AP Syllabus focus: ‘The equivalence point occurs when the analyte is completely consumed; an observable change (often color) that signals this is the endpoint.’
Titrations rely on matching a measurable signal to a chemical completion point. Understanding the difference between equivalence point and endpoint helps you interpret titration results, choose indicators wisely, and minimise experimental error.
Core ideas: what “equivalence” and “endpoint” mean
Equivalence point (chemical condition)
Equivalence point: The point in a titration at which the analyte is completely consumed according to the balanced reaction (stoichiometric completion).
The equivalence point is defined by stoichiometry, not by what you see.
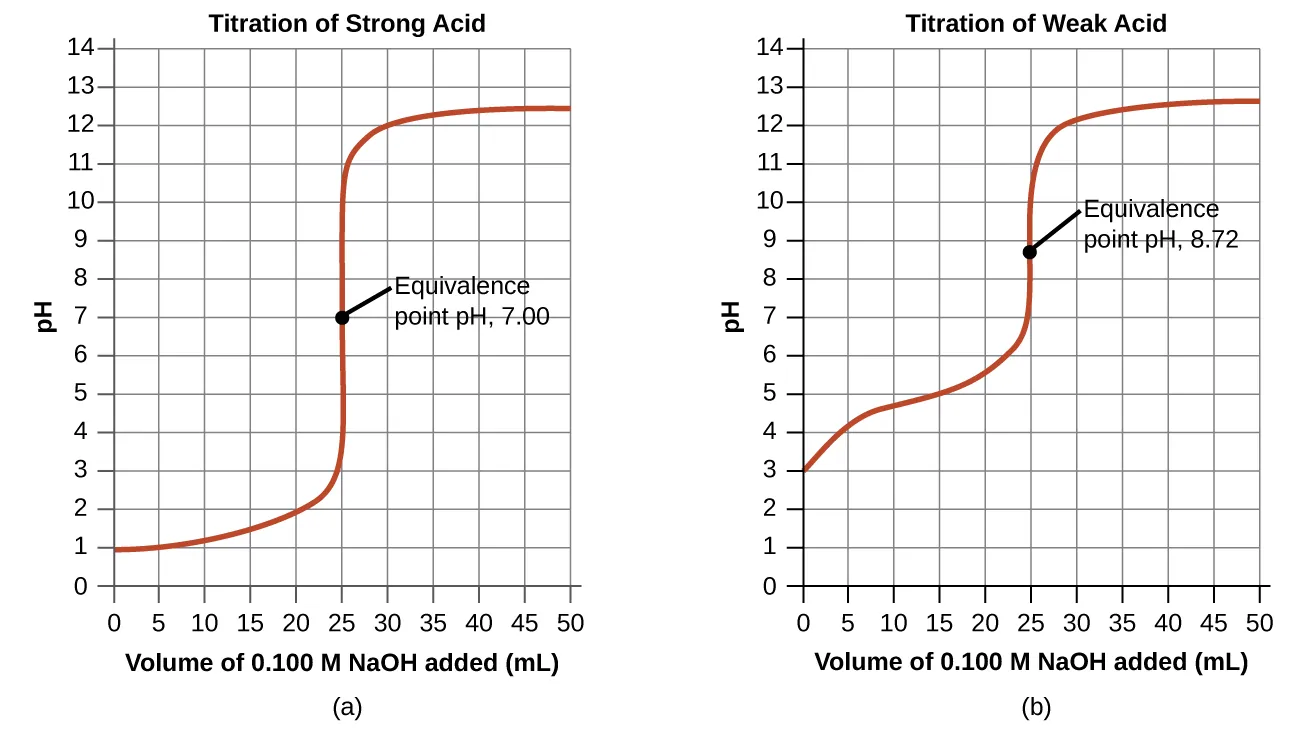
These titration curves plot pH versus volume of titrant added and mark the equivalence point for both a strong acid–strong base titration (equivalence at pH 7.00) and a weak acid–strong base titration (equivalence at pH > 7). The figure highlights the steep pH change near equivalence, which is why small volume differences near this point can produce noticeable indicator color changes. Source
At equivalence, the reacting species have combined in the exact mole ratio required by the balanced equation, so the limiting reactant (typically the analyte) is just used up.
This point is sometimes called the stoichiometric point because it depends only on the reaction’s mole ratio, not on the method used to detect it.
Endpoint (observable signal)
Endpoint: The point in a titration when an observable change (often a color change) indicates that the titration should be stopped.
The endpoint is tied to your detection method (indicator color change, pH meter reading, conductivity change, etc.). In an ideal titration, the endpoint occurs as close as possible to the equivalence point.
A key practical idea is that endpoints are observed, while equivalence points are defined by the chemistry.
Why equivalence point and endpoint may not match
Indicator transition and “indicator error”
Indicator error: The small difference between the volume at the endpoint and the volume at the equivalence point, caused by limitations of the detection method.
Most acid–base indicators change color over a range of pH values, not at a single pH. Because the solution pH changes rapidly near equivalence (for many titrations), the indicator’s transition range must overlap the steep part of the pH change to keep indicator error small.
