AP Syllabus focus: ‘Chemical equations show atoms rearranged into new combinations, so each element has equal atoms before and after; mass and charge are conserved.’
Chemical equations are bookkeeping tools that track how particles rearrange during a reaction. The key idea is conservation: the same atoms (and total charge, when ions are involved) must appear on both sides of the equation.
Conservation is what “balanced” really means
A chemical reaction reorganizes atoms into new groupings; it does not create or destroy atoms. A balanced chemical equation is one whose coefficients enforce that constraint for every element present.
Balanced chemical equation: A symbolic representation of a reaction in which each element has the same number of atoms in reactants and products, consistent with conservation laws.
Balancing is not an aesthetic step; it is the mechanism by which equations align with physical reality in ordinary chemical processes.
Conserving atoms (and therefore mass)
Because matter is made of atoms that persist through chemical change, the count of each type of atom must be identical before and after reaction. This implies that total mass is conserved in the reacting system (since mass is tied to the identities and numbers of atoms), even though phases, volumes, and observable properties may change.
Atom-by-atom accounting
To check conservation of atoms, you compare element counts on each side:
List each element that appears in the equation.
Count how many atoms of that element are present in the reactants.
Count how many atoms of that element are present in the products.
Adjust coefficients (numbers in front of formulas) until all element counts match.
Subscripts inside a chemical formula are part of the substance’s identity and must not be changed when balancing; changing a subscript would describe a different compound.
= number of atoms of element in the reactants (atoms)
= number of atoms of element in the products (atoms)
This atom-conservation check must be satisfied for every element present, not just the ones that are easiest to count.
Coefficients vs. formulas
A coefficient scales the entire formula unit:
If you place a coefficient of 2 in front of , you now have 4 H atoms and 2 O atoms total.
If you were to change to , you would be describing hydrogen peroxide, not water, so the equation would no longer represent the same substances.
Conserving charge (especially in ionic equations)
In addition to atoms, electric charge is conserved in chemical processes. This is most visible when writing ionic equations (with explicit ions), but it is still true in any representation: the total charge on the reactant side must equal the total charge on the product side.
Conservation of charge: The total electric charge of the reactants equals the total electric charge of the products in a chemical process.
Charge conservation provides an independent check alongside atom conservation. An equation can have correct atom counts but still be wrong if the total charge does not match (for example, if an ion’s charge is written incorrectly).
= total charge of the reactants (in units of the elementary charge)
= total charge of the products (in units of the elementary charge)
For molecular equations involving only neutral species, charge conservation is usually automatic because both sides have net charge 0. In ionic equations, you must explicitly verify it.
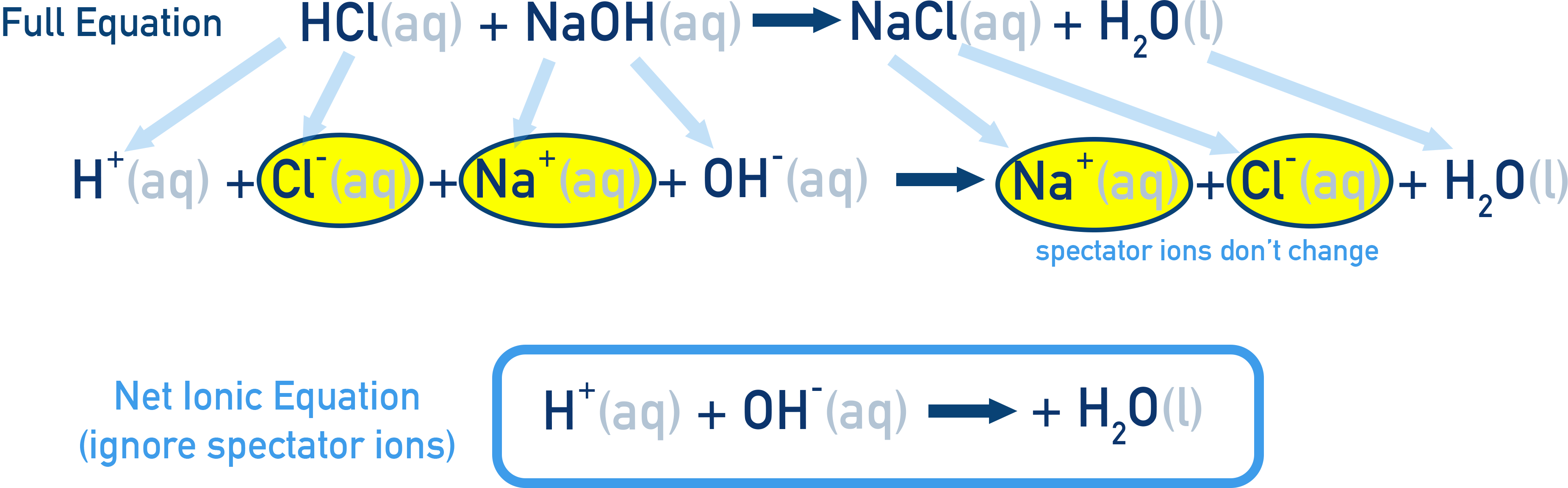
Side-by-side representation of a precipitation reaction written as a molecular equation, a complete ionic equation, and the net ionic equation. By marking spectator ions and showing what cancels, the diagram reinforces that both atom counts and total charge must be conserved even as the representation becomes more chemically focused. Source
