AP Syllabus focus: ‘Balanced chemical equations (in any form) can be translated into consistent symbolic particulate representations.’
Balanced equations are more than bookkeeping: they can be converted into particle-level pictures that show what is present before and after a process.
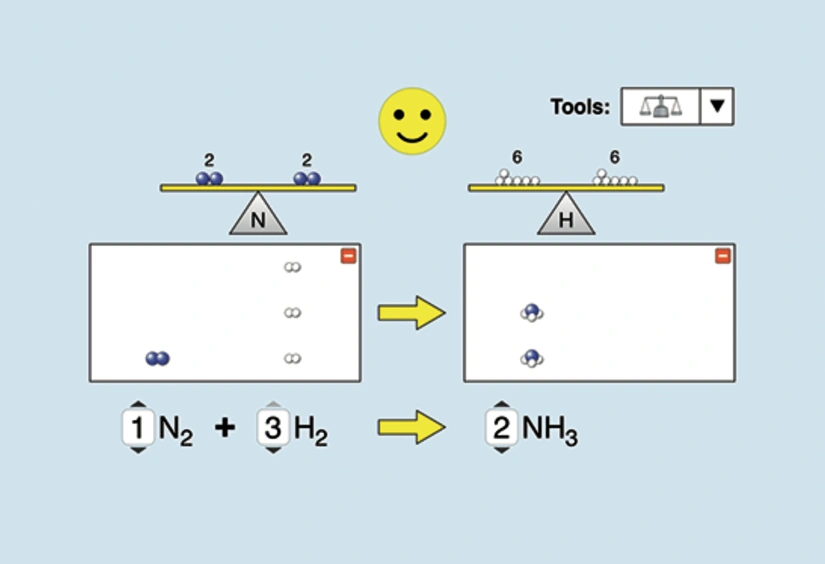
An interactive balancing-equations activity (ACS-hosted, PhET-based) that helps students connect symbolic coefficients to particle-level counts. It reinforces that coefficients control the relative number of molecules/formula units shown in a particulate diagram, while the identities of the species remain fixed by the formulas. Source
A correct particulate model must match the equation’s identities, ratios, and states.
What a particulate model represents
A particulate model is a visual, symbolic depiction of atoms, molecules, and/or ions that is consistent with a chemical equation. It does not need to be to scale, but it must be chemically consistent.
Particulate representation — a diagrammatic model that uses particles (spheres, clusters, or icons) to show the relative numbers and types of species in a system, consistent with a balanced chemical equation and stated phases.
Because the diagram is constrained by the balanced equation, it must reflect:
Identity: correct chemical formulas (including subscripts)
Ratio: correct relative amounts from coefficients
Conservation: same count of each type of atom before and after
Phase: solid, liquid, gas, or aqueous behavior as indicated
Translating a balanced equation into particles
Step 1: Read formulas as “particle types”
Each distinct formula in the equation corresponds to a distinct particle type you must draw:
Monatomic species (e.g., ): single spheres
Diatomic elements (e.g., , , , , , , ): pairs bonded together
Molecular compounds (e.g., , ): bonded groups with the correct atom count and connectivity implied by the formula
Ionic compounds: represented as repeating cation/anion units in a crystal for solids, or as separated ions if the equation is written in ionic form
A common constraint is that subscripts are part of particle identity, not adjustable “counts.” For example, must always appear as a three-atom unit (2 H atoms bonded to 1 O atom) in a molecular representation.
Step 2: Use coefficients as the particle ratio
The coefficients tell you the simplest whole-number ratio of particles (molecules, formula units, or ions as written) that react and form products. Your drawing should include particle counts proportional to these coefficients.
Guidelines for diagramming with coefficients:
You may scale all coefficients by the same factor to make a clearer picture, but you must keep the same ratio.
If a coefficient is “1,” you still draw that species (one particle, or a consistent scaled amount).
Coefficients apply to the entire formula unit; for , draw two molecules, not one molecule with doubled subscripts.
Step 3: Ensure atom-by-atom conservation in the picture
A balanced equation guarantees that each element has the same number of atoms on both sides, so the particulate model must show the same conservation visually.
Checks you can perform directly on a particulate representation:
Count each element’s atoms in the reactant particles you drew.
Count each element’s atoms in the product particles you drew.
The totals must match for every element; otherwise the particulate model contradicts the balanced equation.
