AP Syllabus focus: ‘Heat transferred between two bodies can be quantified using q = mcΔT, relating mass, specific heat capacity, and temperature change.’
Thermochemical problems often reduce to tracking how much thermal energy moves when temperature changes. In AP Chemistry, the key quantitative tool is , linking heat, mass, material properties, and measured temperature change.
Core idea: heat transfer changes temperature
When two objects (or an object and a liquid) are in thermal contact, energy can transfer as heat. If no phase change occurs and the material’s heat capacity is treated as constant over the temperature range, the temperature change provides a direct measure of heat transferred.
Key quantities you must identify
Specific heat capacity: The heat required to raise the temperature of 1 gram of a substance by 1°C (or 1 K).
Specific heat capacity is a property of the substance, so equal heat inputs can produce different temperature changes in different materials.
The heat transfer relationship
To quantify heat transfer during a temperature change, AP Chemistry uses a linear model that is accurate for many common laboratory temperature ranges.
= heat transferred (J)
= mass of the substance whose temperature changes (g)
= specific heat capacity
= temperature change, (°C or K)
The equation is most often applied to liquids (especially aqueous solutions) and solids when temperature changes smoothly and no chemical or phase change is occurring in the sample whose temperature you are tracking.
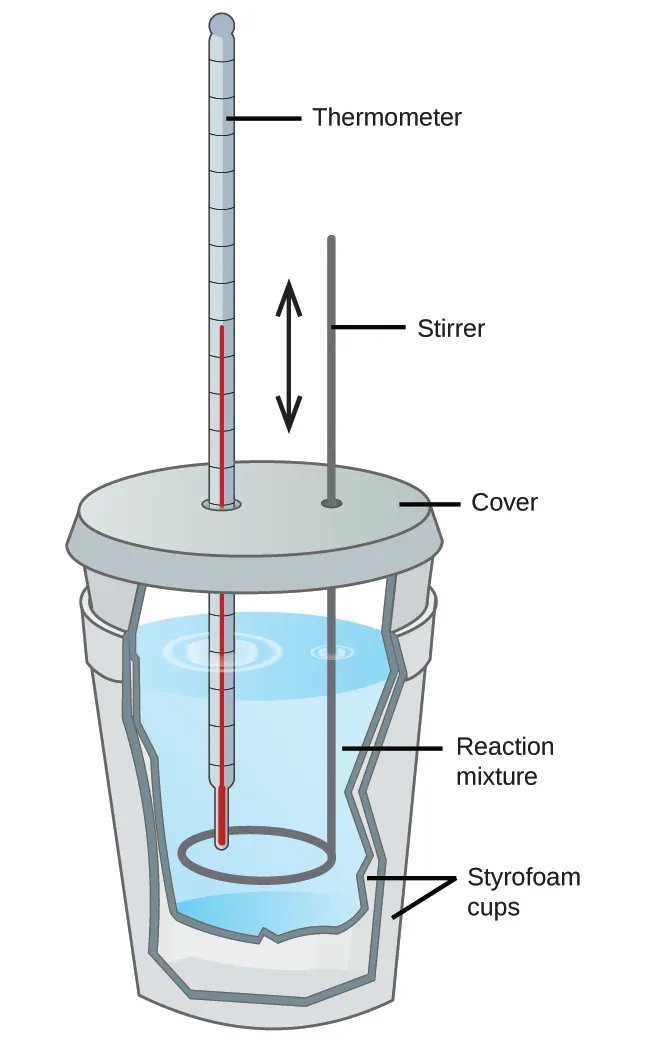
A simple constant-pressure (coffee-cup) calorimeter is shown, constructed from nested polystyrene cups with a lid, thermometer, and stirrer immersed in the reaction mixture. This setup makes explicit that the measured is the temperature change of the solution (the surroundings for the reaction), which is then paired with the solution’s mass and specific heat in . Source
Interpreting the sign of
Using automatically handles direction:
If the substance warms, , so for that substance (it absorbed heat).
If the substance cools, , so for that substance (it released heat).
Always state clearly which object or sample your calculated refers to (the water, the metal, the solution, etc.). The same physical event involves heat lost by one part and gained by another; the sign depends on the chosen “receiver” of heat.
Units and temperature conventions that prevent mistakes
Temperature change: °C vs K
For differences, 1 K equals 1°C, so has the same numerical value in kelvin or Celsius. The crucial step is consistency:
Compute from final minus initial using the same scale for both.
Use units that match the given value (commonly per °C).
