AP Syllabus focus: ‘Qualitatively estimate K: if ΔG° is near zero, K is close to 1; if ΔG° is much larger or smaller than RT, K differs greatly from 1.’
Thermodynamic data often give ΔG° values that can be used to judge whether K is near 1 or extremely large/small. This page focuses on qualitative size comparisons to RT.
Core idea: compare ΔG° to the thermal scale RT
What “relative to RT” means
When judging how far an equilibrium lies to products or reactants, the key is the size of ΔG° compared with RT, which sets a temperature-dependent “energy scale.”
Thermal energy scale, : the product of the gas constant () and absolute temperature (), representing the typical magnitude of thermal energy per mole that competes with ordering/energy constraints.
At a fixed temperature, is a constant, so the ratio tells you whether the free-energy driving force is weak, moderate, or strong.
The ΔG°–K link (why the comparison works)
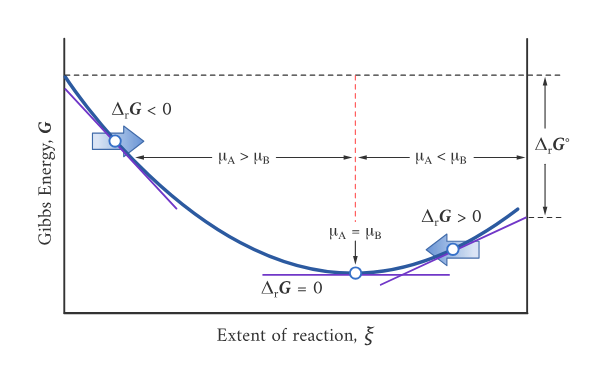
Gibbs energy plotted versus extent of reaction , illustrating that equilibrium occurs at the minimum of (where the slope is zero). Regions where drive the system toward products, while regions where drive it back toward reactants, reinforcing why equilibrium corresponds to a balance point in composition. Source
= standard Gibbs free energy change, J·mol
= equilibrium constant (dimensionless)
= gas constant, 8.314 J·mol·K
= temperature, K
Because the relationship uses , even “moderate” changes in relative to can push far from 1.
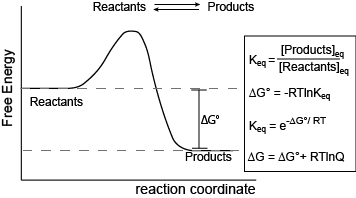
Reaction-coordinate free-energy diagram for a generic exergonic reversible reaction, annotated with the equilibrium relationship . The diagram connects the sign/magnitude of to the tendency to favor products vs. reactants at equilibrium, while highlighting that equilibrium is still a mixture rather than a “completion” statement. Source
Interpreting “near zero” vs “much larger than RT”
If ΔG° is near zero (|ΔG°| ≪ RT)
If , then , so . That implies:
Neither side is strongly favoured at equilibrium.
The equilibrium mixture contains appreciable amounts of both reactants and products (exact proportions depend on stoichiometry, but not “extreme”).
Small changes in conditions can noticeably shift composition because the driving force is weak.
If ΔG° is negative and large in magnitude (ΔG° ≪ −RT)
A strongly negative makes strongly positive, so . Qualitatively:
Products are strongly favoured at equilibrium.
The reaction is often described as “going essentially to completion” in an equilibrium sense (though not necessarily in rate).
Useful mental benchmarks (no calculation required):
on the order of → modestly greater than 1 (product-favoured, but not extreme).
Several times negative → becomes very large, meaning products dominate overwhelmingly.
If ΔG° is positive and large (ΔG° ≫ +RT)
A strongly positive makes strongly negative, so :
Reactants are strongly favoured at equilibrium.
Only a small fraction converts to products under standard conditions.
“Much larger or smaller than RT” (|ΔG°| ≫ RT)
This is the syllabus phrase that signals an extreme equilibrium:
If is many times , then is large.
Large corresponds to differing from 1 by orders of magnitude, not just a factor of 2 or 3.
Temperature dependence in this qualitative comparison
Because the comparison is to , temperature changes the meaning of “large”:
At higher , is larger, so a fixed looks less extreme in , pulling closer to 1.
At lower , is smaller, so the same looks more extreme, pushing farther from 1.
Common interpretation checkpoints (qualitative)
and magnitude small relative to → slightly above 1.
and magnitude large relative to → (products dominate).
and magnitude large relative to → (reactants dominate).
→ (no strong preference).
FAQ
Because $RT$ sets the scale in $\Delta G^\circ/RT = -\ln K$. If $|\Delta G^\circ| \ll RT$, then $|\ln K| \ll 1$, which forces $K$ to be near 1.
The logarithm makes $K$ exponential in $-\Delta G^\circ/RT$.
So linear shifts in $\Delta G^\circ$ translate into multiplicative changes in $K$.
It means $K$ is much larger than 10 (product-heavy) or much smaller than 0.1 (reactant-heavy), not merely 2 or 0.5. This corresponds to $|\ln K|$ being several units.
Logarithms require dimensionless inputs. In rigorous treatments, $K$ is built from activities relative to standard states so units cancel, leaving a pure number.
If $\Delta G^\circ$ is within experimental uncertainty of 0, you cannot confidently claim $K$ is far from 1. Large-magnitude $\Delta G^\circ$ (relative to $RT$) is more robust against uncertainty.
Practice Questions
(1–3 marks) Under standard conditions at a fixed temperature, a reaction has very close to . Estimate the size of and state what that implies about the equilibrium mixture.
States . (1)
Links to comparable amounts of reactants and products at equilibrium / no strong preference. (1)
Uses correct qualitative language (e.g., “neither side strongly favoured”). (1)
(4–6 marks) At the same temperature, Reaction A has and Reaction B has . Compare the relative sizes of and and justify your answer using . No numerical calculation is required.
Uses to relate sign of to whether is greater or less than 1. (1)
For Reaction A, concludes is large and positive, so . (2)
For Reaction B, concludes is large and negative, so . (2)
States that the magnitudes imply both equilibria are extreme (orders of magnitude from 1), in opposite directions. (1)