Step 4: Match the phase labels to particle spacing and arrangement
Phase symbols carry structural meaning that a particulate diagram should reflect:
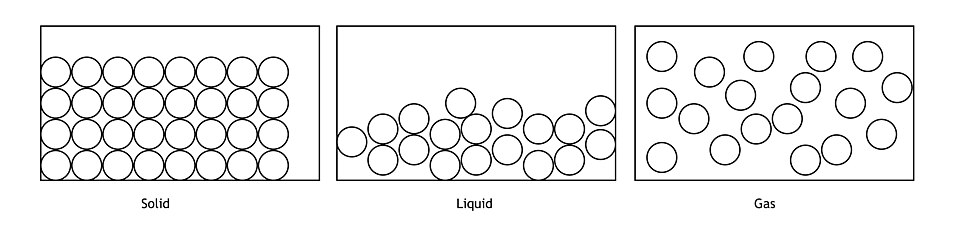
Particle-level models of a solid, liquid, and gas, emphasizing how particle spacing and ordering change with phase. This supports translating phase symbols like , , and into visually distinct arrangements in a particulate representation. Source
Gas, (g): particles far apart with lots of empty space; random distribution
Liquid, (l): particles close together but disordered; able to move past each other
Solid, (s): particles in a fixed, closely packed arrangement; for ionic solids, depict an extended lattice pattern

A crystal-structure diagram of sodium chloride showing a repeating ionic lattice, with Na and Cl ions arranged in a regular 3D pattern. This is a model for how an ionic solid labeled should appear in a particulate representation: ordered, closely packed, and extended rather than dispersed. Source
Aqueous, (aq): particles dispersed throughout solution; species are surrounded by water in reality, but most AP-level particulate diagrams show the solute particles distributed in the liquid phase
When the process forms a new phase, the particulate model should show that change. For example, formation of a solid product in water is depicted as clustered particles separated from the dispersed aqueous particles (often settling at the bottom conceptually), consistent with a phase label of (s).
“In any form”: aligning the diagram with the equation’s representation
Balanced chemical equations can be written in different forms, and the particulate model must follow the form given:
If the equation shows molecules, depict intact molecular units as written.
If the equation shows ions (ionic form), depict separated ions with the charges implied by their formulas.
The key rule is consistency: your particles must represent exactly the chemical species written in the balanced equation, not additional “assumed” species. If the equation is already balanced, your particle counts should require no extra particles to “make it work.”
Common consistency checks (high-utility for AP diagrams)
Use these quick checks to verify a particulate model derived from a balanced equation:
Ratio check: particle counts reduce to the coefficient ratio.
Subscript check: each particle has the correct internal atom count.
Element conservation check: total atoms of each element match before and after.
Phase check: spacing/aggregation matches (g), (l), (s), (aq).
No impossible fragments: avoid drawing partial molecules that are not in the equation (unless the equation itself is written to include them).
FAQ
Draw the polyatomic ion as a single grouped unit (a “cluster”) and keep it intact throughout unless the equation explicitly changes it.
You can:
Use one composite symbol for the whole ion
Keep the same cluster identity on both sides to show it is unchanged
Only include explicit $H_2O$ particles if water is written as a reactant/product in the balanced equation.
If the equation uses (aq) without writing $H_2O$, you may depict a generic liquid background rather than individual water molecules, to avoid adding species not present in the equation.
No. They are symbolic models aimed at conserving identity and number, not depicting scale or electronic structure.
What must be correct is:
Which particles are present
Their ratios
Their phase-appropriate distribution
Divide all coefficients by a common factor if possible to reach the simplest ratio, then draw that ratio.
If already simplest, you may draw a smaller proportional set (e.g., half as many) only if it keeps whole particles and preserves the ratio; never change subscripts to compensate.
Use a fast audit:
Reduce particle counts to a ratio and compare with coefficients
Count each element’s atoms on both sides
Check diatomic elements are paired ($H_2$, $N_2$, $O_2$, $F_2$, $Cl_2$, $Br_2$, $I_2$)
Ensure phases look correct (gas spaced out; solids clustered)
Practice Questions
(2 marks) For the balanced equation , state the number of each type of molecule you would show in a correct particulate diagram for reactants and products.
1 mark: Reactants show 2 molecules of and 1 molecule of .
1 mark: Products show 2 molecules of .
(6 marks) Consider . Describe what a consistent particulate representation should show, including (i) the correct particle identities, (ii) relative numbers, and (iii) how the phases should be depicted.
1 mark: Identify reactant aluminium as particles in a solid arrangement (closely packed/fixed).
1 mark: Identify chlorine as diatomic molecules, , in the gas phase (pairs, far apart).
1 mark: Use the correct relative numbers: 2 Al units to 3 molecules (or any correct scaled multiple).
1 mark: Identify product as units in a solid arrangement (clustered/lattice-like).
1 mark: Use the correct relative number of product units: 2 units of (or scaled consistently).
1 mark: Explicitly indicate conservation in the diagram description (Al atoms conserved; 6 Cl atoms from become part of ).

