AP Syllabus focus: ‘Write and balance molecular and ionic equations, then cancel spectator ions to produce a balanced net ionic equation for the reaction.’
Net ionic equations streamline aqueous reactions to show only the species that actually change. This page focuses on how to write, balance, and justify net ionic equations while preserving mass balance and charge balance.
What a net ionic equation shows (and what it omits)
A net ionic equation represents the chemical change in solution by including only the particles that participate in the reaction. Ions or molecules that appear unchanged on both sides are removed, because they do not contribute to the chemical transformation.
Key terms you must use correctly
Spectator ion: An ion present in solution that appears in the same form on both sides of the complete ionic equation and therefore is cancelled to form the net ionic equation.
Spectator ions still matter for conductivity and the full composition of the solution, but they are not part of the reaction event being modelled.
Strong electrolyte: A soluble ionic compound or strong acid/base that dissociates essentially completely into ions in water and is therefore written as separate aqueous ions in a complete ionic equation.
Because strong electrolytes are written as ions, correctly identifying them is essential to writing the complete ionic equation before cancelling spectators.
Required workflow: molecular → complete ionic → net ionic
1) Write a balanced molecular equation first
Start with correct formulas and state symbols , , , and . Balance the molecular equation for atoms. This step prevents mistakes later when splitting into ions.
Include correct physical states; they determine whether something is split into ions.
Ensure the equation reflects what actually forms (e.g., an insoluble solid, a gas, or water).
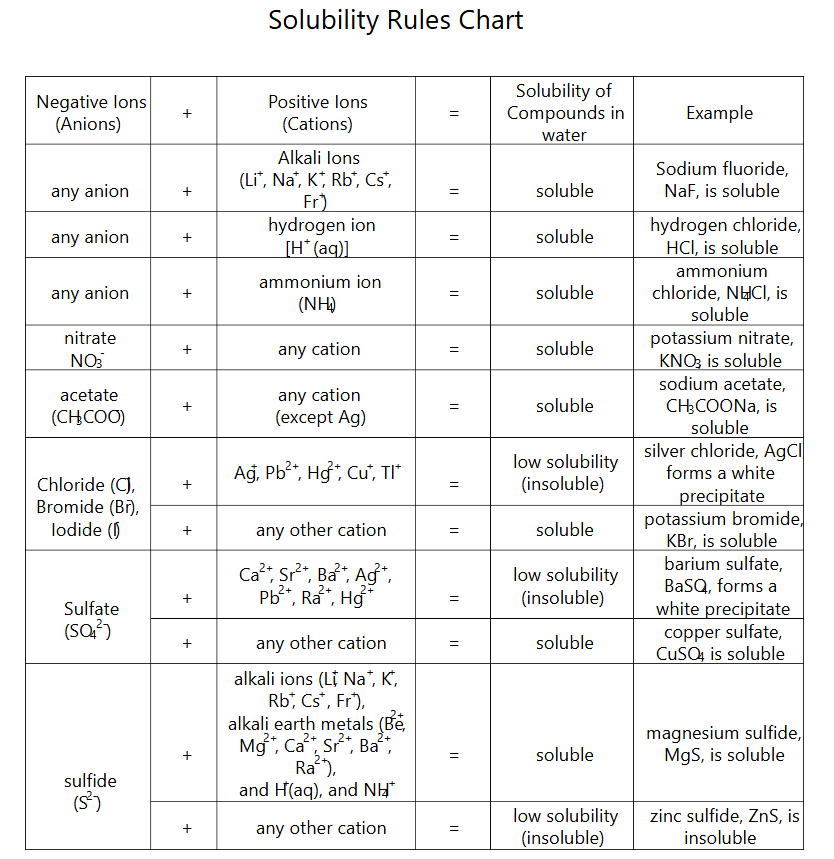
Solubility-rules table summarizing which ionic combinations are typically soluble in water and which commonly produce low-solubility precipitates. This kind of chart is used to predict when a reaction has a driving force (e.g., formation of an insoluble solid), which then guides the correct states in the molecular equation and which species remain intact in the ionic equations. Source
2) Convert to a complete ionic equation
Rewrite the molecular equation by splitting only strong electrolytes in aqueous solution into ions.