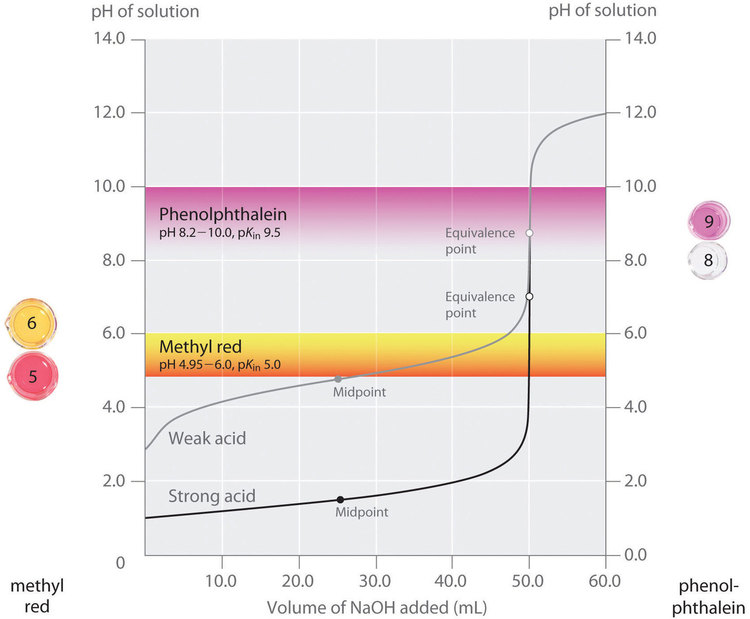
This plot overlays two indicator transition ranges (methyl red and phenolphthalein) onto titration curves, showing where each indicator’s color change occurs relative to the equivalence point. It visually explains indicator error: if an indicator’s transition band does not align with the steep vertical region of the curve, the observed endpoint can occur at a significantly different volume than the true equivalence point. Source
Human and procedural contributions
Even with a good indicator choice, the endpoint can be shifted by technique:
Overshooting: adding titrant past the first persistent color change.
Subjective color judgment: stopping too early or too late because the color change is faint.
Incomplete mixing: local regions of high titrant concentration briefly change color before the bulk solution reaches the endpoint.
Lighting and glassware: background color, dirty flasks, or tinted solutions can mask the transition.
These issues change the observed endpoint without changing the true equivalence point.
Connecting the syllabus statement to titration practice
The syllabus emphasis is that the equivalence point occurs when the analyte is completely consumed, and that an observable change signals the endpoint. In practice, you use an observable endpoint as a proxy for the chemical equivalence point, and you design the titration so the proxy is reliable.
Choosing an indicator to make endpoint ≈ equivalence
To make the endpoint closely match equivalence:
Select an indicator whose transition range falls within the steep region of the titration’s pH change.
Prefer indicators with a clear, high-contrast color change.
Use the minimum effective amount of indicator to reduce its influence on solution chemistry (especially in very dilute titrations).
Recognising a proper endpoint in the lab
A high-quality endpoint is typically defined operationally as:
The first permanent appearance of the indicator’s final color (often required to persist for a short time while swirling).
A color change that is uniform throughout the solution, indicating thorough mixing.
When done carefully, the endpoint can be a close experimental approximation to the equivalence point, even though they are conceptually different.
Common misconceptions to avoid
“The color change is the equivalence point.” The color change marks the endpoint, not the stoichiometric completion.
“Equivalence always occurs at pH 7.” The equivalence point depends on the reaction system; pH at equivalence can vary widely.
“Any indicator works if you’re careful.” Careful technique helps, but indicator choice determines whether the endpoint can realistically coincide with equivalence.
FAQ
Common alternatives include using a pH probe, conductivity sensor, or spectrophotometer.
These methods can reduce subjectivity but still require a defined criterion for “endpoint.”
An early endpoint occurs before equivalence; a late endpoint occurs after equivalence.
Either shifts the measured titre volume, introducing a systematic bias.
Strongly coloured analytes or products can mask the indicator transition.
A sharper-contrast indicator or instrumental detection can help.
In principle, adding indicator introduces additional acid/base species.
In practice, using only a few drops makes this effect negligible for typical titrations.
It defines an operational endpoint to avoid stopping at a transient colour caused by incomplete mixing.
This improves consistency between trials.
Practice Questions
(1–3 marks) State the difference between the equivalence point and the endpoint in a titration, and give one reason they may not coincide.
Equivalence point defined as analyte completely consumed / stoichiometric completion (1)
Endpoint defined as observable change (often colour) used to stop titration (1)
One valid reason for mismatch, e.g. indicator changes over a pH range / subjective judgement / overshooting (1)
(4–6 marks) In an acid–base titration using a colour indicator, describe how you would ensure the endpoint is as close as possible to the equivalence point. Include two sources of error that can shift the endpoint and how to reduce them.
Choose indicator with transition range within steep part of pH change near equivalence (1)
Describe stopping at first permanent, uniform colour change while swirling (1)
Error source 1 identified (e.g. overshooting / poor mixing / subjective colour judgement) (1)
Mitigation for error 1 (e.g. add dropwise near endpoint / swirl thoroughly / use white tile) (1)
Error source 2 identified (1)
Mitigation for error 2 (1)