Mass and specific heat capacity compatibility
Common pitfalls come from mismatched units:
If is in , then must be in grams.
If is in , then must be in kilograms.
Heat will come out in joules if is expressed in joule-based units.
What “counts” as the system in
The equation itself does not choose a system; you do. For any chosen sample:
Treat it as a single “lump” with a uniform temperature (well-mixed liquid, or a solid that equilibrates quickly).
Use the sample’s own , , and to calculate the heat it gained or lost.
This is why stirring and good thermal contact matter experimentally: they help ensure the measured temperature represents the whole sample.
A classroom coffee-cup calorimeter is depicted with two nested polystyrene cups, a lid, and probes (thermometer and stirrer) extending into the solution. The image emphasizes the practical steps used to approximate a single, well-mixed temperature so that the measured meaningfully represents the whole sample in . Source
Limits of applicability (when not to use it directly)
The model assumes a temperature change with no hidden energy pathways. It becomes unreliable or incomplete when:
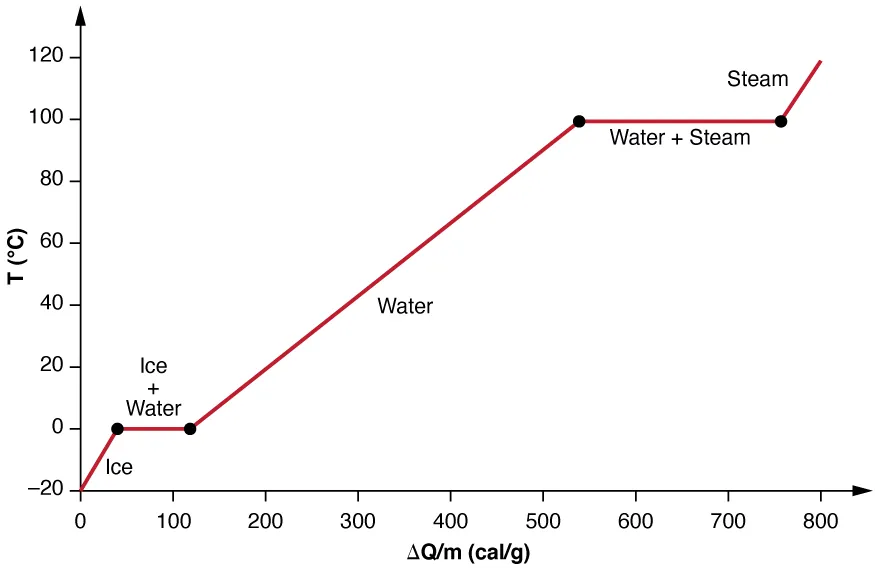
This heating curve plots temperature versus energy added and highlights flat segments at phase changes where temperature stays constant even though heat is still being absorbed. Those plateaus illustrate why cannot capture latent-heat energy during melting/boiling, because while . The sloped regions correspond to single-phase warming where the linear model is appropriate over modest temperature ranges. Source
A phase change occurs (temperature may stay constant while heat is still transferred).
changes significantly over the temperature interval (large temperature ranges).
The temperature is not uniform within the sample (poor mixing or slow conduction).
In these cases, may describe only part of the energy transfer or may require additional information.
FAQ
Because kelvin and Celsius have the same-sized degree. Only the zero points differ, so differences cancel: $(T_f+273.15)-(T_i+273.15)=T_f-T_i$.
It means the chosen sample lost heat overall. Typically its temperature decreased, so energy flowed from that sample to something else in thermal contact.
Not exactly. It varies with temperature and (slightly) with pressure, but over modest lab temperature ranges it’s often treated as constant to make $q=mc\Delta T$ usable.
The formula assumes a single temperature represents the whole mass. Stirring reduces temperature gradients so the measured $T$ matches the sample’s average thermal energy.
Not by itself. During a phase change, temperature can remain constant while heat transfers; you’d need a latent-heat approach rather than relying only on $\Delta T$.
Practice Questions
(2 marks) State the equation used to calculate heat transferred during a temperature change and define each symbol, including a suitable unit for .
1 mark: States .
1 mark: Correctly defines symbols (any three of ) with at least one correct unit (e.g., in J).
(5 marks) A sample of water cools from to . Using , calculate for the water and state whether heat was absorbed or released.
1 mark: Calculates .
1 mark: Substitutes into .
1 mark: Correct numerical magnitude (allow rounding): .
1 mark: Correct sign: .
1 mark: Interprets sign: heat released by the water.

