AP Syllabus focus: ‘Use stoichiometric ratios to determine the limiting reactant and predict theoretical product amounts when reactants are not in exact proportions.’
Limiting-reactant problems apply the idea that a balanced equation is a fixed “recipe.”
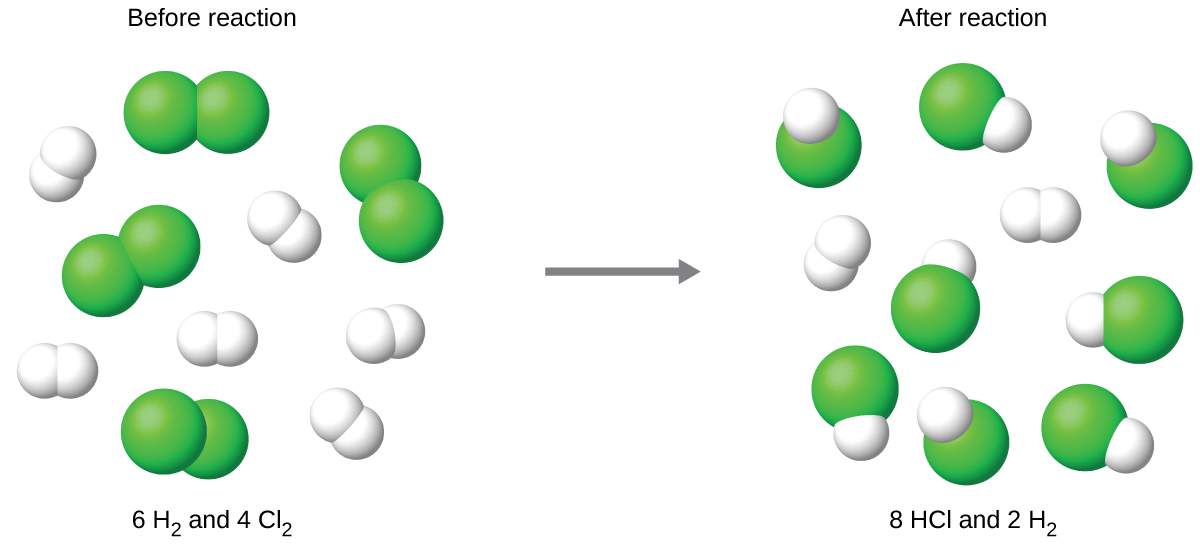
Particle-level illustration of a reaction mixture where one reactant is present in excess and the other is limiting. The diagram emphasizes that the balanced-equation coefficients act like a recipe: once the limiting reactant is fully consumed, product formation stops and any excess reactant remains unreacted. Source
When reactants are mixed in nonstoichiometric amounts, one reactant runs out first, controlling how much product can form.
Core idea: reactions stop when a required reactant is exhausted
In any chemical reaction, reactants are consumed according to the mole ratios set by the balanced chemical equation. If the initial amounts do not match those ratios, the reaction cannot consume all reactants completely.
Key terms you must use precisely
Limiting reactant: The reactant that is completely consumed first, causing the reaction to stop and thereby limiting the amount of product formed.
A limiting reactant is identified by comparing how far each reactant can drive the reaction, not by comparing masses directly (because molar masses differ).
Excess reactant: A reactant present in more than the stoichiometric amount required to consume the limiting reactant; some of it remains unreacted when the reaction stops.
In AP Chemistry contexts, “in excess” means left over after completion (not merely “a larger starting mass”).
Theoretical yield: The maximum amount of product predicted by stoichiometry from the limiting reactant, assuming the reaction goes to completion as written.
Theoretical yield is a prediction from the balanced equation under ideal completion, before considering experimental loss or side reactions.
How to determine the limiting reactant (application workflow)
A robust method is to convert each reactant into the same comparison basis using the balanced equation.
Method A: compare “moles of product possible” from each reactant
Write the balanced equation (coefficients are essential).
Convert each given reactant amount to moles.
