AP Syllabus focus: ‘A reaction may be written as a balanced molecular, complete ionic, or net ionic equation; the best form depends on the context.’
Chemical reactions can be communicated at different “zoom levels.” In aqueous chemistry, choosing between molecular, complete ionic, and net ionic equations determines whether you emphasise compounds, dissociated ions, or only the species that actually undergo change.
Three common equation forms (and what they show)
Molecular equation
A molecular equation keeps substances written as intact chemical formulas, even if they are dissolved in water.
Molecular equation: A balanced chemical equation that represents reactants and products as neutral formulas (or solids/liquids/gases) rather than as separated aqueous ions.
Use this form when the chemical identity of compounds matters (for example, communicating which reagents were mixed), or when species do not exist primarily as ions in solution.
Complete ionic equation
A complete ionic equation expands aqueous strong electrolytes into their constituent ions, explicitly showing all ions present in solution.
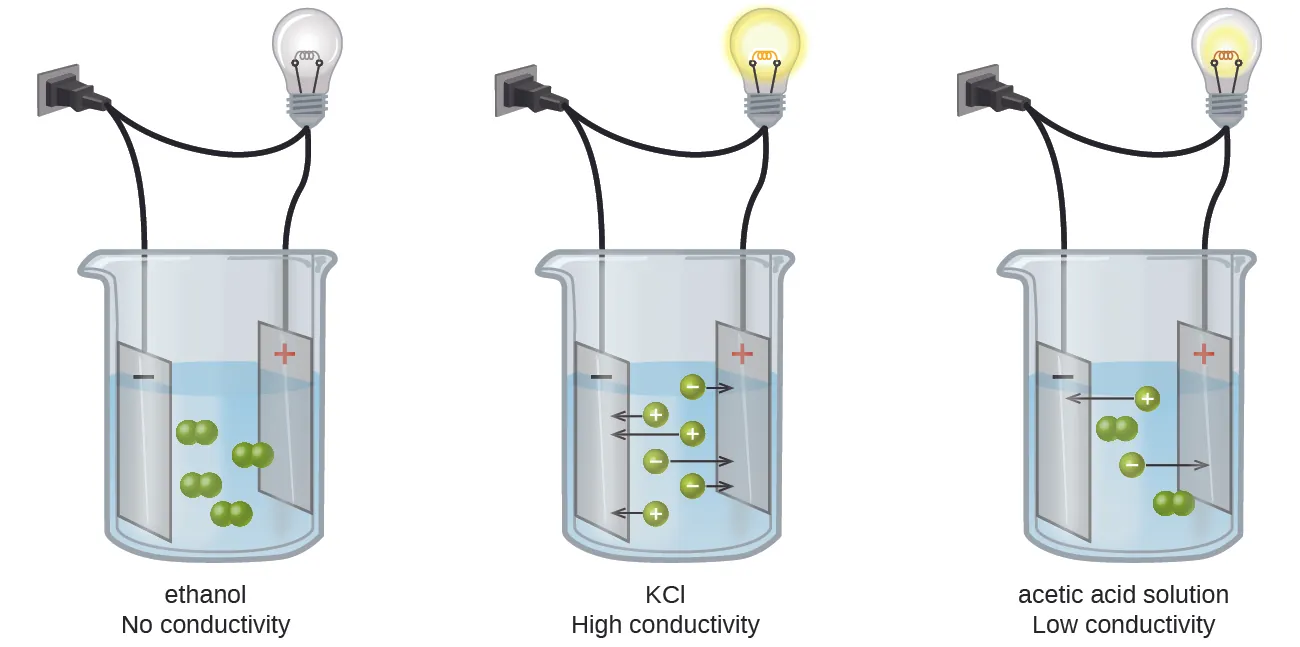
This OpenStax figure contrasts nonelectrolyte, strong-electrolyte, and weak-electrolyte solutions by showing whether particles appear as intact molecules or as separated ions that can carry charge. It reinforces why strong electrolytes are written as ions in a complete ionic equation, while nonelectrolytes remain molecular. Source
Complete ionic equation: A balanced equation in which dissolved strong electrolytes are written as separated ions, while substances that do not dissociate (and solids/liquids/gases) remain written as formulas.
