AP Syllabus focus: ‘Represent a transformation with an energy diagram, using appropriate axes labels, scale, and units to communicate energy changes.’
Energy diagrams are visual models for tracking how a system’s energy changes during a transformation. A high-quality sketch depends on correct axes, consistent units, clear labels, and a scale that communicates the size and direction of the energy change.
What an energy diagram must communicate
An energy diagram should make these features unambiguous:
Whether the system ends at higher energy or lower energy than it started
The magnitude of the energy change (with units)
Which state is reactants (or initial state) and which is products (or final state)
The pathway from initial to final state along a reaction/progress axis
Axes: labels, meaning, and units
Reaction coordinate (reaction progress): A horizontal-axis variable that tracks progress from initial state to final state; it is not time and does not have standard units.
Label the axes as follows:
y-axis: Energy (or Enthalpy, if specified by the problem context)
Typical units: kJ/mol for chemical reactions; kJ for a specific sample; sometimes J for small-scale processes
Include the unit directly on the axis label, such as Energy (kJ/mol)
x-axis: Reaction coordinate or Reaction progress
Do not add time units (s, min) because the diagram is not a rate plot
Use a clean origin and a sensible vertical range so the energy difference between initial and final states is visually apparent.
Core labels: initial, final, and the energy change
Place and label two horizontal levels (or plateaus):
Reactants (initial state) on the left
Products (final state) on the right
Then indicate the vertical difference between them with a bracket or arrow labeled ΔH (or “energy change,” if the prompt avoids thermodynamic symbols).
The sign should match the direction:
Products lower than reactants: ΔH < 0
Products higher than reactants: ΔH > 0
A diagram should never force the reader to infer the sign from text alone; the vertical placement must show it.
= enthalpy change of the transformation (kJ/mol or kJ, matching the axis)
= energy/enthalpy level of products (kJ/mol or kJ)
= energy/enthalpy level of reactants (kJ/mol or kJ)
After writing a value on the diagram (for example, “ΔH = −120 kJ/mol”), ensure the y-axis unit is consistent with that value.
Scale and units: making the sketch quantitatively meaningful
Even when a sketch is not to scale, it must still be internally consistent:
If two diagrams are compared, use the same y-axis units and a comparable vertical scale
If numerical values are given, reflect relative magnitudes (a 10 kJ/mol change should not look larger than a 100 kJ/mol change)
Avoid mixing per-mole and per-sample units (kJ/mol vs kJ) in the same diagram
If the prompt provides energy values, mark them clearly:
Label the reactant level (e.g., “50 kJ/mol”)
Label the product level (e.g., “−20 kJ/mol”)
Or label only ΔH, provided the relative heights are correct
Optional features sometimes required by prompts
Some prompts ask for additional annotations.
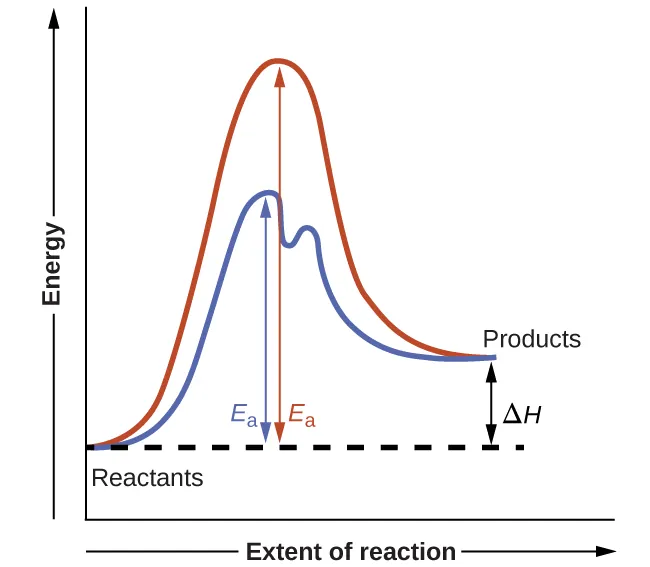
Reaction diagrams compare energy versus extent of reaction and explicitly label both the overall energy change () and the activation energy barrier (). The two curves show how a catalyst provides an alternative pathway with a lower peak (smaller ) while leaving the reactant and product energy levels—and thus —unchanged. Source
Include them only if requested:
A single “hump” along the path to represent an energy barrier
A labelled highest point (“peak” or “transition state”) and a vertical arrow from reactants up to the peak
When adding such features, keep labels clear and ensure they do not replace the required reactant/product levels and ΔH label.
Sketching checklist (what graders look for)
Correct axes: Energy (units) vs Reaction coordinate
Reactants on left, products on right, each clearly labelled
Correct relative energy levels (higher/lower)
ΔH labelled with sign and units consistent with the y-axis
Clean, readable scaling and annotations (no conflicting units or ambiguous arrows)
FAQ
Use kJ/mol when the energy change is given per mole of reaction (most common in chemistry problems).
Use kJ when the prompt refers to a specific sample or calorimetry amount rather than per mole.
No. Reaction coordinate is a conceptual progress variable from initial to final state.
Even a “slow” reaction can be drawn with the same horizontal length as a fast one.
“To scale” means vertical distances match numerical energy differences proportionally.
“Not to scale” is acceptable if the direction (up/down) and relative magnitudes are still consistent and units are correctly shown.
Yes, if the problem context is general energy changes.
If the prompt explicitly references constant pressure heat or $ \Delta H $, label the y-axis as enthalpy (or energy/enthalpy) to match.
Draw a vertical bracket or double-headed arrow between reactant and product levels and write $ \Delta H $ with its sign and units beside it.
Avoid placing $ \Delta H $ near the curve without indicating which two levels are being compared.
Practice Questions
(2 marks) Sketch an energy diagram for a transformation with . Include axis labels with units and label reactants, products, and .
1 mark: Axes correctly labelled: Energy (kJ mol) on y-axis; Reaction coordinate/progress on x-axis.
1 mark: Products drawn above reactants with clearly indicated.
(5 marks) A student draws an energy diagram with the y-axis labelled “Temperature (°C)” and writes next to the curve, but the reactant and product levels are not labelled. State five specific corrections to make the diagram scientifically acceptable.
Change y-axis label to Energy or Enthalpy, not temperature.
Add appropriate energy units on y-axis (kJ or kJ mol) consistent with the stated .
Label the left level as reactants (initial state).
Label the right level as products (final state).
Ensure products are drawn lower than reactants for negative .
Indicate as a vertical difference between reactant and product levels (arrow/bracket), not floating without reference.
Use “Reaction coordinate/progress” on x-axis (not time).

