AP Syllabus focus: ‘When particles are in thermal contact, collisions can transfer energy; this energy transfer is called heat transfer (heat exchange).’
Heat transfer is best understood by zooming in to the particle level. When two objects touch, countless collisions at their boundary redistribute energy, driving a net flow from hotter to cooler until the transfer no longer has a preferred direction.
Core idea: collisions move energy
Whenever matter is made of moving particles, collisions are the mechanism that allows energy to be passed from one place to another. In thermochemistry, the energy being redistributed by these collisions is tracked as heat transfer.
When two bodies are brought together, the key requirement is thermal contact—particles from each body must be able to interact directly (or through immediate neighboring particles) so collisions can occur across the boundary.
Heat (q): Energy transferred between a system and its surroundings due to a temperature difference.
A temperature difference matters because it creates an imbalance in how much kinetic energy particles tend to carry, so collisions have a directional “bias” on average.
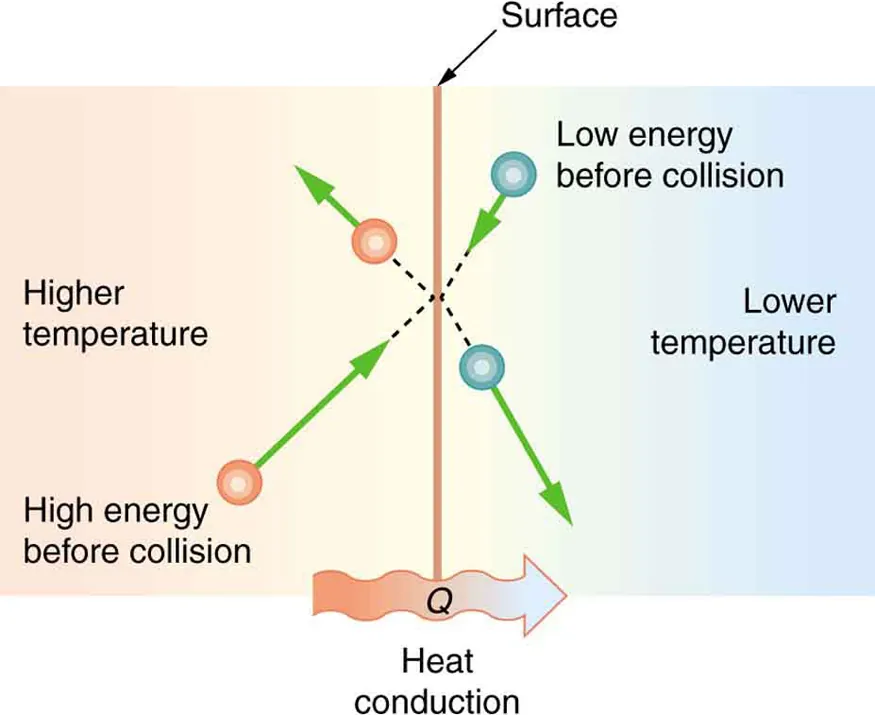
Collision-mediated heat transfer is inherently statistical: particles on the hotter side have higher average kinetic energy, so collisions at the interface tend to transfer energy to particles on the cooler side. This schematic makes the “directional bias” of many random collisions visible, linking the microscopic picture to macroscopic net heat flow ( from hot to cold). Source
What “thermal contact” means at the particle level
Contact creates a collision pathway
Thermal contact does not require mixing; it requires a route for collisions (or collision chains) to transmit energy.
Solid–solid contact: boundary atoms vibrate and collide through the lattice
Liquid–solid contact: fluid particles collide with the solid surface particles
Gas–solid or gas–liquid contact: fast gas particles strike the surface and exchange energy
Thermal contact: A physical situation in which particles from two bodies can interact so that energy can be transferred as heat.
In AP Chemistry terms, once thermal contact exists, the observed macroscopic change (warming/cooling) is explained microscopically by collision-driven energy exchange.
How collisions actually transfer energy
Individual collisions exchange kinetic energy
In any collision, energy can be redistributed among the colliding particles. Even though any one collision might transfer energy in either direction, the net effect depends on which side has particles that, on average, are moving more energetically.
