AP Syllabus focus: ‘In liquids, particles remain in close contact while moving and colliding; arrangement and movement depend on the nature and strength of forces such as polarity, hydrogen bonding, and temperature.’
Liquids occupy a middle ground between solids and gases: particles stay close together, yet they can move past one another. Their macroscopic behaviour is best explained by particle motion competing with intermolecular attractions.
Particle-level model of liquids
Close contact and short-range order
Liquid particles are in close contact, meaning neighbouring particles are separated by distances comparable to their sizes. Unlike gases, there is very little empty space between particles, so liquids are nearly incompressible.
Although liquids lack the repeating 3D pattern of a crystal, they often exhibit short-range order: over a few particle diameters, attractions and particle shape create transient local arrangements that continually form and break.
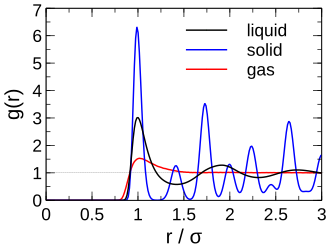
Radial distribution functions for solid, liquid, and gaseous argon compare how particle density varies with distance from a reference particle. The liquid curve shows a strong first-neighbor peak (and weaker subsequent peaks), capturing short-range order without the long-range periodicity of a crystal. This connects a particle-level structure metric to the macroscopic idea that liquids are dense yet disordered beyond a few molecular diameters. Source
Constant motion, collisions, and flow
Particles in a liquid are in continuous, random motion. They frequently collide and exert attractive forces on nearby particles. The key difference from a solid is that particles can translate (move position) by slipping past neighbours, so the substance can flow and take the shape of its container.
Viscosity: Resistance of a liquid to flow, arising from intermolecular attractions and how easily particles can slide past one another.
A useful way to think about a liquid is as a crowded environment: particles are always interacting, so their paths are constantly redirected by collisions and attractions.
Forces in liquids: what controls arrangement and movement?
Intermolecular attractions compete with particle motion
Liquid structure and dynamics depend on a balance between:
Intermolecular forces (IMFs) that pull particles together and favour more organised local arrangements
Thermal motion that disperses particles and disrupts local ordering
Stronger attractions generally lead to less freedom of movement (slower rearrangement), while higher temperature increases particle motion and makes it easier for particles to overcome attractions during collisions.
Polarity and hydrogen bonding in liquids
Many liquids are molecular substances where the dominant attractions depend on molecular structure:
Polar molecules experience dipole–dipole attractions, which favour particular orientations (partial positive ends near partial negative ends).
Molecules capable of hydrogen bonding (involving H bonded to N, O, or F and attracted to a lone pair on N, O, or F) often form transient, directional interaction networks that strongly affect mobility and local arrangement.
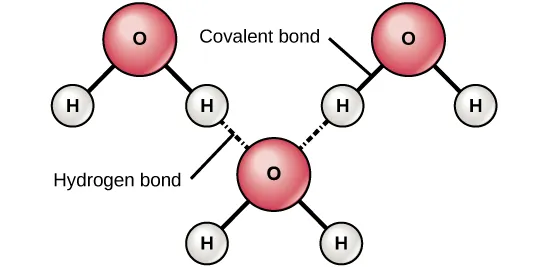
The figure depicts hydrogen bonding between water molecules, distinguishing intramolecular covalent bonds from the intermolecular hydrogen bond (shown as a dotted interaction). Because hydrogen bonds are directional, they favor specific relative orientations between molecules and can form transient networks in the liquid. These networks are a key reason hydrogen-bonding liquids often have higher viscosity and surface tension than similar-sized non–hydrogen-bonding liquids. Source
Even nonpolar liquids have attractive forces between particles, but these are typically less directional, allowing easier sliding and reorientation.
Because particles remain close together, these attractions operate essentially all the time; the liquid’s properties reflect an ongoing tug-of-war between “stick together” and “move around.”
Surface tension: The tendency of a liquid surface to minimise area because surface particles experience a net inward attraction.
Surface effects matter because particles at the surface have fewer neighbours, so the balance of forces differs from the interior.
