AP Syllabus focus: ‘A favored process that does not proceed measurably is under kinetic control; a high activation energy is a common cause.’
Thermodynamics tells whether a reaction is energetically allowed, but kinetics tells whether it happens on a useful timescale. Kinetic control explains why some favorable processes appear not to occur because of large activation energy barriers.
Kinetic Control: What It Means Mechanistically
A reaction can be thermodynamically favored and still be essentially unobservable in the lab if its rate is extremely small under the given conditions. In that case, the system is said to be under kinetic control: the outcome is governed by how difficult it is to cross the energy barrier, not by how stable the products are.
Kinetic control: A situation in which a process’s observable behavior is determined primarily by reaction rate (barrier height), so a thermodynamically favored change may not occur measurably.
Kinetic control is especially important when:
The activation barrier is large, so only a tiny fraction of collisions lead to reaction.
Conditions (temperature, catalyst presence, surface area, mixing) are not sufficient to overcome the barrier.
A system is “stuck” in a metastable state (not the lowest-energy state, but long-lived).
Activation Energy Barriers and the Transition State
Reaction rate depends strongly on activation energy, the energy required to reach the transition state from reactants. Even if products are lower in energy than reactants, the system must still pass through a high-energy configuration.
Activation energy (): The minimum energy barrier that must be overcome for reactants to form products via the transition state.
A large creates kinetic control because:
At a given temperature, relatively few particles have enough kinetic energy to reach the transition state.
Raising temperature increases the fraction of particles energetic enough to react, often dramatically.
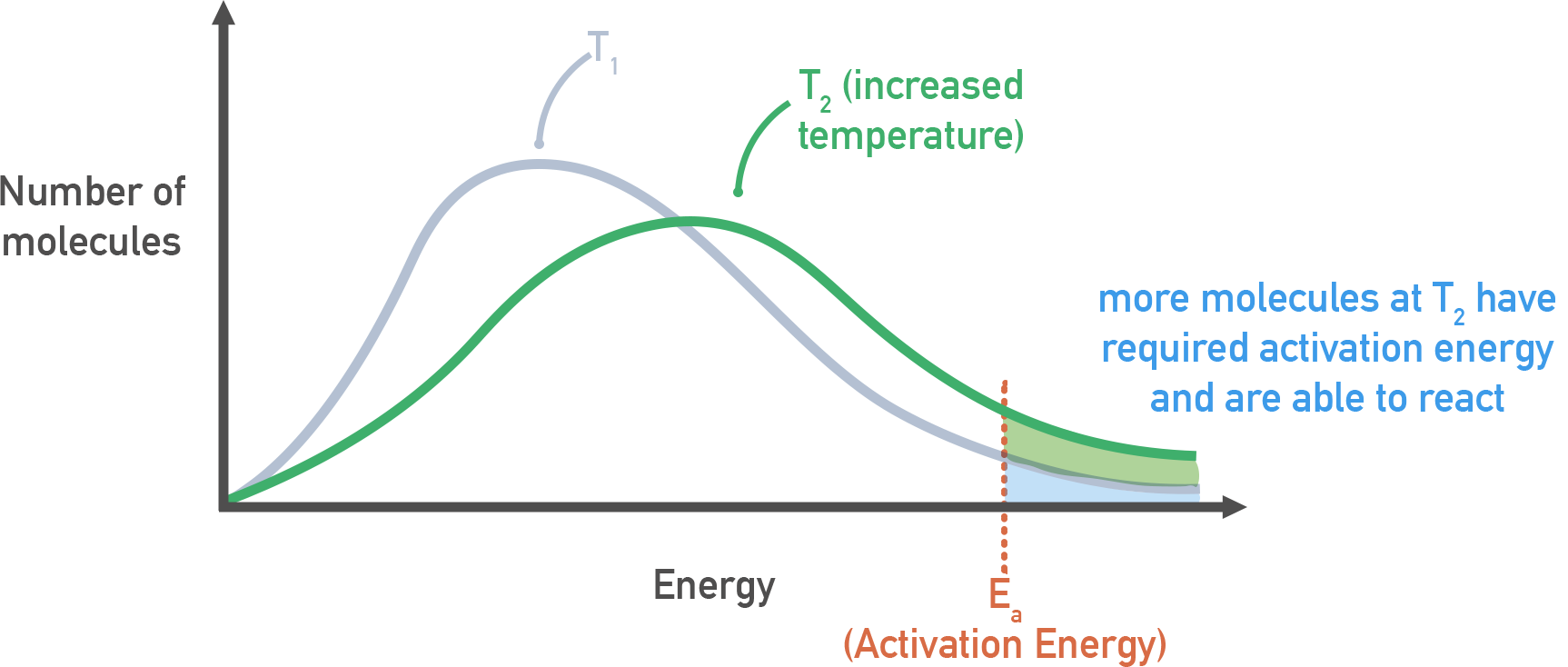
Maxwell–Boltzmann energy distributions at two temperatures ( and higher ) with the activation energy threshold, , marked. The shaded region beyond increases at higher temperature, illustrating why heating can make a previously unmeasurably slow reaction proceed at an observable rate. Source
Between understanding “high barrier” and predicting rate behavior is the quantitative idea that rate constants depend exponentially on .
