AP Syllabus focus: ‘AP Exam questions will not assess derivation of Henderson–Hasselbalch or computing ΔpH from adding acid/base to a buffer, but using the equation to find pH is in scope.’
This page clarifies exactly what you are expected to do with the Henderson–Hasselbalch equation on the AP Chemistry Exam, and what common buffer-related tasks are explicitly not assessed.
What the AP Exam Will and Will Not Test
In scope: using Henderson–Hasselbalch to find pH
You are responsible for applying the Henderson–Hasselbalch relationship as a tool to determine buffer pH when given the needed chemical information (typically pK and a conjugate pair ratio).
You may be expected to:
Recognise when a solution contains a conjugate acid–base pair (e.g., HA/A⁻).
Identify which species is the acid form (HA) and which is the base form (A⁻).
Use provided concentrations (or other directly convertible quantities) to form the ratio .
Use logarithms correctly (including interpreting as base 10).
Out of scope: derivation and buffer “addition” pH-change calculations
The AP Chemistry Exam will not require algebraic derivations or multi-step buffer change computations tied to adding reagents.
You will not be assessed on:
Deriving the Henderson–Hasselbalch equation from equilibrium expressions (e.g., starting from and solving for pH).
Computing pH (or a new pH) after adding a strong acid or strong base to an existing buffer via stoichiometry-and-then-Henderson–Hasselbalch workflows.
Essential Tool You Can Use (Without Deriving It)
Henderson–Hasselbalch equation: A logarithmic relationship that connects buffer pH to the acid’s pK and the ratio of conjugate base to conjugate acid, .
On the exam, treat Henderson–Hasselbalch as a provided/accepted model: your job is correct substitution and interpretation, not proof.
= measure of acidity (unitless)
= for the weak acid HA (unitless)
= equilibrium concentration of conjugate base (mol·L)
= equilibrium concentration of weak acid (mol·L)
Because the AP Exam focuses on application, you should be comfortable distinguishing A⁻ from HA in chemical context (for example, from a chemical formula, a reaction, or a buffer description).
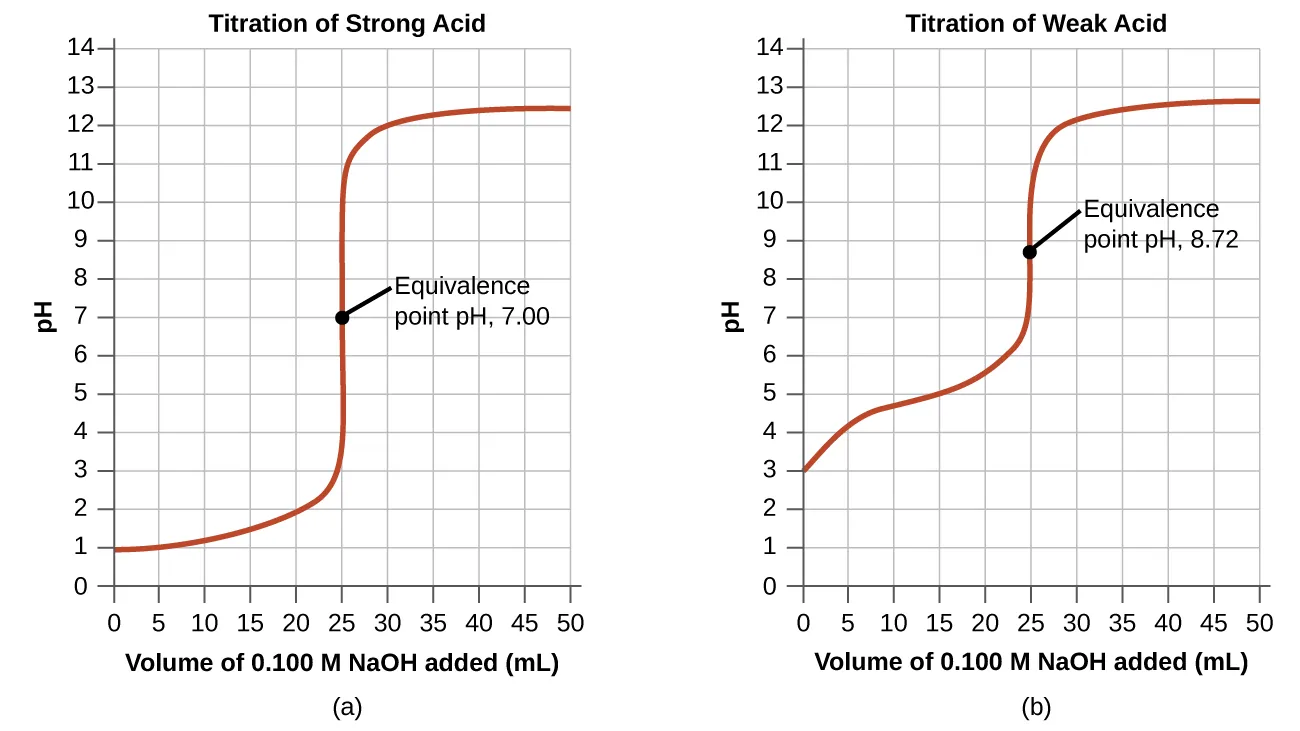
These titration curves compare a strong acid–strong base titration to a weak acid–strong base titration, highlighting how weak-acid systems exhibit a broad, gently sloped region before the equivalence point. That flatter region corresponds to conditions where both and are present in appreciable amounts, so buffer behavior—and Henderson–Hasselbalch reasoning—applies most naturally. Source
How to Recognise Exam-Friendly Henderson–Hasselbalch Setups
What “using the equation to find pH” typically looks like
Prompts that align with the assessed scope often provide:
A named weak acid (so pK is given directly, or is given so you can compute pK).
A buffer composition that clearly indicates both members of a conjugate pair are present.
