AP Syllabus focus: ‘At equivalence, relate titrant and analyte amounts using the balanced reaction to calculate unknown concentration or moles, assuming the reaction goes to completion.’
Titration problems become straightforward at equivalence because the reacting amounts are fixed by the balanced equation. The key skill is converting measured volume and known concentration into moles, then using stoichiometric ratios to find the unknown.
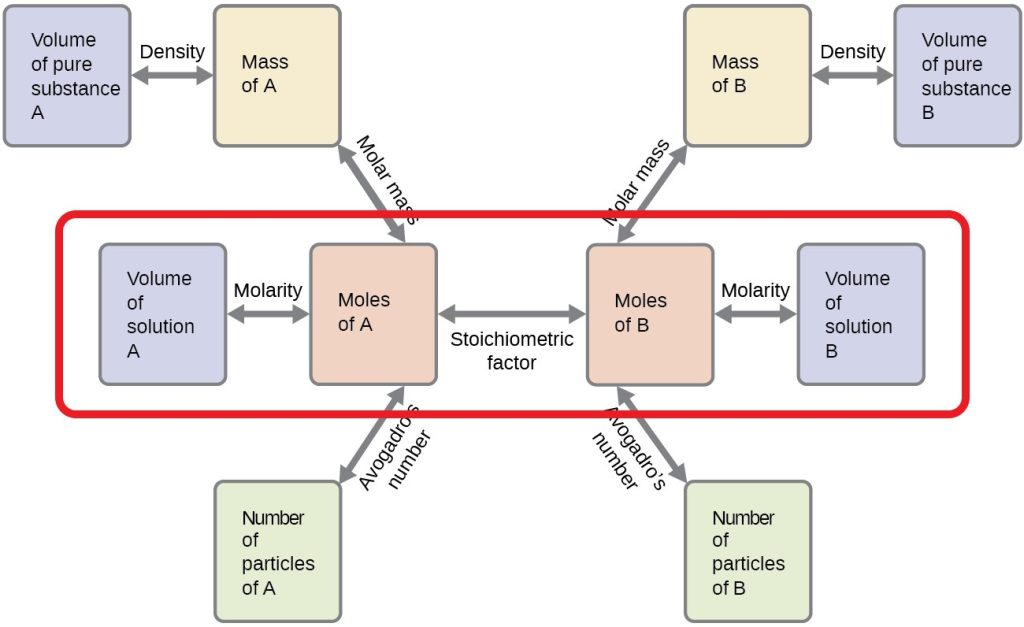
Stoichiometry “road map” for titration-style calculations: volume and molarity are first converted to moles, then a stoichiometric factor (coefficient ratio from the balanced equation) links reactant amounts at equivalence. The diagram also emphasizes that volume alone is not chemically meaningful until it is converted to moles. Source
Stoichiometry at the equivalence point
Core idea
At the equivalence point, the titrant and analyte have reacted in exact proportions dictated by the balanced chemical equation (mole ratio from coefficients). This allows you to determine an unknown moles or concentration from the measured titrant volume.
Equivalence point: The point in a titration at which the titrant added is stoichiometrically equivalent to the analyte present (neither reactant is in excess, based on the balanced reaction).
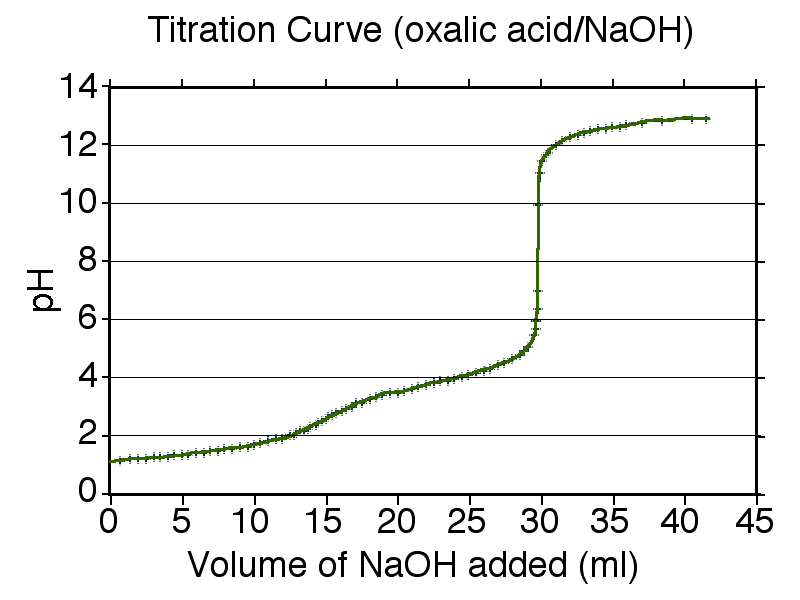
Example titration curve (pH vs. volume of titrant added) showing the sharp rise near the equivalence region, where stoichiometry dictates that reactants have reacted in the exact mole ratio required by the balanced equation. For a polyprotic analyte, multiple equivalence points can appear, reflecting successive neutralization steps. Source
This approach relies on the AP assumption that the reaction is quantitative: it proceeds to completion with a well-defined stoichiometric relationship.