= rate constant (units depend on overall reaction order)
= activation energy (J mol)
= gas constant (8.314 J mol K)
= temperature (K)
= frequency/orientation factor (same units as )
This relationship explains why modest temperature increases can turn an “imperceptibly slow” reaction into a measurable one, while a high can keep a favorable process effectively halted.
Recognising Kinetic Control in Practice
Kinetic control is inferred when a process is known (from energy considerations) to be favorable, yet it does not proceed measurably on the observation timescale. In AP Chemistry terms, the key reasoning is that a high activation energy is a common cause of the lack of observable change.
Common indicators consistent with kinetic control include:
No visible reaction (no gas, precipitate, color change) despite favorable driving forces.
Reaction proceeds only when heated, initiated (spark), or provided with a catalyst.
A reaction occurs rapidly once started, suggesting initiation required crossing a high barrier.
How Catalysts Relate to Kinetic Control
A catalyst changes the pathway to one with a lower effective , increasing rate without changing reactant/product energies. This directly addresses kinetic control: if the barrier is the problem, lowering the barrier can make the same favorable reaction occur at an observable rate.
Key catalyst ideas aligned to kinetic control:
Catalysts provide an alternative mechanism with a lower barrier.
Catalysts are not consumed overall; they improve rate by increasing successful transition-state formation.
Removing a catalyst can return a system to appearing “stuck,” even if the overall change remains favorable.
Timescale, Conditions, and “Not Measurably”
“Does not proceed measurably” is about experimental timescale. A kinetically controlled reaction may occur, but so slowly that:
Product formation is below detection limits, or
The time required is impractically long at the stated conditions.
Therefore, kinetic control is always conditional on the environment:
Temperature (most influential), since it alters the fraction of particles able to overcome .
Physical state and contact (e.g., solids reacting at surfaces can be limited by available reactive area).
Initiation requirements (some mechanisms require an initial high-energy step before rapid propagation can occur).
FAQ
If $E_a$ is very large, the rate constant is tiny at that temperature, so the timescale for detectable product is far longer than the experiment.
No. $E_a$ affects how quickly equilibrium is approached, not where equilibrium lies.
Initiation energy is a practical threshold to start a mechanism (e.g., forming radicals). Activation energy is the barrier for a specific elementary step.
Only surface particles react. Low surface area or passivating layers can limit collisions, making an otherwise favourable process appear not to proceed.
No. A catalyst changes the pathway and lowers $E_a$, but it does not change the overall thermodynamic favourability.
Practice Questions
Q1 (1–3 marks) Explain why a thermodynamically favourable reaction might not occur at a noticeable rate at room temperature.
States reaction is under kinetic control / rate-limited (1)
Identifies high activation energy barrier as the cause (1)
Links high to too few particles having sufficient energy at that temperature (1)
Q2 (4–6 marks) A reaction is known to be thermodynamically favourable, yet no change is observed over several hours. Using kinetic ideas, explain this observation and describe two ways experimental conditions could be changed to make reaction progress observable.
Concludes the process is kinetically controlled (1)
Explains lack of observable change due to high / difficulty reaching transition state (1)
Links temperature to increased fraction of particles with (1)
Suggests increasing temperature (1)
Suggests adding a catalyst to lower via alternative pathway (1)
Mentions an additional valid rate-increasing change (e.g., increased surface area/mixing for heterogeneous systems) (1)