Numerical information that directly yields the ratio without requiring modelling how that ratio changes due to adding external acid/base.
Common student pitfalls the scope implies you should avoid
Even when a question is in scope, errors often come from setup rather than mathematics:
Flipping the ratio: using instead of changes the sign of the log term.
Mixing up which pK applies: the pK must correspond to the HA ⇌ H⁺ + A⁻ pair used in the ratio.
Using species that are not a conjugate pair (e.g., combining two unrelated weak acids in one ratio).
Treating strong acids/bases as if they belong in Henderson–Hasselbalch (the equation is meant for weak acid/conjugate base systems).
Interpreting What “Not Assessed” Means in Practice
What you can assume about buffer-change scenarios
If a prompt involves adding acid/base to a buffer and would normally require a stoichiometric update of buffer components before applying Henderson–Hasselbalch, that calculation pathway is not the target skill in this course scope. If such contexts appear, the question is typically reframed so that you are still only “using the equation to find pH” (for instance, by giving the final ratio directly).
What you should still be able to communicate
Even without doing pH calculations, you should be able to:
State what information Henderson–Hasselbalch requires (pK and a conjugate ratio).
Explain qualitatively that pH depends on the log of the ratio, so changes in the ratio affect pH less dramatically than linear changes would.
FAQ
Yes, conceptually. It helps you remember which species belong in the ratio and why $pK_a$ matches the HA/A⁻ pair.
A full algebraic derivation is not required.
Often yes, provided both species are in the same solution volume so the volume cancels.
If volumes differ or change, you must be careful about cancellation.
Either can be provided. If $K_a$ is given, you may be expected to compute $pK_a=-\log(K_a)$ before using Henderson–Hasselbalch.
It can break down for very dilute buffers or when one component is extremely small, because equilibrium assumptions become weaker.
AP questions generally avoid these edge cases or guide you with appropriate data.
No. The equation uses base-10 logarithms.
If a calculator offers both, choose $\log$, not $\ln$.
Practice Questions
(2 marks) Which of the following is within AP Chemistry Exam scope regarding Henderson–Hasselbalch? A. Deriving the equation from the definition of B. Calculating pH after adding mol HCl to a buffer C. Using to find buffer pH D. Proving mathematically when Henderson–Hasselbalch fails
1 mark: Identifies option C
1 mark: Rejects derivation/pH/proof options as out of scope (any clear statement)
(5 marks) A buffer is prepared with a weak acid HA (given ). The mixture contains and . Calculate the pH using Henderson–Hasselbalch.
1 mark: Writes Henderson–Hasselbalch equation
1 mark: Substitutes correct values with correct ratio
1 mark: Evaluates ratio to 3 (or equivalent)
1 mark: Correctly takes (method shown or correct calculator use)
1 mark: Correct final pH (to appropriate significant figures)

