AP Syllabus focus: ‘Understanding or interpreting phase diagrams is excluded from assessment; focus instead on particulate models that explain phase properties and behavior.’
Phase diagrams are a common way to summarise phase changes, but AP Chemistry testing avoids them here. Instead, you should explain phase behaviour using particle-level reasoning about spacing, motion, and attractions.
What this syllabus point means
You are expected to recognise that phase diagrams are not an assessed tool for this unit. In practice, that means you should not spend study time mastering diagram features (lines, regions, special points) for exam purposes. Your explanations should come from particulate models that connect microscopic particle behaviour to macroscopic observations.
What you should do instead of using phase diagrams
On AP questions about solids, liquids, and gases in this topic area, prioritise:
Particle spacing (very close in solids and liquids; far apart in gases)
Particle motion (vibration in solids; sliding/flow in liquids; rapid random motion in gases)
Relative strength of attractions between particles (stronger attractions favour condensed phases)
Energy and temperature changes as drivers of changes in motion and the ability to separate particles
Pressure effects on gases described qualitatively (compression brings particles closer, increasing collision frequency)
Particulate models you should be able to use
A particulate model is a drawing or description showing many particles and how they are arranged and moving. A strong AP-level particulate explanation includes both structure and motion.
Particulate model: A representation that shows particles (atoms, molecules, or ions), their spacing/arrangement, and their motion to explain macroscopic properties of matter.
Use particulate models to communicate ideas such as:
In a solid, particles are in close contact and organised; motion is limited to vibrations, so shape and volume are fixed.
In a liquid, particles remain in close contact but rearrange; this allows flow while maintaining nearly fixed volume.
In a gas, particles are far apart and move freely; weak attractions and large spacing lead to no fixed shape or volume.

A kinetic-molecular-theory sketch shows gas particles widely spaced inside a container, moving in straight-line segments and colliding with the walls. This visual supports qualitative explanations of gas pressure (wall collisions) and why gases are compressible (mostly empty space between particles). Source
Linking particle ideas to observable phase behaviour
When you explain phase behaviour without phase diagrams, the most important causal links are:
Heating increases average particle kinetic energy, increasing motion and helping overcome attractions.
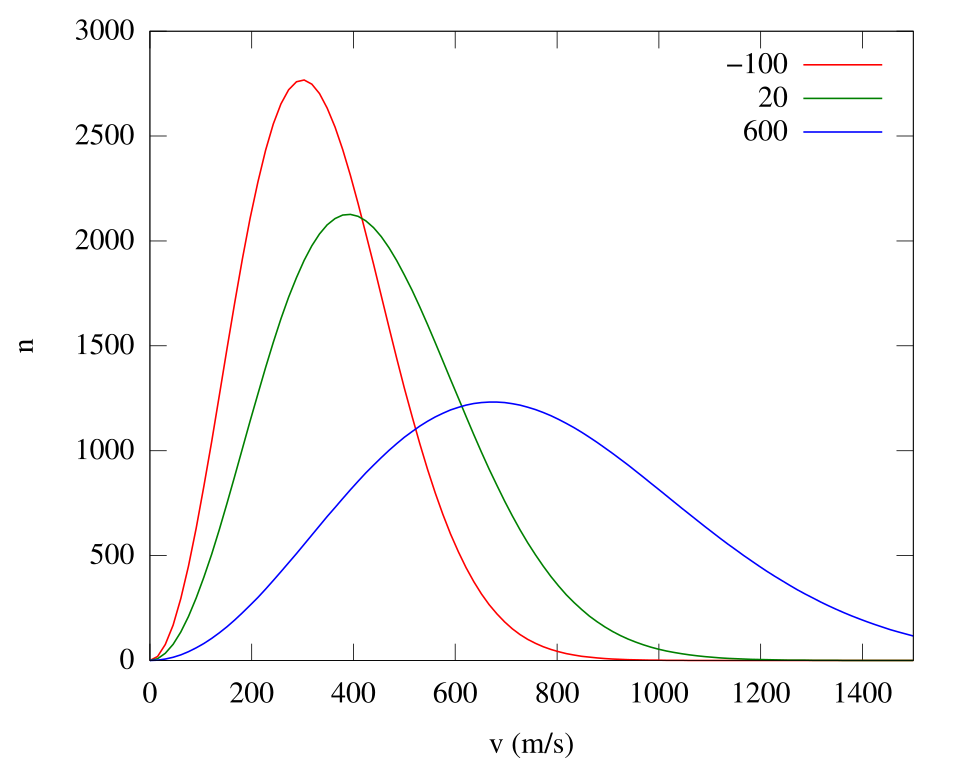
A Maxwell–Boltzmann speed distribution plot shows how increasing temperature shifts the curve to higher speeds and broadens the distribution. It provides a quantitative-style visual for the statement that higher temperature means higher average kinetic energy and faster particle motion, which helps particles overcome intermolecular attractions during phase changes. Source
Cooling decreases motion, allowing attractions to pull particles into closer, more ordered arrangements.
