AP Syllabus focus: ‘Processes with ΔG° < 0 favor products (K > 1), while processes with ΔG° > 0 favor reactants (K < 1).’
Thermodynamic signs quickly tell you whether equilibrium lies mostly to the product side or reactant side under standard conditions. This section focuses on interpreting ΔG° and K together, not on calculating them.
Product-favored vs reactant-favored: the meaning of the signs
Key thermodynamic quantities
Standard Gibbs free energy change (ΔG°): The free energy change for a reaction when all species are in their standard states, used to judge thermodynamic favorability under those conditions.
A common interpretation error is to treat “favored” as meaning “happens completely.”
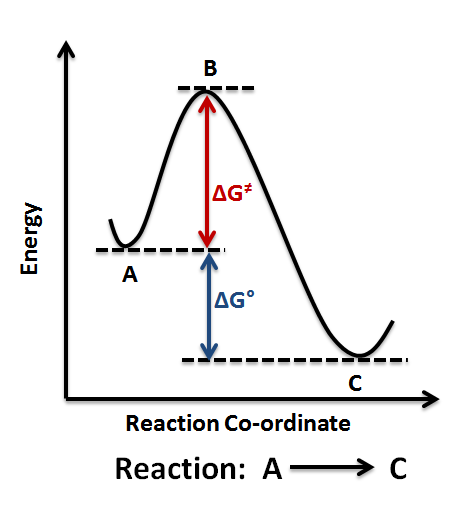
Reaction-coordinate (free-energy) diagram showing reactants converting to products via a transition state (the peak). The vertical gap between reactants and products represents the reaction’s free-energy change (), which is negative when products lie lower in free energy than reactants. The peak highlights that thermodynamic favorability does not imply a fast reaction (a barrier can still exist). Source
In AP Chemistry, favored means the equilibrium position is shifted toward one side, not that the reaction goes to completion.
Equilibrium constant (K): A dimensionless number that indicates the equilibrium composition; larger values mean equilibrium lies further toward products for the balanced reaction as written.
Interpreting the sign of ΔG°
Under standard conditions:
ΔG° < 0 means the reaction is thermodynamically favored in the forward direction.
Equilibrium tends to contain more products than reactants (relative to stoichiometry).
This corresponds to K > 1.
ΔG° > 0 means the forward reaction is thermodynamically unfavored.
Equilibrium tends to contain more reactants than products.
This corresponds to K < 1.
ΔG° = 0 is the boundary case.
The system is at the tipping point under standard conditions.
This corresponds to K = 1.
Interpreting the size of K (without calculating)
The value of K indicates “how far” equilibrium lies to one side:
K much greater than 1: strongly product-favored; products dominate at equilibrium.
K close to 1: neither side is strongly favored; significant amounts of both reactants and products.
K much less than 1: strongly reactant-favored; reactants dominate at equilibrium.
These statements are always tied to the balanced equation as written. If you reverse the reaction, the interpretation flips (product-favored becomes reactant-favored) because K becomes 1/K and ΔG° changes sign.
The ΔG°–K connection (how the signs are linked)
The sign relationship in the syllabus comes from the fundamental thermodynamic connection between free energy and equilibrium.
= standard Gibbs free energy change (J·mol)
= gas constant, J·mol·K
= absolute temperature (K)
= equilibrium constant (dimensionless)
Because and are positive, the sign logic is fixed:
If K > 1, then is positive, so ΔG° is negative.
If K < 1, then is negative, so ΔG° is positive.
What “favored” does (and does not) claim
“Product-favored” (ΔG° < 0, K > 1) means products are lower in free energy relative to reactants under standard conditions, so equilibrium lies to the product side.
It does not guarantee:
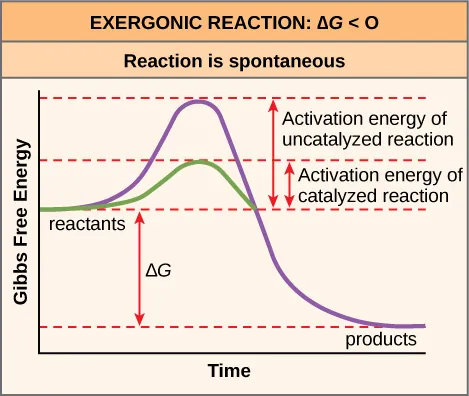
Energy profile comparing uncatalyzed vs catalyzed pathways for an exergonic reaction (). Both curves have the same reactant-to-product free-energy change, but the catalyzed pathway has a lower activation energy barrier. This reinforces that (thermodynamics) and reaction rate (kinetics) are different ideas. Source
a fast reaction rate,
a complete reaction (100% yield),
or that nonstandard starting conditions will behave the same way.
FAQ
Equilibrium means forward and reverse rates are equal, not that reactants vanish.
Even when $K>1$, the equilibrium mixture can contain reactants because the expression for $K$ allows nonzero reactant terms, especially if $K$ is only moderately above 1.
Strictly, $K$ is defined using activities, which are ratios relative to a standard state, so the units cancel.
In many AP problems, concentrations/pressures are substituted directly, but the intended interpretation is still unitless.
If you reverse a reaction, $K$ becomes $1/K$ and $\Delta G^\circ$ changes sign.
If you multiply coefficients by a factor $n$, the new equilibrium constant is $K^{n}$ and $\Delta G^\circ$ scales by $n$.
No. It means the forward direction is unfavoured under standard conditions.
The reaction can still proceed to some extent, and it can be driven by nonstandard conditions or by coupling, but those are separate ideas from the sign interpretation under standard states.
Both have $K>1$ (so $\Delta G^\circ<0$), but different magnitudes of $K$ indicate different equilibrium positions.
A much larger $K$ corresponds to a more negative $\Delta G^\circ$ and a stronger tendency for products to dominate at equilibrium.
Practice Questions
(2 marks) Under standard conditions, a reaction has kJ·mol. State whether the reaction is product-favoured or reactant-favoured, and whether is greater than 1 or less than 1.
Reactant-favoured (1)
(1)
(5 marks) Consider three reactions at the same temperature:
Reaction A:
Reaction B:
Reaction C: For each reaction, state (i) whether is positive, negative, or zero, and (ii) whether equilibrium is product-favoured, reactant-favoured, or neither. Justify each using the relationship between and .
A: negative (1); product-favoured (1) because / (1)
B: positive (1); reactant-favoured (1) because / (1)
C: (1); neither side favoured (1) because (1) (Max 5: award any 5 correct marking points)

