AP Syllabus focus: ‘Buffer pH depends on the acid’s pKa and the concentration ratio of the conjugate pair, described by the Henderson–Hasselbalch equation: pH = pKa + log([A−]/[HA]).’
Buffers are designed to resist pH change, so predicting their pH quickly is essential. The Henderson–Hasselbalch equation links buffer pH to pKa and the conjugate pair ratio.
Core idea: buffer pH is controlled by a ratio
A buffer contains appreciable amounts of a weak acid and its conjugate base (or a weak base and its conjugate acid). The Henderson–Hasselbalch equation expresses that the pH is set by how much base-form you have compared to acid-form, not by either concentration alone.
Key terms you must recognise
Buffer: A solution that contains significant amounts of a conjugate acid–base pair and therefore resists large changes in pH when small amounts of acid or base are added.
A buffer’s two components must be present together; otherwise, the “ratio control” on pH is lost.
pKa: The negative base-10 logarithm of the acid dissociation constant, , which indicates the strength of a weak acid.
Lower pKa means a stronger acid; higher pKa means a weaker acid.
Conjugate acid–base pair: Two species that differ by one proton, such as , where can donate and can accept .
Henderson–Hasselbalch equation (what you use on buffers)
Use Henderson–Hasselbalch when the solution contains both and in meaningful amounts (a true buffer situation).
= acidity of the buffer solution (unitless)
= acid dissociation logarithmic constant for (unitless)
= equilibrium concentration of conjugate base (mol L)
= equilibrium concentration of weak acid (mol L)
This equation is base-10 logarithmic and depends on the ratio .
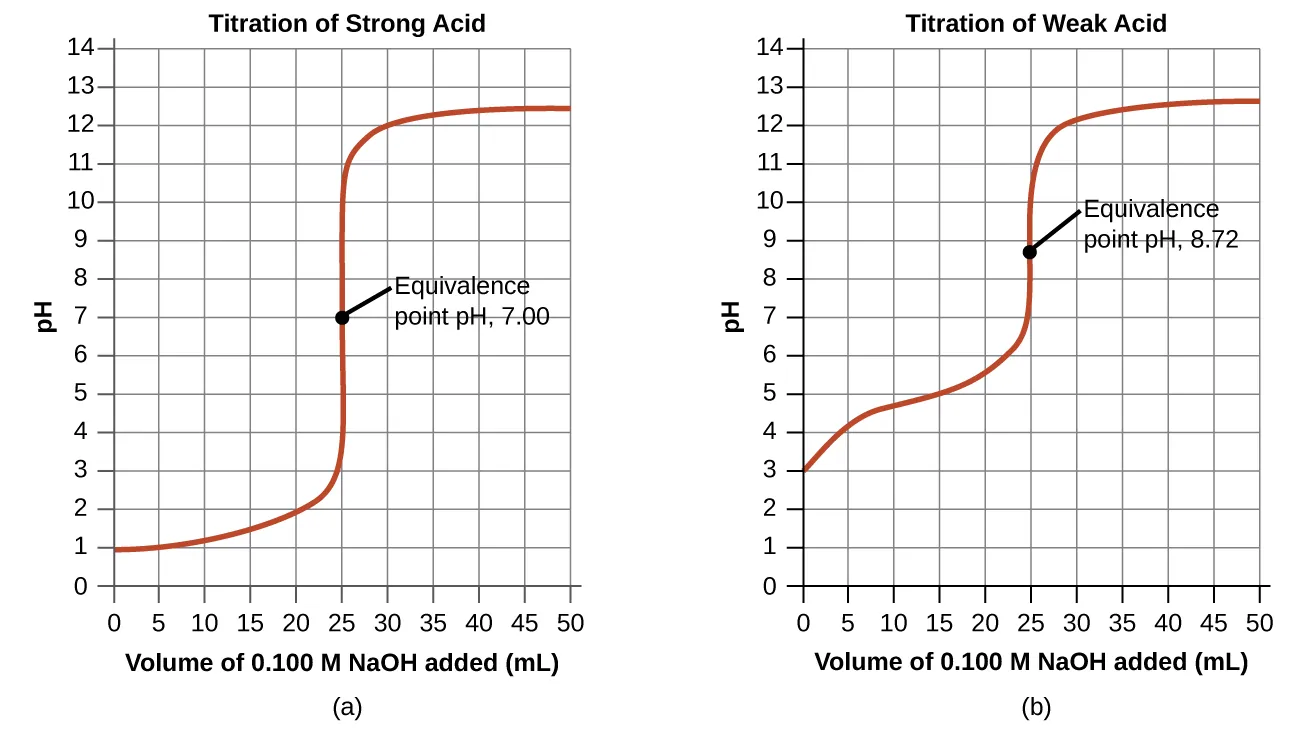
These titration curves plot pH versus volume of titrant added, contrasting a strong acid with a weak acid titrated by a strong base. The weak-acid curve shows an extended buffer region where both and are present, so Henderson–Hasselbalch applies and the midpoint (half-equivalence) corresponds to (therefore ). Source
How to apply the equation correctly
1) Identify the conjugate pair
Confirm the buffer pair is of the form weak acid and conjugate base .
Match the given pKa to the correct acid (not to ).
2) Determine the needed ratio term
Use the ratio exactly as written.
If the buffer is made by mixing solutions, it is often more reliable to use moles:
When both species are in the same final solution volume, because the volume cancels.
Maintain consistent units for concentrations (typically molarity).
3) Interpret the equation (quick qualitative checks)
If , then , so pH = pKa.
If , then , so pH > pKa (more basic).
If , then the log term is negative, so pH < pKa (more acidic).
Increasing both and by the same factor keeps the ratio (and thus the pH) unchanged.
Common pitfalls (and how to avoid them)
Swapping the ratio: Writing flips the sign and gives the wrong pH shift direction.
Using the wrong pKa: Ensure the provided corresponds to .
Forgetting it is a buffer requirement: If one component is essentially absent (all or all ), Henderson–Hasselbalch no longer describes the solution well.
Confusing concentration with amount: When solutions are mixed, compare final concentrations or use moles so dilution is handled correctly.
Log rules errors: Remember changes slowly; large pH changes require large ratio changes.
FAQ
It is arranged so that increasing the base form $A^-$ increases pH (a positive log term), matching the chemical expectation for a more basic buffer.
Reversing the ratio would require subtracting the log term instead.
You may use $\log(n_{A^-}/n_{HA})$ when both species are in the same final solution volume, because the common volume cancels.
This is especially helpful after mixing two solutions.
Practically, it performs best when both $HA$ and $A^-$ are present in comparable, non-negligible amounts, so the ratio is not extreme.
Very large or very small ratios can make real behaviour deviate from the simple model.
It means the buffer has equal “acid form” and “base form” available.
In that condition, the buffer is often most balanced in how it responds to added acid versus added base.
Because Henderson–Hasselbalch depends on the ratio $[A^-]/[HA]$, multiplying both concentrations by the same factor leaves the ratio unchanged.
However, the amount of acid/base the buffer can absorb before the ratio shifts substantially can change.
Practice Questions
(2 marks) A buffer contains and . State the Henderson–Hasselbalch equation and state the pH when .
1 mark:
1 mark: when
(5 marks) A buffer is prepared with . The final concentrations are and . (a) Calculate the buffer pH using Henderson–Hasselbalch. (b) State whether the pH is greater than, less than, or equal to , and justify using the ratio.
1 mark: correct substitution into
1 mark: ratio
1 mark: (allow -0.60)
1 mark: (allow 4.16–4.17)
1 mark: pH is less than because (or log term negative)

