AP Syllabus focus: ‘Titrations determine the amount of an analyte in solution using a titrant of known concentration that reacts specifically and quantitatively with the analyte.’
Titration is a core quantitative technique in aqueous chemistry. It links careful volume measurement to reaction stoichiometry, allowing you to determine the amount of a dissolved substance when direct measurement is difficult.
Purpose of titration
A titration is used to determine an unknown amount (moles) or concentration of a dissolved substance by reacting it with a solution of accurately known concentration. The known solution is added until the reaction is judged complete based on a chosen signal (often an indicator or instrument response). The measured volume required is the central experimental data.
In AP Chemistry contexts, titration is most often used when:
The analyte is too dilute, reactive, or impure to weigh reliably.
A reaction can be made to proceed in a predictable, stoichiometric way.
High precision is needed from glassware-based volume measurements.
Core key terms (what you must be fluent with)
Substances and solutions
Titration: A volumetric analytical method in which a solution of known concentration is used to determine the amount or concentration of another solution via a specific, quantitative reaction.
Titrations are designed so the reacting species are measured indirectly by volume, but interpreted chemically.
Analyte: The substance in solution whose amount (moles) or concentration is being determined.
The analyte is typically placed in a flask so it can be mixed thoroughly during addition of the other solution.
Titrant: A solution of known concentration that is added to the analyte until the reaction is complete.
The titrant must be prepared (or verified) so its concentration is trusted; otherwise, the titration cannot produce a reliable result.
Standard solution: A solution with a precisely known concentration, used as a reference in quantitative analysis.
A standard solution is often made by dissolving a known amount of solute and diluting to an exact final volume.
Concentration language used in titration
A titration fundamentally connects moles to measured volume through concentration.
= amount concentration of solution,
= amount of solute,
= solution volume,
In titration lab work, is usually obtained from high-precision glassware (especially a buret).

This close-up of a buret illustrates how delivered volume is read from the meniscus using the buret’s graduations. Because buret markings increase downward, the correct reading comes from the last graduation above the meniscus plus an estimated final digit. Accurate buret readings are the foundation for computing moles via . Source
Common titration setup and measurement terms
Glassware roles
Buret: Delivers the titrant and measures the delivered volume using initial and final readings.
Volumetric pipet: Transfers a fixed, highly precise volume of analyte (or another solution).
Erlenmeyer (conical) flask: Holds the analyte and allows swirling/mixing without splashing.
Volumetric flask: Used to prepare a standard solution to an exact total volume.
Volume and result terms
Initial reading / final reading: The two buret readings used to determine volume delivered (recorded to the correct decimal place for the buret).
Titre (titer): The volume of titrant delivered to reach the chosen completion signal.
Trial: One run of a titration; multiple trials improve confidence and reveal random error.
What “reacts specifically and quantitatively” means
For titration to work as intended, the underlying reaction should meet these criteria:
Specific: The titrant reacts primarily with the analyte (minimal side reactions with other species in the flask).
Quantitative: The reaction proceeds essentially to completion under the conditions used, so the measured titrant volume reflects the analyte amount.
Known stoichiometry: The balanced reaction provides the mole relationship linking titrant to analyte.
Good titration design also emphasizes observable, reproducible detection of completion (chosen to match the chemistry and concentration range), because the measured titre is only meaningful if the stopping point is consistent.