For each reactant, use the coefficient ratio to calculate the moles of a chosen product (or any common species) that could form if that reactant were used up.
The reactant that produces the smallest predicted amount of that product is the limiting reactant.
Use only the limiting reactant amount (with mole ratios) to find the theoretical yield of any product.
If asked, determine excess remaining by subtracting the amount of excess reactant consumed from the initial excess amount.
This approach works for any reaction (including those with multiple products) because the limiting reactant restricts the entire reaction extent.
Method B: compare “reaction extent” using coefficients
Conceptually, you can compare how many “sets” of the balanced equation each reactant can supply:
Minimal schematic illustrating the limiting-reagent concept by comparing how available reactant amounts relate to required stoichiometric ratios. It supports the shortcut logic behind extent-style comparisons such as to determine which reactant is depleted first. Because it is a simplified graphic, it works best as a conceptual cue alongside worked examples. Source
Compute for each reactant (in moles).
The smallest value corresponds to the limiting reactant (fewest complete reaction “sets” possible).
Multiply that smallest extent by product coefficients to get product moles.
This is often efficient when several reactants are present, but you must still start from a correctly balanced equation.
Predicting theoretical product amounts (what you’re really doing)
Once the limiting reactant is known, every product amount is fixed by stoichiometry.
You are applying conservation of atoms through the equation’s coefficients: the limiting reactant determines how many times the reaction “can happen.”
= moles of a specified product (mol)
= moles of the limiting reactant (mol)
= coefficient of the product in the balanced equation (unitless)
= coefficient of the limiting reactant in the balanced equation (unitless)
To report theoretical yield in grams (if required), you would convert product moles to mass using molar mass; the limiting-reactant logic remains the central step.
Excess reactant remaining (leftover) and why it matters
After identifying the limiting reactant, you can determine how much excess reactant was actually used:
Use the limiting reactant moles and the coefficient ratio to compute moles of excess reactant consumed.
Leftover excess reactant initial moles consumed moles (convert units afterward if needed).
Excess can be important for lab design and interpretation: it helps ensure complete consumption of a costly reactant, but it also affects purification and mass of mixture after reaction.
Common pitfalls that cost points
Using unbalanced equations (incorrect coefficients give incorrect limiting reactants).
Comparing masses directly rather than converting to moles.
Assuming the reactant with the smaller numerical amount is limiting without using mole ratios.
Forgetting that coefficients apply to moles (or particles), not grams.
Calculating product from the excess reactant after the limiting reactant is found; theoretical yield must come from the limiting reactant only.
FAQ
They are algebraically equivalent.
Comparing product moles converts each reactant to a common outcome; comparing $n/\nu$ directly compares how many full “reaction sets” each reactant can supply.
Yes, unless the problem explicitly guarantees only one reactant can possibly limit.
“Excess” is sometimes qualitative; stoichiometric confirmation prevents choosing the wrong limiting reagent.
No.
Any valid product comparison uses the same balanced-equation ratios, so the reactant that yields the smallest reaction extent remains the limiting reactant.
Convert to moles or compare consistently in particles using coefficients.
Because coefficients apply to particle counts, you may work in particles provided every species is treated in the same unit.
Each reactant gives its own maximum possible reaction extent.
The smallest extent identifies the limiting reactant; all other reactants are excess to varying degrees, and leftovers are found by subtracting what that limiting extent consumes.
Practice Questions
(3 marks) For , a mixture contains mol and mol . Identify the limiting reagent and state the theoretical amount (in mol) of formed.
Correct comparison using ratio and identification of as limiting (1)
Moles of from limiting reagent: (1)
Final value with unit (mol) stated clearly (1)
(6 marks) For , mol is mixed with mol . Determine (i) the limiting reagent, (ii) the theoretical amount (mol) of formed, and (iii) the amount (mol) of the excess reagent remaining.
Use of stoichiometric requirement (1)
Identification of as limiting (since mol would require mol ) (1)
Theoretical from mol : mol (2)
Moles consumed: mol (1)
Excess remaining: mol (1)

