AP Syllabus focus: ‘For a given substance, solid and liquid phases typically have similar molar volumes because particles in both phases remain in close contact at all times.’
Solids and liquids often seem very different, yet they occupy nearly the same volume per mole. This similarity comes from how particles are arranged: in both phases, particles touch or nearly touch, leaving little empty space.
Core idea: particles stay close in both phases
In both solids and liquids, particles are held near a preferred separation distance where attractions and repulsions balance.

Side-by-side particle-level views of a solid, liquid, and gas highlight that solids and liquids remain closely packed (condensed phases) while gases contain large gaps. The images emphasize that melting changes particle mobility and arrangement more than average spacing, which helps explain why solid and liquid molar volumes are often similar. Source
Changing from solid to liquid mostly changes mobility and arrangement, not average particle spacing.
In a solid, particles vibrate about fixed positions and maintain a stable, closely packed structure.
In a liquid, particles can move past one another, but they still remain in close contact because intermolecular attractions keep them from separating widely.
Because particles are already near their minimum-energy separation, melting does not suddenly create large gaps; it mainly increases disorder and allows flow.
Molar volume and why it changes little on melting
Molar volume — the volume occupied by 1 mole of a substance at specified conditions (units commonly or ).
A useful way to connect molar volume to measurable properties is through density.
= molar volume ( or )
= molar mass ()
= density ( or )
For a given substance, the molar mass is the same whether the sample is solid or liquid. Therefore, similar molar volumes imply that the densities of the solid and liquid are often similar. That happens because the particles remain packed closely in both phases, so the amount of empty space per mole stays relatively small.
Close contact: attraction, repulsion, and limited compressibility
At typical temperatures and pressures, solid and liquid particles sit at separations dominated by two competing effects:
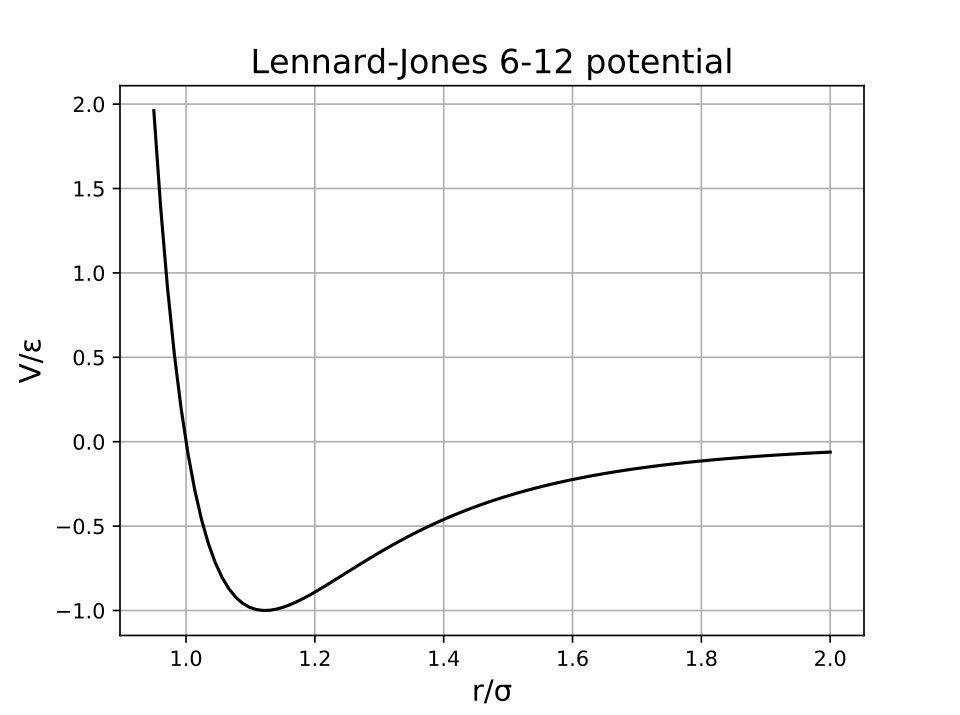
A Lennard–Jones potential graph shows how potential energy varies with intermolecular distance: strong repulsion at very short range and attraction at intermediate range. The minimum of the curve corresponds to the preferred separation where attractions and repulsions balance, helping explain both limited compressibility and why melting does not greatly change average spacing. Source
Attractive forces (overall intermolecular attractions) pull particles together.
Short-range repulsions (electron cloud overlap/Pauli repulsion) push back strongly when particles get too close.
This steep repulsion makes both solids and liquids hard to compress. Since particles are already “as close as they can reasonably get” without strong repulsion, melting does not significantly reduce particle spacing, and it also does not greatly increase spacing because attractions still keep particles nearby.
What actually changes from solid to liquid
When a substance melts, the key change is in structure and motion, not average separation distance.
Solids: more ordered particle positions; motion is mostly vibrational.
Liquids: less ordered positions; motion includes translation (particles slide and rearrange), but neighbours remain nearby.
This is why liquids can take the shape of their container while still having a definite volume similar to the solid’s volume: the particles are still essentially “touching” throughout the sample.
Contrast with gases (why gases are different)
A gas has a dramatically larger molar volume because its particles are separated by large distances, with substantial empty space. In solids and liquids, the “empty space” is limited to small gaps between closely packed particles, so the molar volume remains relatively small and similar between the two condensed phases.
FAQ
No. “Similar” means the change is usually small compared with the change on vaporisation.
A small difference can still be measurable and depends on how efficiently particles pack in each phase.
Typically by measuring mass and volume to obtain density, then converting using $V_m=M/\rho$.
For liquids, volume is read directly (e.g., volumetric glassware). For solids, volume may be found by geometric measurement or displacement.
Flow comes from particles being able to continually break and reform local arrangements.
Close contact is maintained, but particles can translate past neighbours rather than remaining in fixed positions.
There are small interparticle gaps and irregular voids due to imperfect packing and thermal motion.
These voids are tiny compared with the vast empty space between gas particles.
Not always. Attractions influence preferred spacing, but the dominant limit at very short distances is steep repulsion.
In condensed phases, changes in structure/packing can matter as much as attraction strength, so the net effect on molar volume can be modest.
Practice Questions
Explain why the molar volumes of a substance in the solid and liquid phases are typically similar.
States that in both solids and liquids particles remain in close contact/closely packed (1).
Links close contact to limited empty space and therefore similar volume per mole (1).
Mentions that melting mainly changes mobility/arrangement rather than average particle spacing (1).
A pure substance has a molar mass of . At the same pressure, its solid density is and its liquid density is near the melting point. Using particle-level reasoning, explain why these densities (and hence molar volumes) are similar, and what this indicates about particle spacing in the two phases.
Recognises that similar densities imply similar molar volumes for the same molar mass (1).
Explains that particles are in close contact in both phases, so there is little empty space per mole (1).
Describes that melting increases freedom of movement/disorder but particles remain neighbouring/close due to attractions (1).
Mentions that strong short-range repulsion prevents particles getting much closer, limiting compression in both phases (1).
Concludes that average particle spacing changes only slightly from solid to liquid (1).
Coherent linkage between macroscopic observation (density) and particulate model (packing/spacing) (1).

