AP Syllabus focus: ‘Adding small amounts of acid or base does not greatly change the [A−]/[HA] ratio, so the pH change is much smaller than it would be without the buffer.’
Buffers resist pH change because added acid or base is consumed by abundant conjugate partners. The key idea is that small additions barely disturb the conjugate ratio that controls pH.
Core idea: buffers minimise changes in the conjugate ratio
A buffer’s pH is governed primarily by the relative amounts of a conjugate acid–base pair (often written HA/A−). When a small amount of strong acid or base is added, a reaction occurs that converts one member of the pair into the other, causing only a small shift in their ratio.
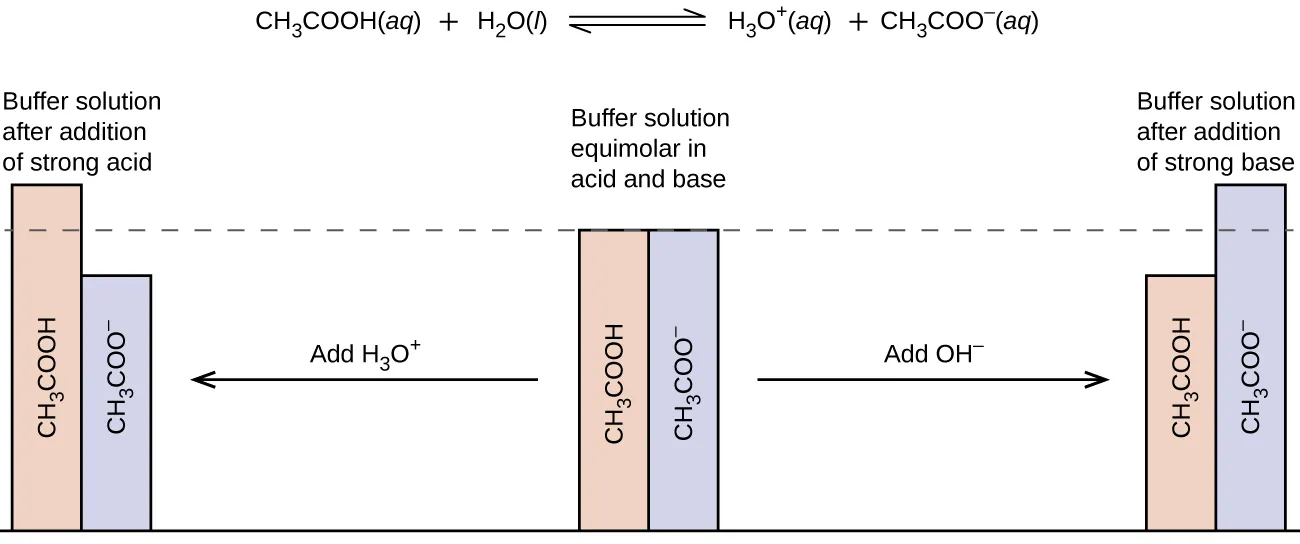
Bar-graph schematic showing how an equimolar buffer (acetic acid/acetate) responds to adding strong base versus strong acid. The diagram emphasizes that the buffer works mainly by converting one conjugate partner into the other, so the ratio changes modestly instead of producing large amounts of free or . Source
Buffer: a solution that resists pH change because it contains substantial amounts of both a weak acid and its conjugate base (or a weak base and its conjugate acid).
A “small” addition means the added moles are much smaller than the moles of HA and A− already present, so the pair acts like a chemical “reservoir.”
What happens when strong acid is added
Added strong acid increases H3O+ in solution, but in a buffer the conjugate base A− quickly consumes most of it:
Net effect: A− is converted to HA
Typical reaction: A− + H3O+ → HA + H2O
Result: [A−] decreases slightly, [HA] increases slightly, and free H3O+ does not accumulate much
Because the change in each component is small relative to its initial amount, the ratio [A−]/[HA] changes only a little.
What happens when strong base is added
Added strong base increases OH− in solution, but in a buffer the weak acid HA consumes most of it:
Net effect: HA is converted to A−
Typical reaction: HA + OH− → A− + H2O
Result: [HA] decreases slightly, [A−] increases slightly, and free OH− does not accumulate much
Again, the ratio [A−]/[HA] changes only a little when the added base is small compared with the buffer components.
Why “small ratio change” means “small pH change”
The relationship between buffer pH and the conjugate ratio is logarithmic, so even moderate-looking ratio changes translate into relatively small pH shifts.
= acidity of the solution (unitless)
= for the weak acid HA (unitless)
= equilibrium concentrations of conjugate base and weak acid (mol L)
Because pH depends on a log term, keeping [A−]/[HA] nearly constant keeps pH nearly constant. This is the specific reason stated in the syllabus: small additions do not greatly change the [A−]/[HA] ratio, so the pH change is much smaller than in an unbuffered solution.
