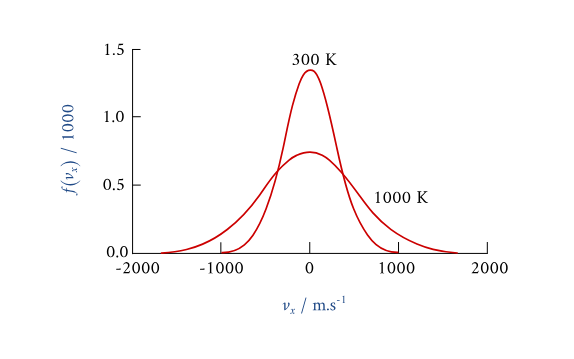
A temperature-dependent molecular-velocity distribution for a gas, showing different curves at two temperatures. The higher-temperature curve places more probability density at larger speeds, demonstrating how heating increases the average kinetic energy without making all particles move at one single speed. Source
Comparing different substances at the same temperature
Temperature fixes the average kinetic energy, not the average speed, across different particle masses.
At the same temperature, different gases have the same average kinetic energy per particle (idealised).
Lighter particles must move faster on average than heavier particles to have the same kinetic energy.
So, at the same , helium atoms have a higher typical speed than oxygen molecules, even though the average kinetic energy is the same.
Beyond gases: particles in liquids and solids
The “warmer means faster particles” idea applies to all phases, with phase-specific motion:
Solids: particles vibrate about fixed positions; higher means larger-amplitude, higher-energy vibrations.
Liquids: particles both vibrate and move past one another; higher increases the typical speeds of this motion.
Gases: particles move freely between collisions; higher increases typical translational speeds.
In every phase, a higher temperature corresponds to greater average microscopic kinetic energy of the particles’ allowed motions, which is why temperature rises when particle motion becomes more energetic.
FAQ
Kelvin is an absolute scale with a true zero tied to microscopic motion.
Celsius is offset by 273.15, so proportional relationships with kinetic energy only work with $T$ in K.
Temperature reflects an average over an enormous number of particles.
Random collisions and energy sharing produce a spread of kinetic energies, giving a distribution of speeds at the same $T$.
For ideal monatomic gases, temperature directly tracks average translational kinetic energy.
For molecules and condensed phases, energy is also stored in rotational/vibrational motions, but temperature still correlates with the average kinetic energy of the accessible particle motions.
Temperature is an average per particle, not a total.
A large object has many more particles, so the sum of their energies can be greater even if each particle has a lower average kinetic energy.
Two key changes occur:
The most probable speed increases (the “typical” speed rises).
The high-speed tail grows, meaning a much larger fraction of particles are moving very fast at higher $T$.
Practice Questions
(2 marks) Two sealed containers hold ideal gases, A at 290 K and B at 310 K. Which container’s particles have the greater average kinetic energy, and why?
Identifies B / 310 K as greater (1)
States that average kinetic energy is proportional to absolute temperature (in K) / higher means higher average kinetic energy (1)
(5 marks) Helium and nitrogen are each in separate containers at the same temperature. a) Compare the average kinetic energy per particle in the two gases. (1 mark)
b) Compare the typical particle speeds in the two gases and justify your answer. (2 marks)
c) Sketch description only (no diagram required): describe how the Maxwell–Boltzmann speed distribution for nitrogen changes when temperature increases. (2 marks)
(a) Same average kinetic energy per particle at the same temperature (ideal gas assumption) (1)
(b) Helium particles move faster on average / have higher typical speed (1)
Justification: to have the same kinetic energy, lower mass must correspond to higher speed (can reference ) (1)
(c) Distribution shifts to higher speeds (peak moves right) (1)
Distribution broadens / peak lowers and higher-speed tail increases (1)

