AP Syllabus focus: ‘If a process is known to be thermodynamically favored yet does not occur measurably, it is reasonable to conclude it is kinetically controlled.’
Many chemical changes are “allowed” by thermodynamics but are rarely observed in real time. This page focuses on using observable evidence to justify when a system is kinetically controlled despite being thermodynamically favored.
What “reasoning from evidence” means here
You are expected to make an argument that connects two kinds of information:
Thermodynamic evidence that the process should proceed overall.
Experimental/observable evidence that the process does not proceed to a noticeable extent on the timescale of observation.
When both are true, the most defensible inference is a kinetic limitation (a large barrier to reaction progress).
The key inference: favored but not observed
A process can be thermodynamically favored yet show no meaningful change because the pathway is blocked by a barrier.
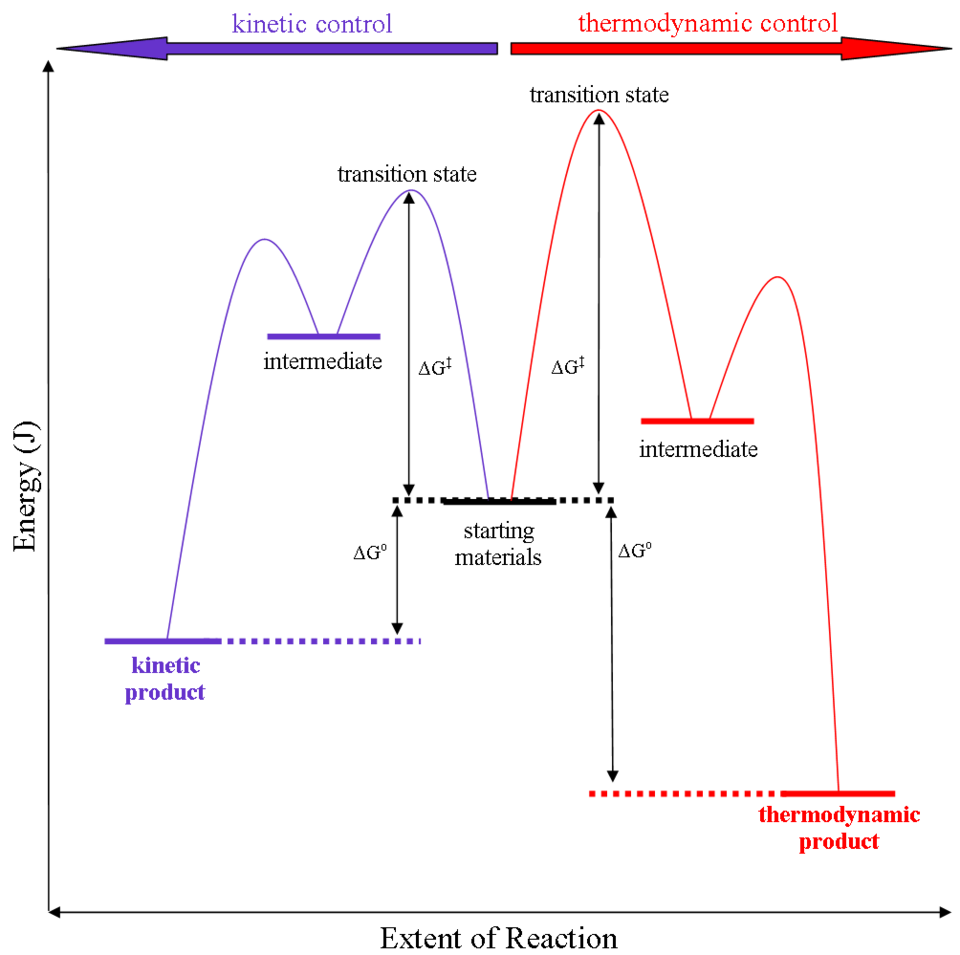
Generalized reaction-coordinate diagram comparing kinetic and thermodynamic products. The kinetic pathway has a smaller activation barrier (smaller ), so it can dominate at short times/low temperatures, while the thermodynamic product lies at lower free energy and is favored at equilibrium (given enough time to cross the barrier). Source
Your claim should match the syllabus statement: if it is known to be thermodynamically favored yet does not occur measurably, conclude kinetic control.
“Thermodynamically favored”: what counts as evidence
Thermodynamic favorability means products are lower in free energy than reactants under the stated conditions. Acceptable supporting evidence can include:
The process is described as spontaneous under those conditions.
Data/claims implying for the overall change (standard or nonstandard as stated).