Stronger attractions make it harder to separate particles, favouring condensed phases at a given temperature.
Increased pressure forces gas particles closer together, making attractions more significant and favouring condensation qualitatively.
What to avoid (because it is “not assessed” here)
Because the specification explicitly excludes phase diagrams, you should avoid relying on:
Identifying or interpreting phase boundaries on a diagram
Discussing triple points or critical points as required knowledge
Using phase-diagram slopes or regions to justify what phase is present
Explaining unusual cases (for example, special melting-curve behaviour) via diagram features
If a phase diagram appears in a classroom resource, treat it as optional enrichment. For AP assessment aligned to this statement, your score-relevant skill is to explain phase properties and changes using particle arrangement, motion, and attractions, not by reading a diagram.
How to write an AP-quality explanation without phase diagrams
When prompted to explain why a substance is solid/liquid/gas under some conditions, build responses with:
A clear statement of the phase in terms of particle spacing and motion
A cause-and-effect link to temperature (energy and motion) and attractions
A macroscopic consequence (shape, volume, compressibility, ability to flow) traced back to the particles
Common high-utility phrases (used accurately) include:
“Particles remain in close contact” (solids and liquids)
“Particles move past one another” (liquids)
“Particles are far apart with negligible attractions” (idealised gases)
“Energy is required to overcome attractions” (changes toward gas)
“Reducing motion allows attractions to dominate” (changes toward solid/liquid)
FAQ
They can shift assessment towards diagram-reading conventions rather than particle-based causality.
Excluding them keeps the focus on explaining observable behaviour from spacing, motion, and attractions.
They are not intended as a tested interpretation skill here.
If any graphic is shown, questions would still be answerable from particle reasoning rather than identifying diagram features.
They describe only spacing and forget motion or attractions.
AP explanations usually require a causal chain: energy/temperature → motion → ability to overcome attractions → macroscopic property.
They should show multiple particles, not a single molecule.
Clarity matters more than artistry: correct spacing, some indication of motion, and consistent representation across phases.
Remove words like “region,” “boundary,” or “curve,” and replace them with particle causes.
A good check is whether your explanation would still work with no graph present at all.
Practice Questions
Question 1 (1–3 marks) State what the AP Chemistry specification indicates about phase diagrams in this topic, and identify what should be used instead to explain phase properties.
1 mark: States that understanding/interpreting phase diagrams is excluded from assessment.
1 mark: States that the focus should be on particulate models.
1 mark: Mentions that particulate models explain phase properties/behaviour via particle arrangement/motion/attractions.
Question 2 (4–6 marks) A student tries to justify that a sample will be a liquid by referring to “the region on a phase diagram.” Explain, without using phase diagrams, how you would justify whether a substance behaves as a solid, liquid, or gas under given conditions. Your answer should refer to particle spacing, motion, and attractions, and must link to at least two macroscopic properties.
1 mark: Describes particle spacing for at least two phases (close vs far apart).
1 mark: Describes particle motion differences (vibration; sliding/flow; free motion).
1 mark: Links attractions to phase stability (stronger attractions favour condensed phases / harder to separate).
1 mark: Correctly links to one macroscopic property (e.g., fixed shape, ability to flow, compressibility).
1 mark: Correctly links to a second macroscopic property (e.g., fixed volume vs variable volume, density trends).
1 mark: Explicitly avoids phase-diagram reasoning by framing the justification as particulate-model based.