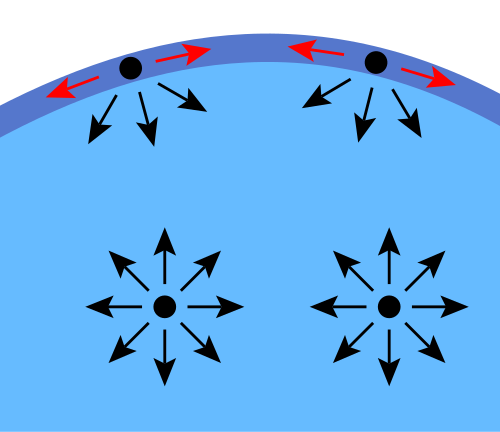
This schematic shows that molecules in the interior of a liquid experience attractions from all directions that largely cancel, whereas molecules at the surface have fewer neighbors above them. The resulting imbalance produces a net inward pull at the surface, which helps explain why liquids minimize surface area. The same particle-level argument underlies surface tension and many surface phenomena (droplets, capillarity, and floating objects). Source
Temperature effects within the liquid state
How increasing temperature changes behaviour
Temperature changes the kinetic energy distribution of particles. As temperature increases:
Particles move faster and collisions are more energetic.
Short-lived local arrangements are disrupted more frequently.
The liquid’s structure becomes, on average, less ordered over short distances.
As a result, liquids often show these trends with increasing temperature:
Lower viscosity (particles can slip past each other more easily).
Greater ability to escape the surface (more particles have enough energy to overcome attractions at the surface).
Evaporation as a surface process
Even below the boiling point, some particles at the surface can escape into the gas phase. This is easier when:
The liquid’s particle attractions are weaker (less energy needed to separate particles).
The temperature is higher (more particles have sufficient energy at a given moment).
At the particulate level, evaporation does not require all particles to “break free,” only those near the surface that momentarily have enough energy and the right trajectory.
Connecting microscopic forces to macroscopic observations
When comparing liquids, the key explanatory links are:
Stronger or more directional attractions (often associated with polarity and especially hydrogen bonding) tend to reduce particle mobility, increasing viscosity and strengthening surface tension.
Higher temperature increases motion, disrupting attraction-based ordering and increasing how rapidly particles rearrange during collisions.
FAQ
Attractions keep particles close, but they are not fixed in place. Constant collisions and thermal motion let particles exchange neighbours, so the liquid can deform continuously under stress rather than fracture.
High viscosity is promoted by:
Strong attractions (especially directional ones)
Molecular shapes that tangle or hinder sliding
Temporary interaction networks that must break and reform for flow to occur
Interior particles are surrounded on all sides and strongly held by neighbours. Surface particles have fewer neighbours, so they require less energy to escape; only a fraction have enough energy at any moment.
Hydrogen bonds form transient, local structures (short-range order) that constantly rearrange. The interactions are strong enough to influence orientation and mobility, but not permanent enough to lock particles into a repeating lattice.
The frequency of rearrangements increases: local clusters form and break more rapidly, orientation correlations persist for shorter times, and collisions more often disrupt attraction-favoured configurations, making the liquid dynamically less structured.
Practice Questions
(2 marks) Using a particle model, explain why liquids are difficult to compress.
Particles are in close contact / very little empty space between particles (1)
Compression would require forcing particles much closer, strongly opposed by repulsions/limited free volume (1)
(5 marks) Liquid A is polar and forms hydrogen bonds; liquid B is non-polar. Compare (i) the viscosity and (ii) the ease of particles escaping from the surface of the two liquids at the same temperature. Then describe how increasing temperature affects each comparison. Explain using intermolecular attractions and particle motion.
A has higher viscosity than B due to stronger/more directional attractions (hydrogen bonding) restricting flow (1)
Particles escape less easily from A’s surface than from B’s due to stronger attractions to neighbours (1)
Increasing temperature decreases viscosity for both by increasing particle motion and overcoming attractions more frequently (1)
Increasing temperature increases surface escape for both because more particles have sufficient energy to overcome attractions (1)
Clear linkage to “close contact + moving/colliding” with forces affecting arrangement/movement (1)