What is “equal” at equivalence (and what is not)
At equivalence:
Moles are related by coefficients, not generally equal.
Volumes are not inherently meaningful without concentration (volume only helps you compute moles).
The reacting species must match the reaction as written (correct chemical forms and states).
Common mistake: assuming even when the coefficients differ (for example, a 2:1 stoichiometry).
Required tools: converting measured data to moles
Titration measurements typically provide titrant volume delivered and titrant molarity.
= amount of substance (moles), mol
= molarity (concentration), mol L
= volume of solution used, L
Use consistent units: convert mL to L before multiplying. In AP-style work, moles are the bridge between solution data and the balanced reaction.
Using the balanced equation to link titrant and analyte
Stoichiometric mapping step
Write the balanced reaction for the titration (the coefficients matter). Then relate amounts using the coefficient ratio.
= moles of titrant that reacted at equivalence, mol
= stoichiometric coefficient of titrant in the balanced equation (unitless)
= initial moles of analyte present (consumed at equivalence), mol
= stoichiometric coefficient of analyte in the balanced equation (unitless)
From this relationship, you can solve for the unknown moles of analyte (or titrant, depending on what is unknown).
From moles to the desired unknown
Once you have moles of analyte:
To find analyte concentration (if analyte volume is given), use the molarity relationship rearranged conceptually: concentration is moles divided by solution volume.
To find mass of analyte (if it’s a solid dissolved to make the solution), convert moles to grams using molar mass.
To find percent composition or purity, compare moles (or mass) of analyte found to the sample’s stated amount.
Practical checks that keep stoichiometry correct
Confirm the reaction is the one that goes to completion (AP typically frames titrations so it does).
Ensure you use the species actually reacting (for example, if a species is polyprotic, the balanced equation must reflect the intended stoichiometry).
Use appropriate significant figures based on given buret readings and stated concentrations.
The equivalence calculation uses the titrant volume at equivalence (often the final buret reading minus the initial reading), not simply the final reading.
FAQ
Start with the species that has both a measured volume and a known concentration (usually the titrant), because it gives moles directly via $n=MV$.
Write the coefficient ratio explicitly before substituting numbers.
Put coefficients under the matching substances
Keep “moles over coefficient” consistent on both sides
No. Completion refers to stoichiometric consumption of reactants. The pH at equivalence depends on the acid/base strengths, not on the stoichiometry step.
Because molarity is defined per dm$^3$. Using cm$^3$ without conversion gives moles off by a factor of 1000.
Compare orders of magnitude: if the titrant and analyte volumes are similar and the reaction is near 1:1, the concentrations should be similar; large mismatches often indicate a coefficient or unit error.
Practice Questions
(2 marks) In a titration, of an acid requires of NaOH to reach equivalence. The reaction is 1:1. Calculate the acid concentration.
Converts titrant volume to dm and finds moles NaOH using . (1)
Uses 1:1 stoichiometry to set moles acid = moles NaOH, then divides by to get concentration. (1)
(5 marks) of HSO is titrated with NaOH. Calculate the NaOH volume at equivalence. Use the balanced stoichiometry.
Finds moles HSO from with correct volume conversion. (1)
Writes/uses correct stoichiometric ratio: HSO reacts 1:2 with NaOH. (2)
Calculates required moles NaOH from the ratio. (1)
Converts moles NaOH to volume using and reports in cm with sensible sig figs. (1)

