AP Syllabus focus: ‘A reaction not occurring at a noticeable rate does not mean the system is at equilibrium; it may simply be kinetically inhibited.’
Slow chemistry can be misleading: a system that barely changes may be “stuck” behind an energy barrier rather than balanced at equilibrium. This page clarifies what reaction rate can—and cannot—tell you.
Slow rate is not evidence of equilibrium
A mixture that shows little visible change (no colour shift, gas, precipitate, or temperature change) is often assumed to be “done.” In AP Chemistry, that assumption is unsafe because rate and equilibrium position describe different ideas.
Kinetics addresses how fast the system moves toward products (or back toward reactants).
Equilibrium addresses the composition the system tends toward if it has time and a viable pathway.
A key implication of the syllabus statement is that “no noticeable reaction” can mean the reaction is possible but occurring too slowly to observe on the available time scale.
What equilibrium actually means (vs “no reaction”)
At equilibrium, microscopic changes continue even if macroscopic properties remain constant.
Equilibrium (dynamic): A state in which the forward and reverse processes occur at equal rates, so concentrations (or partial pressures) remain constant over time.
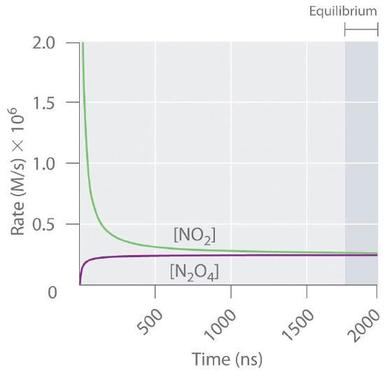
Forward and reverse reaction rates for a reversible system approach the same value as the system reaches dynamic equilibrium. The key idea is that macroscopic constancy corresponds to equal opposing rates, not to the reactions “stopping.” Source
A system can fail to change measurably for a completely different reason: the forward process may be extremely slow (and the reverse process equally irrelevant on the time scale), so the system is not necessarily at equilibrium.
Observable constancy has two possible explanations
Dynamic equilibrium: two opposing processes are occurring at equal rates.
Kinetic inhibition: the process is thermodynamically allowed but the rate is so low that change is not detected.
Kinetic inhibition: why “nothing happens”
Many reactions and phase/structural changes require surpassing a barrier before substantial progress occurs.
If typical particle collisions do not supply enough energy (or proper orientation), the process can be negligibly slow.
Common causes of kinetic inhibition that can mimic equilibrium-like behaviour:
High activation energy barrier (few successful collisions per unit time).
Low temperature, reducing collision energy and frequency.
Physical separation of reactants (limited contact area; poor mixing).
Protective layers (oxide coatings; passivation) blocking reactive surfaces.
Unfavourable molecular orientation requirements, making successful collisions rare.
Multi-step mechanisms with one very slow step controlling the overall rate.
Importantly, these factors affect how quickly a system approaches whatever state is ultimately favoured; they do not, by themselves, prove the system is at equilibrium.
What slow rates do imply
A slow or unobservable rate can justify conclusions about kinetics, not equilibrium.
The reaction may have a large activation energy or an otherwise slow mechanism.
The reaction may require specific conditions (temperature, catalyst, surface area, agitation) to proceed at a measurable rate.
Measurements taken over a short window may not reflect the system’s long-term behaviour.
What slow rates do not imply
A slow rate does not justify equilibrium claims without additional evidence.
It does not mean the forward and reverse rates are equal.
It does not mean concentrations have reached their equilibrium values.
It does not mean the system is “product-favoured” or “reactant-favoured.”
It does not rule out substantial change if conditions are adjusted to reduce kinetic barriers.
How to think like an AP Chemist about “no noticeable reaction”
When you see minimal change, separate the questions:
1) Is the system at equilibrium?
To support equilibrium, you need evidence of a stable composition that persists and is consistent with a dynamic balance (not merely “no visible change”).
2) Or is it kinetically inhibited?
To support kinetic inhibition, look for signs that changing conditions affects the rate dramatically:
Increasing temperature causes the process to begin or speed up.