This form is especially useful for visualising what is actually present in beakers before and after mixing, and for detecting ions that appear unchanged.
Net ionic equation
A net ionic equation removes ions that do not participate in the chemical change, leaving only the species that undergo a change in composition, bonding, or phase.
Net ionic equation: A balanced equation showing only the ions/molecules that participate in the reaction, after eliminating spectator ions; it conserves both mass (atoms) and total charge.
Net ionic equations are often preferred when the reaction’s driving force is ionic (such as formation of a precipitate, gas, or weak electrolyte), because they highlight the essential chemical change.
Spectator ions and “what counts as reacting?”
In many aqueous reactions, some ions are present but unchanged.
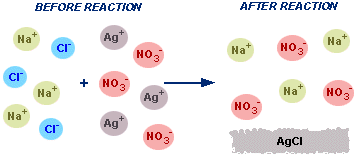
This University of Wisconsin tutorial uses a before-and-after particle view of mixing aqueous salts to show which ions remain unchanged (spectators) and which ions form a new substance. The visuals support the logic of canceling identical ions from the complete ionic equation to obtain the net ionic equation. Source
Identifying these explains why different equation forms can describe the same process.
Spectator ion: An ion that appears in the reaction mixture but undergoes no chemical change, appearing identically on both sides of the complete ionic equation.
A key idea is that the “best” equation form depends on context:
If you are tracking which solutions were combined, the molecular equation may be clearest.
If you are tracking all dissolved species, the complete ionic equation is most explicit.
If you are focusing on the chemical change that occurs regardless of counterions, the net ionic equation is most informative.
What gets written as ions (and what stays together)
When moving from molecular to ionic representations, only certain dissolved substances are written as separated ions:
Split into ions (typical): soluble ionic compounds and strong acids/bases (they exist mostly as ions in water).
Do not split (typical): solids (s), liquids (l), gases (g), and species that remain largely molecular in water (commonly weak electrolytes).
Keep meaningful units together: polyatomic ions (like sulfate) are usually kept as intact ions when they appear as ions in solution.
Correct equation choice also relies on including appropriate phase labels (such as aqueous vs solid), since many net ionic reactions are driven by forming a new phase (for example, an insoluble solid).
= phase labels indicating aqueous solution and solid
= ions present but unchanged in this reaction
= shows only species undergoing the chemical change
Interpreting what each form communicates
Each representation encodes a different chemical message:
Molecular: emphasises reactant/product formulas and is often closer to the lab setup (what bottles were used).
Complete ionic: emphasises the full ionic inventory in solution and makes charge-carrying species explicit.
Net ionic: emphasises the minimal chemical event; it is especially useful for comparing reactions that differ only by counterions (since spectators are omitted).
FAQ
Only for species that exist predominantly as ions (strong electrolytes). If a substance remains largely molecular in water, it is typically kept together.
They omit spectator ions, keeping only species that undergo change, which removes counterions that do not affect the reaction’s chemistry.
Write them as intact ions (e.g. $\mathrm{SO_4^{2-}}$) when they remain unchanged; only their overall charge and formula matter unless they are chemically altered.
Yes. Different soluble salts can supply the same reacting ions; after removing spectators, the same net ionic equation can result.
When the driving force is best described by ions forming a new phase or species (e.g. an insoluble solid), making spectators irrelevant to the core change.
Practice Questions
(1–3 marks) State which equation form (molecular, complete ionic, or net ionic) is most useful for identifying spectator ions, and explain why.
Identifies complete ionic equation (1)
Explains that all aqueous ions are shown explicitly (1)
Links to spectator ions appearing unchanged on both sides (1)
(4–6 marks) Three equations describe the same aqueous reaction: A.
B.
C.
(i) Classify A, B, and C as molecular/complete ionic/net ionic.
(ii) Identify the spectator ions.
(iii) One equation is not an acceptable net ionic equation; state which and why.
A is molecular; B is complete ionic; C is intended net ionic (2 marks: 1 each for two correct classifications, +1 for the third)
Spectator ions: and (2)
C is unacceptable because spectators were not fully removed / appears on both sides so should cancel (1)