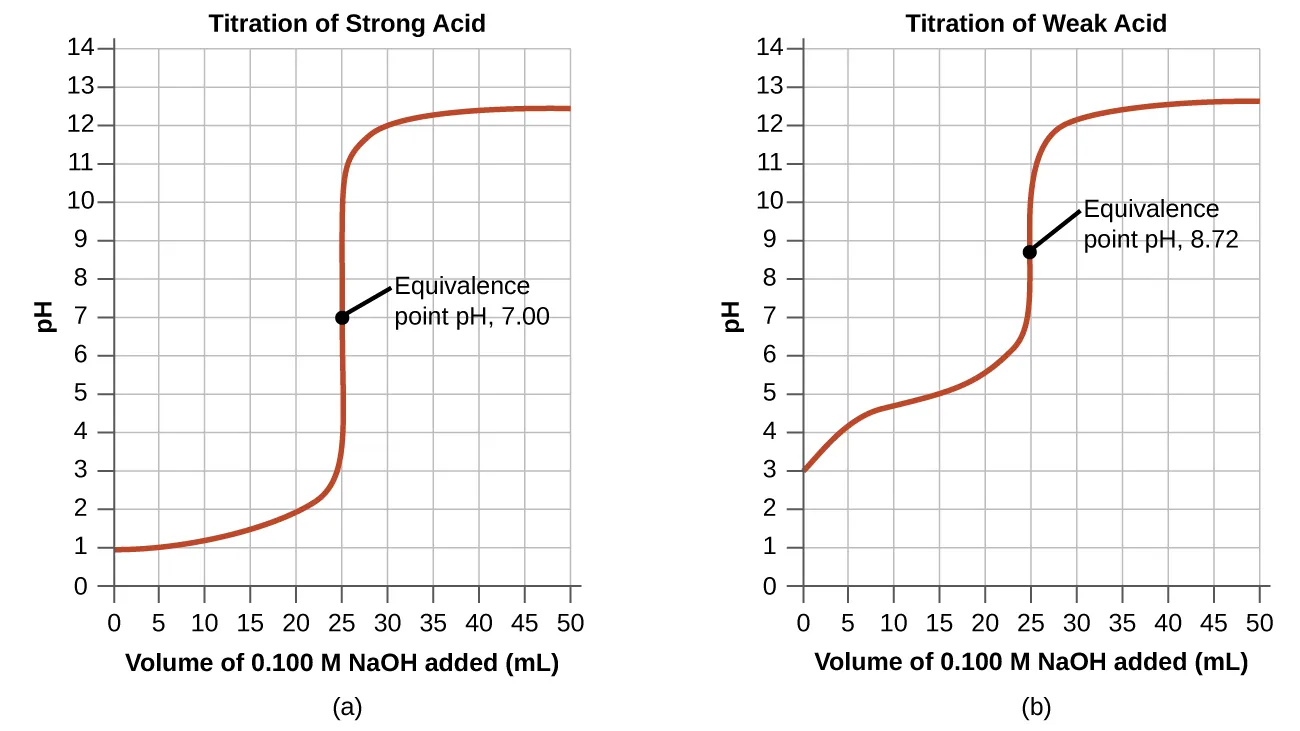
These titration curves show how pH changes as titrant volume increases for two common cases: strong acid–strong base and weak acid–strong base. The steep vertical region corresponds to the rapid pH change near the equivalence point, where stoichiometric amounts of acid and base have reacted. Comparing the two curves highlights that weak-acid titrations have an equivalence-point pH above 7 because the conjugate base hydrolyzes water. Source
FAQ
Use an analytical balance to weigh the solid, dissolve it fully, transfer to a volumetric flask, rinse transfer equipment into the flask, then dilute to the calibration line and invert to mix thoroughly.
Typically it is:
very high purity
stable in air (not hygroscopic, not oxidised by air)
reasonably high molar mass (reduces relative weighing error)
readily soluble to give a stable solution
Because the burette scale measures volume delivered, and small zeroing offsets or non-ideal filling levels are cancelled by using initial and final readings taken from the same burette.
Common causes include temperature differences affecting volume, tiny air bubbles in the burette tip, inconsistent mixing, contamination/dilution from wet glassware, or difficulty judging the completion signal consistently.
Choose a titrant concentration that gives a measurable titre with low relative uncertainty (often a mid-range burette volume), while keeping the reaction rapid and the completion signal sharp enough to judge reproducibly.
Practice Questions
State the purpose of a titration and define the terms analyte and titrant.
Purpose: determine the amount (moles) or concentration of a substance in solution by reaction with a solution of known concentration (1)
Analyte: the solution/species of unknown amount or concentration being determined (1)
Titrant: the solution of known concentration added to react with the analyte (1)
Describe how volumetric glassware is used in a titration to obtain the titre, and state what information must be known for the titrant to be suitable for determining the analyte amount.
Titrant placed in a burette; analyte placed in a conical flask (1)
A fixed volume of analyte is measured using a volumetric pipette (1)
Burette initial and final readings are taken; difference gives volume delivered (titre) (1)
The flask is swirled/mixed during addition to ensure reaction throughout solution (1)
Titrant concentration must be known accurately (standard solution) (1)
Reaction between titrant and analyte must be specific and quantitative with known stoichiometry (1)