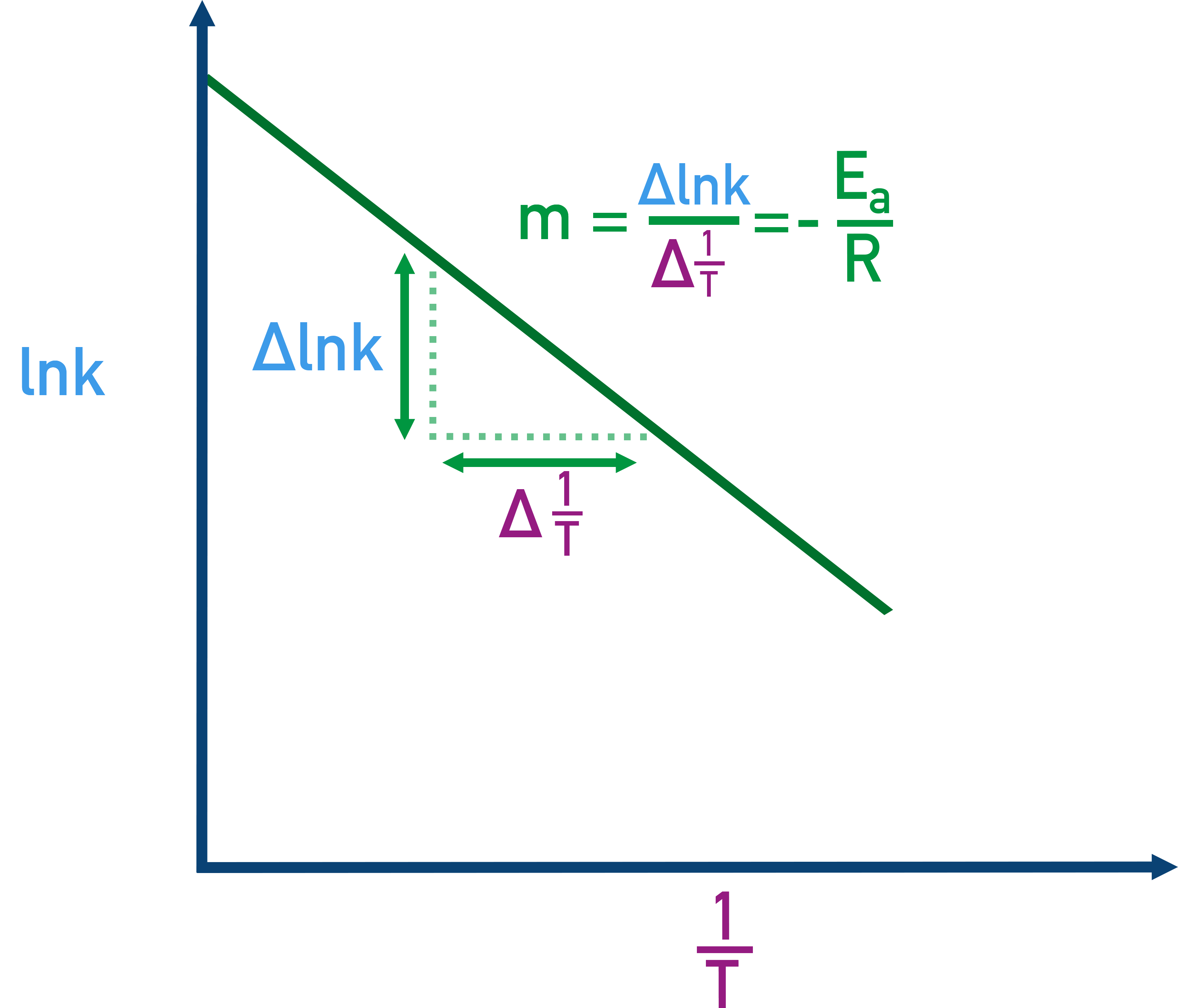
An Arrhenius plot of versus with the slope identified as . The negative slope shows that higher temperature (smaller ) corresponds to larger , explaining why heating often “turns on” a kinetically inhibited reaction. Source
Adding a catalyst increases rate without being consumed.
Increasing surface area (powdering a solid) increases rate.
Stirring or better mixing increases rate by improving contact.
These observations indicate a kinetic limitation was suppressing the rate, so the earlier lack of change was not strong evidence for equilibrium.
FAQ
At equilibrium, individual molecules continue reacting in both directions.
Because forward and reverse rates are equal, the net change is zero, so macroscopic properties stay constant even though reactions continue.
Visual cues alone (no bubbles, no precipitate, no colour change) are unreliable.
Some reactions have subtle changes, and some are too slow to observe without instruments or long time scales.
A catalyst lowers the activation barrier, increasing rate.
If adding a catalyst “starts” an apparently stalled process, that supports kinetic inhibition; it still does not, by itself, establish that the final state is equilibrium.
Yes. If both forward and reverse pathways have large barriers, the system can remain trapped in a long-lived non-equilibrium state.
Such states are sometimes called metastable and can persist until conditions change.
Track composition (concentration/partial pressure) over time under fixed conditions.
If composition becomes constant reproducibly and remains so even with small perturbations, that supports equilibrium; if it changes when barriers are reduced (e.g., heating), that supports kinetic trapping.
Practice Questions
(2 marks) Explain why a reaction that shows no noticeable change over several minutes is not necessarily at equilibrium.
States that equilibrium requires forward and reverse reactions occurring at equal rates / dynamic balance (1)
States that the reaction may be kinetically inhibited (very slow due to a barrier such as high activation energy), so macroscopic change is not observed (1)
(5 marks) A student mixes two reactants and observes no visible change. They claim the system has reached equilibrium. Evaluate this claim using kinetic vs equilibrium reasoning, and describe two experimental changes that would help distinguish equilibrium from kinetic inhibition.
Explains that lack of visible change alone does not prove equilibrium (1)
Correctly describes equilibrium as dynamic with equal forward and reverse rates and constant concentrations (1)
Identifies kinetic inhibition as an alternative explanation (slow rate; barrier) (1)
Proposes two valid changes (any two): increase temperature, add catalyst, increase surface area, stir/agitate, improve mixing/contact; each linked to increasing rate (2)