Split:
Soluble ionic compounds written as
Strong acids and strong bases written as
Do not split:
Solids , liquids , and gases
Weak electrolytes (they are mostly molecular in water)
Molecular compounds (unless reacting as strong acids/bases)
Maintain coefficients when splitting; coefficients apply to each ion produced.
3) Cancel spectator ions to obtain the net ionic equation
Remove species that are identical on both sides (same formula, charge, and phase).
Cancel only when the species matches exactly (e.g., cancels only with ).
After cancelling, the remaining equation is the net ionic equation.
4) Check that the net ionic equation is balanced
A correct net ionic equation must conserve:
Atoms: each element has the same count on both sides.
Charge: total charge on the left equals total charge on the right.
If atoms balance but charge does not, you have not written or cancelled ions correctly.
Balancing expectations for net ionic equations
Net ionic equations are typically balanced by inspection once written correctly, but you must verify both atom and charge conservation. In many aqueous reactions, charge balance is the fastest way to catch an error (such as forgetting a coefficient or cancelling an ion that should remain).
= total charge on the reactant side (unitless)
= coefficient (count) of ionic species (unitless)
= charge number of ionic species (unitless)
= total charge on the product side (unitless)
Use this charge-accounting idea as a consistency check; you are not expected to compute charges formally every time, but the equality must hold.
Common pitfalls that lower accuracy
Splitting species incorrectly
Writing a precipitate as ions (incorrect): insoluble products stay as .
Splitting weak acids/bases (often incorrect in net ionic context): they are typically written as molecules unless specifically treated as ions by the prompt.
Cancelling incorrectly
Cancelling ions that are not identical (different phase or different charge).
Cancelling a species that is produced or consumed (it must appear unchanged on both sides to be a spectator).
Missing states
States determine whether a species can be cancelled and whether it should be written as ions. Include them throughout, especially in the molecular equation and the final net ionic equation.
When net ionic form is the most appropriate
Net ionic equations are most informative when:
The reaction occurs in aqueous solution and involves ions.
You want to highlight the driving force (formation of a precipitate, gas, or weakly ionised product such as water).
They are less useful when all species remain as strong electrolytes with no net change (in that case, cancellation can leave no reaction).
FAQ
Use the species’ behaviour in water.
Strong electrolytes (soluble ionic salts, strong acids, strong bases) are written as ions.
Weak acids/bases are usually written molecularly.
If the prompt provides a “complete ionic equation,” follow its conventions.
No. Liquid water is written as $H_2O(l)$ in net ionic equations.
Only aqueous ions explicitly present (or formed by dissociation of strong electrolytes) are written as ions.
That indicates no chemical change in aqueous solution.
This commonly happens when mixing two soluble salts whose ions remain aqueous with no precipitate, gas, or weak electrolyte formed.
Yes, if they appear unchanged on both sides with the same charge and phase, e.g., $NO_3^-(aq)$.
Do not break polyatomic ions into atoms; treat them as intact ions.
Check three features:
Atom balance for each element
Total charge balance left vs right
Presence of a driving-force product: $(s)$, $(g)$, or $H_2O(l)$
If all species are $(aq)$ strong electrolytes, re-check whether a reaction should occur at all.
Practice Questions
Question 1 (3 marks) Aqueous solutions of and are mixed. Write the balanced net ionic equation, including state symbols.
Writes correct ions and product with states: and form (1)
Balanced atoms (1)
Balanced charge (1)
Question 2 (6 marks) Aqueous reacts with aqueous .
(a) Write the balanced molecular equation with state symbols. (2 marks)
(b) Write the balanced complete ionic equation. (2 marks)
(c) Write the balanced net ionic equation. (2 marks)
Correct formulas and products with states: and (1)
Correct balancing: (1) (b)
Correctly splits strong electrolytes: (2) (c)
Cancels no spectators incorrectly; net ionic shows both processes: (2)