The equilibrium position is strongly product-favored (for example, a very large equilibrium constant), even if equilibrium is not reached quickly.
You do not need to compute anything; you need to justify that, energetically, the change is “downhill.”
“Does not occur measurably”: what counts as evidence
“Not measurable” is about rate on the observation timescale, not about equilibrium. Evidence might be:
No visible change (no gas, precipitate, color change, mass change) over minutes/hours/days as specified.
Instrument readings (pressure, conductivity, concentration, voltage) remain essentially constant within stated uncertainty.
The change occurs only when a trigger is added (heating, spark, catalyst), suggesting the untriggered rate is negligible.
Be explicit about the timescale: “no change over 10 minutes” supports a kinetics claim; “no change ever” is usually too strong.
Kinetic control: the most reasonable explanation
Kinetic control — a situation where the observed extent of reaction is limited by a slow rate (often due to a large activation energy barrier), even if thermodynamics would favor product formation.
A strong AP-style justification links:
Favorability (thermodynamics) + negligible rate (observation) → high barrier → kinetic control.
What you are (and are not) allowed to conclude
From “favored but not happening,” you may reasonably conclude:
The system is not reaching equilibrium on the timescale observed.
The activation energy barrier is effectively preventing measurable progress under the given conditions.
You should not conclude (without extra evidence) that:
The reaction is impossible.
The reaction is at equilibrium.
The reaction has (that contradicts the premise).
Distinguishing kinetic control from “equilibrium” language
A common reasoning error is to treat “no observable change” as “equilibrium.” Kinetic control differs because:
At equilibrium, forward and reverse reactions occur at equal rates, and the equilibrium composition is established.
Under kinetic control, the system may still be far from equilibrium; it simply cannot get there quickly.
If the prompt indicates a strongly product-favored equilibrium but you observe mostly reactants persisting, that mismatch is classic evidence for kinetic inhibition.
Common experimental patterns that support kinetic control
Use these patterns as evidence chains (not as calculations):
Trigger required: Reaction proceeds only after heating/spark/light/catalyst is introduced → barrier-limited at baseline conditions.
Temperature sensitivity: Reaction becomes noticeable only at higher temperature → consistent with overcoming a barrier.
Metastable persistence: A higher-energy form remains (e.g., a material or arrangement that “should” convert) → conversion is kinetically blocked.
Keep the claim tightly scoped: “kinetically controlled under these conditions and timescale.”
FAQ
A catalyst lowers the activation barrier but does not change $\Delta G$ between reactants and products.
If adding a catalyst makes a previously “stuck” but thermodynamically favoured process proceed rapidly, that pattern strongly supports kinetic inhibition as the original cause.
It depends on what the prompt gives (seconds, minutes, hours, days) and the method (visible change vs instrument).
Your reasoning should explicitly reference the stated timeframe and detection method (e.g., “no pressure change within instrument uncertainty over 10 minutes”).
Yes. “Kinetically controlled” can still allow an extremely slow reaction.
The key is that the rate is too small to produce a meaningful or easily detectable change on the timescale of interest.
Look for confirmation that reactants are present and able to interact (mixing, phase contact, correct concentrations).
If composition is verified yet change remains negligible while thermodynamics predicts favourability, kinetic control becomes the stronger inference.
Surface passivation can physically block reaction even when thermodynamics favours it.
In that case, the underlying reasoning is still kinetic (rate-limited), but the mechanistic cause is a barrier created by the surface layer rather than an intrinsic bond-breaking barrier in solution.
Practice Questions
(2 marks) A reaction is reported to have at , but when the reactants are mixed at no observable change occurs over 30 minutes. What is the most reasonable conclusion, and what evidence supports it?
1 mark: Concludes the process is under kinetic control / kinetically inhibited.
1 mark: Cites both pieces of evidence (thermodynamically favoured: ; and no measurable change on the stated timescale).
(5 marks) A student claims “because nothing happened, the reaction must be at equilibrium.” You are told the reaction is thermodynamically favoured under the conditions used. Evaluate the student’s claim using evidence-based reasoning, and state what conclusion is justified instead. Include two distinct reasons.
1 mark: States the claim is not justified (no change does not imply equilibrium).
1 mark: Uses the given thermodynamic favourability as evidence the equilibrium position favours products.
1 mark: Explains equilibrium requires the system has reached equilibrium composition (not merely no visible change).
1 mark: States the justified conclusion: kinetic control / large activation barrier causes negligible rate.
1 mark: Provides a second supporting reason tied to observation timescale/measurement limits (e.g., “not measurable over the timeframe” indicates slow kinetics, not equilibrium).

