AP Syllabus focus: ‘Free energy and equilibrium are related by K = e^(−ΔG°/RT) and ΔG° = −RT ln K.’
Thermodynamic driving force and equilibrium position are mathematically linked. This relationship lets you translate between standard Gibbs free energy change and the equilibrium constant, tying “how favorable” to “how product-favored” at equilibrium.
Key quantities and what they represent
Standard Gibbs free energy change, ΔG°
ΔG° refers to the free energy change for a reaction as written, using standard-state reference conditions for each species.
Standard Gibbs free energy change (ΔG°): The change in Gibbs free energy for a reaction under standard-state conditions, indicating the inherent thermodynamic tendency of reactants and products to form equilibrium.
ΔG° is an extensive property for the reaction as written, so changing stoichiometric coefficients changes ΔG° accordingly.
Equilibrium constant, K
K is the equilibrium constant expression for the balanced reaction, written in terms of activities (effective concentrations).
Equilibrium constant (K): A dimensionless ratio of product activities to reactant activities at equilibrium, each raised to their stoichiometric coefficients, for a specific balanced reaction at a given temperature.
Because K is tied to a particular balanced equation, reversing a reaction inverts K, and scaling coefficients changes K in a predictable way (consistent with the underlying mathematics).
The mathematical relationship between ΔG° and K
Thermodynamics shows that the standard free energy change is proportional to the natural logarithm of the equilibrium constant.
= standard Gibbs free energy change of reaction, J mol
= gas constant, J mol K
= absolute temperature, K
= equilibrium constant (dimensionless)
= base of the natural logarithm (unitless constant)
This equation encodes the sign connection without extra assumptions: since and are positive, the sign of ΔG° depends on .
If K > 1, then is positive, so ΔG° is negative.
If K = 1, then , so ΔG° = 0.
If K < 1, then is negative, so ΔG° is positive.
Why K must be unitless (and how it happens)
The natural logarithm function requires a dimensionless argument, so K cannot carry units. In rigorous thermodynamics, K is built from activities, not raw concentrations or pressures.
Key implications for writing K appropriately:
Pure solids and pure liquids have activity defined as 1, so they do not appear in K.
Solutes are treated using activity relative to a standard reference (often approximated using concentration divided by a standard concentration), which removes units.
Gases are treated using activity relative to a standard reference pressure (often approximated using partial pressure divided by a standard pressure), which removes units.
In AP Chemistry practice, you may see K written using concentrations (Kc) or partial pressures (Kp); conceptually, the ΔG° relationship is with the unitless, activity-based K.
Temperature’s role in the relationship
For a given reaction, temperature must be specified: both ΔG° and K depend on T, and the equation explicitly includes T.
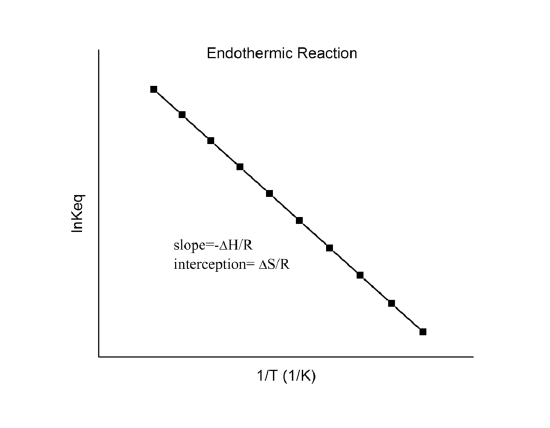
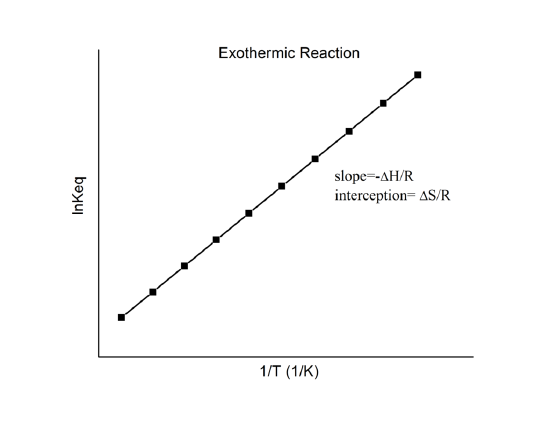
Van ’t Hoff plots show how the equilibrium constant changes with temperature by graphing versus . The straight-line form highlights that the slope is proportional to and the intercept is proportional to , so temperature shifts in equilibrium are tied directly to reaction enthalpy and entropy. Source
Therefore:
You cannot compare K values at different temperatures without accounting for the temperature change.
A reported ΔG° value implies a particular temperature (commonly 298 K unless stated otherwise).
FAQ
The thermodynamic derivation naturally produces the natural logarithm because it arises from integrating exponential (Boltzmann) relationships.
If you use $\log_{10}$, a conversion factor appears: $2.303$.
You can use $ \Delta G^\circ = -2.303,RT,\log_{10}K $.
This is equivalent to the $\ln$ form, just using a different logarithm base.
Scaling the reaction by a factor $n$ scales $\Delta G^\circ$ by $n$ and raises $K$ to a power: $K_{\text{new}} = K^{,n}$.
This keeps $ \Delta G^\circ = -RT\ln K $ consistent.
Reversing changes the sign of $\Delta G^\circ$ and inverts the equilibrium constant: $K_{\text{rev}} = 1/K$.
This matches the property $\ln(1/K) = -\ln K$.
With $R=8.314$ in J mol$^{-1}$ K$^{-1}$ and $T$ in K, $RT$ is in J mol$^{-1}$.
So $\Delta G^\circ$ comes out in joules per mole; convert to kJ mol$^{-1}$ only after the calculation.
Practice Questions
(2 marks) Using , state the sign of when (i) and (ii) . Justify each using the sign of .
(1) Correct sign for with justification that so .
(1) Correct sign for with justification that so .
(6 marks) Explain why the equilibrium constant in must be dimensionless, and describe how activities ensure this for (i) solutes, (ii) gases, and (iii) pure solids/liquids.
(1) States that requires a dimensionless argument, so must be unitless.
(2) Solutes: explains activity is defined relative to a standard state (units cancel; concentration used as an approximation to activity).
(2) Gases: explains activity is defined relative to a standard pressure (units cancel; partial pressure used as an approximation).
(1) Pure solids/liquids: states their activity is 1, so they are omitted from .