A higher-energy particle striking a lower-energy particle tends to leave the lower-energy particle with more kinetic energy afterward
That gain may appear as faster translational motion (especially in gases and liquids) or as increased vibration (especially in solids)
Over many collisions, the initially cooler region gains kinetic energy and the initially warmer region loses kinetic energy
Many collisions create a net heat flow
Because collisions occur in enormous numbers, AP Chemistry focuses on the statistical result:
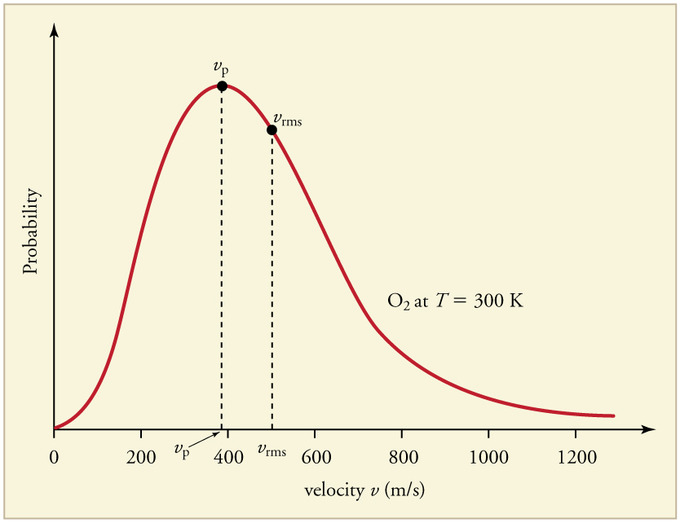
A Maxwell–Boltzmann distribution plot shows that, at a given temperature, particles occupy a range of speeds (and therefore kinetic energies), not a single value. Comparing curves at different temperatures illustrates why a hotter sample has a larger fraction of faster, higher-kinetic-energy particles, which underlies the net direction of collision-driven energy exchange during thermal contact. Source
If two regions have different temperatures, collisions produce net heat transfer from the warmer region to the cooler region
As energy is redistributed, the temperature difference shrinks, reducing the “driving force” for net transfer
This aligns with the syllabus statement: when particles are in thermal contact, collisions can transfer energy, and that energy transfer is called heat transfer (heat exchange).
Conduction as collision chains (the most direct case)
Solids: vibrations pass energy along
In solids, particles are not free to flow past one another; instead they vibrate about fixed positions. Energy transfer occurs when:
A more energetic particle collides with (pushes/pulls on) a neighbor
That neighbor vibrates more strongly and then interacts with its next neighbor
The effect propagates through the solid as a chain of energy-sharing interactions
Liquids and gases: collisions plus motion
In fluids, collisions still transfer energy, but particles also move and continually bring energy to new locations.
Collisions rapidly redistribute energy locally
Particle motion continually creates new collision partners, spreading energy through the sample
What you should connect to lab observations
Temperature change is evidence of collision-driven transfer
In a typical calorimetry-style setup, you cannot see collisions, but you can observe their outcome:
If an object warms up while in contact with another body, collisions have transferred energy into it as heat
If it cools down, collisions have transferred energy out of it as heat
“System” and “surroundings” language stays consistent
Regardless of which piece you label as the system, heat transfer refers specifically to energy crossing the boundary due to the temperature difference, mediated by collisions at (and near) that boundary.
FAQ
Heat transfer can still occur because collisions occur at the microscopic contact points.
If the true contact area is small (rough surfaces), the collision pathway is limited, so the rate of transfer is reduced.
A single collision transfers energy between particles (often kinetic and vibrational components).
“Heat” is the macroscopic bookkeeping term for energy crossing a boundary due to temperature difference.
Collisions are random, but the distribution of particle energies differs between hot and cold bodies.
Statistically, more energy is passed from the higher-energy side to the lower-energy side per unit time.
Greater pressure typically increases real contact area between surfaces.
More microscopic contact points means more collision pathways, usually increasing the rate of heat transfer.
Not by collision-driven transfer across a gap.
Across a vacuum gap, collisions cannot occur; any energy transfer would require a different mechanism rather than heat transfer by particle collisions.
Practice Questions
(2 marks) Explain, in terms of particles and collisions, why energy is transferred when a hot metal block is placed in contact with cooler water.
States that particles are in thermal contact and collisions occur between metal and water particles (1)
Explains that collisions transfer kinetic energy on average from hotter (more energetic) particles to cooler (less energetic) particles, i.e. heat transfer/heat exchange (1)
(5 marks) Two solids, A and B, are pressed together. A is initially at a higher temperature than B. Describe how collisions lead to heat transfer across the boundary and how the net direction of transfer changes as time passes.
Mentions thermal contact enabling interactions at the boundary (1)
Describes collisions at the interface transferring energy between particles (1)
Explains energy spreads through each solid by collision/vibration chains between neighbouring particles (1)
States net transfer is from A to B initially because A’s particles are, on average, more energetic (1)
Explains that as temperatures become closer, the net transfer decreases and can cease when there is no temperature difference (1)

