AP Syllabus focus: ‘Solids may be crystalline with a regular 3D arrangement or amorphous without orderly arrangement; in both, particle motion is limited and overall translation does not occur.’
Solids are defined by closely spaced particles and restricted motion, but not all solids are structurally identical. Understanding the difference between crystalline and amorphous solids helps explain melting behavior, mechanical properties, and experimental observations.
Crystalline vs Amorphous: Core Ideas
A solid’s classification depends on how its particles are arranged over large distances, not on what the particles are made of.
Crystalline solid: A solid whose particles (atoms, ions, or molecules) are arranged in a regular, repeating 3D pattern (a crystal lattice) extending throughout the sample.
Crystalline solids exhibit long-range order, meaning the repeating pattern continues over many particle spacings.

Side-by-side schematic contrasting a crystalline solid (periodic, repeating arrangement) with an amorphous solid (disordered network). This visual makes the idea of long-range order intuitive: the crystalline pattern repeats far beyond nearest neighbors, while the amorphous structure does not. Source
Amorphous solid: A solid that lacks an orderly, repeating arrangement of particles; it has no long-range pattern, even though particles remain close together.
In amorphous solids, there may be short-range order (local packing over a few neighbors), but no repeating 3D pattern across the bulk material.
Particle Motion in Both Types of Solids (Syllabus Emphasis)
Even though their structures differ, both crystalline and amorphous solids have limited particle motion, and overall translation does not occur:
Particles primarily vibrate about fixed positions
Particles do not continuously move past one another as in a liquid
The solid maintains a fixed shape and volume under ordinary conditions
Structural Consequences You Can Observe
The presence or absence of a repeating structure leads to different macroscopic behaviors that you can connect to particle-level models.
Melting and Softening Behavior
Crystalline solids typically have a sharp melting point because disrupting a uniform lattice occurs over a narrow temperature range.
Amorphous solids often soften over a range of temperatures because different local regions require different amounts of energy to begin flowing.
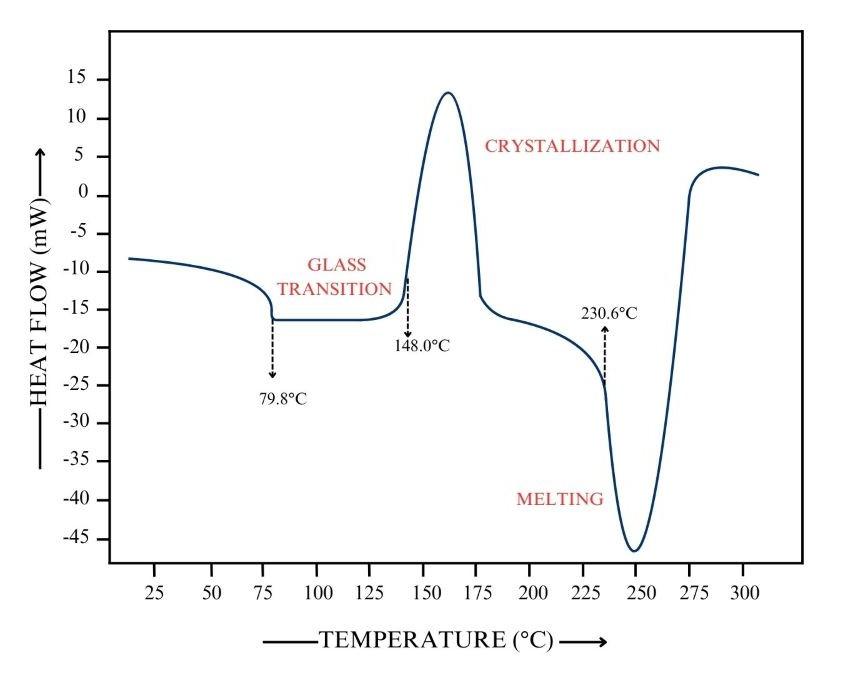
Differential scanning calorimetry (DSC) curve illustrating a glass transition as a baseline step and melting as a sharp endothermic peak. The plot helps connect structure to observable behavior: amorphous regions show a gradual transition ( range), whereas crystalline regions produce a distinct melting event (). Source
Shape, Surfaces, and Cleavage
Crystalline solids may form flat faces and can cleave along specific planes where the lattice breaks more easily.
Amorphous solids usually fracture irregularly because there are no repeating planes of weakness.
Direction-Dependent Properties (Anisotropy)
In a single crystal, properties (such as hardness or conductivity) can depend on direction because the lattice is oriented.
In an amorphous solid, properties are typically more uniform in all directions because there is no consistent long-range orientation.
How to Identify Them Conceptually (No Special Tools Required)
A helpful mental test is to imagine zooming out from a particle diagram:
If the pattern repeats predictably as you move outward in many directions, it is crystalline.
If the arrangement is random-looking beyond the nearest neighbors, it is amorphous.
Common Examples (For Recognition)
Crystalline: many salts and minerals (ordered lattices)
Amorphous: glassy solids and many plastics (disordered networks)
FAQ
Glass is amorphous, so different regions have slightly different local structures. On heating, parts of the structure gain mobility at different temperatures, producing a gradual softening (often described using a glass transition rather than a true melting point).
Crystalline solids give sharp, well-defined diffraction peaks due to regular lattice spacing.
Amorphous solids give broad humps because there is no long-range periodic spacing to reinforce specific diffraction angles.
Yes. Given sufficient molecular mobility (often aided by heating below melting), some amorphous materials can crystallise. This process is influenced by nucleation sites, time, and thermal history (cooling rate and annealing).
$T_g$ is the temperature range where an amorphous solid changes from brittle/glassy to rubbery as molecular segments gain mobility. It is not a lattice-breaking event, so it differs from a crystalline melting point.
Amorphous solids can dissolve faster than crystalline forms because their particles are less tightly organised. Faster dissolution can improve bioavailability, but amorphous drugs may be less stable and more likely to crystallise during storage.
Practice Questions
State one structural difference between a crystalline solid and an amorphous solid, and state one similarity in particle motion for both types.
1 mark: Crystalline has a regular repeating 3D arrangement / amorphous lacks long-range order.
1 mark: In both, particle motion is limited to vibrations about fixed positions and there is no overall translation.
A student heats two solids, A and B. Solid A melts sharply at one temperature. Solid B gradually softens over a temperature range.
(a) Deduce which solid is more likely crystalline and which is more likely amorphous. (2 marks)
(b) Explain, in terms of particle arrangement and motion, why their heating behaviour differs. (3 marks)
(a)
1 mark: A is crystalline.
1 mark: B is amorphous.
(b)
1 mark: Crystalline solids have a regular repeating lattice (long-range order).
1 mark: Amorphous solids lack long-range order / have a range of local environments.
1 mark: Both have limited motion (vibration) in the solid state, but disrupting a uniform lattice occurs at one temperature whereas disordered regions begin to move at different temperatures.