Practical charge-check steps
Compute the net charge on each side by summing ionic charges times their coefficients.
Ensure the totals match exactly.
If charges differ, the equation is not physically possible as written (it violates conservation), even if the atom counts appear consistent.
What balancing does and does not do
Balancing enforces conservation constraints without changing the chemistry being described.
Balancing does
Ensures each element has equal atom counts in reactants and products.
Ensures total charge matches on both sides when ions and charges are shown.
Produces coefficients that define the simplest whole-number particle ratios consistent with conservation.
Balancing does not
Indicate reaction speed, extent, or mechanism.
Guarantee that a reaction will occur; it only ensures the written equation is consistent with conservation if it does occur.
Change the identities of substances (so it cannot involve changing subscripts or charges to “make it work”).
High-utility consistency checks students should use
Element inventory check: Every element appearing on one side must appear on the other side; otherwise atoms would be created/destroyed.
Coefficient distribution check: When you change a coefficient, recount all atoms in that formula unit (and charges for ions).
Charge sanity check: Common ions have characteristic charges (for example, group 1 metals are typically in ionic compounds); incorrect charges often lead to charge nonconservation.
Whole-number requirement: Chemical equations are conventionally scaled to whole-number coefficients because they represent ratios of discrete particles.
FAQ
Subscripts define the compound’s identity (its fixed ratio of atoms). Changing them turns one substance into a different substance.
Balancing must preserve the substances and only change how many formula units participate.
If each element’s atom count is unchanged, the total mass is unchanged because atomic masses are properties of the atoms present.
Apparent mass changes in labs usually come from matter entering or leaving an open container.
Multiply each ion’s charge by its coefficient and sum for each side.
If the sums match, charge is conserved; if not, the equation cannot be correct as written.
No. Conservation applies to the total charge of each side, not to each species.
Ions can combine into neutral compounds or form different ions, as long as the net charge is conserved.
In ordinary chemical reactions, mass is conserved to an excellent approximation.
In nuclear processes, tiny mass changes can occur due to mass–energy equivalence; these are outside typical chemical equation balancing.
Practice Questions
Question 1 (1–3 marks) State two conservation laws that a correctly balanced chemical equation must obey.
Conservation of atoms: each element has the same number of atoms on both sides (1)
Conservation of mass: total mass of reactants equals total mass of products in the system (1)
Conservation of charge: total charge of reactants equals total charge of products (1) (Award any two valid laws for full credit if only two are requested; if a student lists all three, credit the best two unless the question wording allows two.)
Question 2 (4–6 marks) The ionic equation below is not balanced. Add coefficients to balance it and then show that both atoms and charge are conserved.
Correct coefficients: (2)
Atom conservation shown for Al: 2 Al on both sides (1)
Atom conservation shown for S (or groups): 3 S on both sides (1)
Atom conservation shown for O: 12 O on both sides (1)
Charge conservation shown: left charge , right charge (1)