Comparing to an unbuffered solution (conceptually)
Without a buffer, added acid or base largely remains as excess H3O+ or OH−, so the controlling concentration changes directly and dramatically:

Side-by-side beaker photographs comparing an unbuffered solution and a buffer at the same initial pH, then showing their different responses after adding a small amount of strong acid. The buffered sample shows little visible indicator change, illustrating how the conjugate pair removes most added before it can accumulate. Source
No reservoir reaction to “absorb” added H3O+ or OH−
The pH responds to the full added amount rather than a small residual
The pH jump is larger because [H3O+] or [OH−] changes by a larger factor
Limits of the “small addition” assumption
The buffering effect depends on having enough of both HA and A−. If the addition is not small, one component can be depleted substantially:
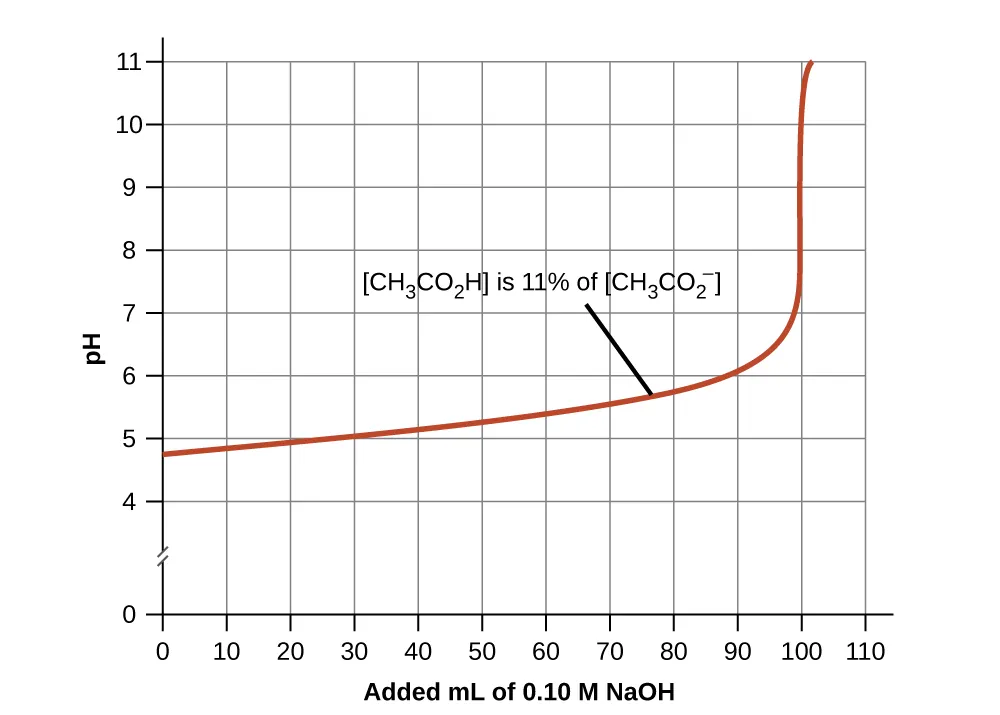
Plot of pH versus volume of strong base added to an acetic acid/acetate buffer, showing a gradual pH change while the buffer components are both present, followed by a sharp increase once buffering capacity is surpassed. The curve visually reinforces that buffering is strongest near and breaks down when one conjugate partner becomes too depleted. Source
If too much acid is added, A− is used up and H3O+ begins to accumulate
If too much base is added, HA is used up and OH− begins to accumulate
Once the ratio shifts a lot, the log term changes more, and pH moves more noticeably
FAQ
If the total volume change is negligible, the concentration ratio tracks the mole ratio closely.
If volume changes noticeably, use concentrations (or correct moles for the new volume) so the ratio is accurate.
A log function compresses multiplicative changes.
For example, doubling a ratio changes $\log$ by only about 0.30, so pH shifts modestly even when the ratio changes by a factor of 2.
When one component dominates, small additions can cause larger fractional changes in the smaller component.
That makes the ratio more “fragile,” so pH can start changing more per added amount.
If initial amounts of HA and A− are unequal, one direction of addition consumes the smaller component faster.
The ratio then changes more quickly in that direction, so pH can drift sooner.
Yes. At higher ionic strength, activities differ from concentrations, so the effective ratio can shift differently.
The qualitative reason remains the same (ratio changes little), but predictions using concentrations can be less accurate.
Practice Questions
(2 marks) Explain why adding a small amount of strong acid to a buffer causes only a small change in pH.
A− reacts with (consumes) added to form HA, so little free remains (1)
The ratio changes only slightly, so pH changes only slightly (1)
(5 marks) A buffer contains HA and A−. A small amount of strong base is added. Using the Henderson–Hasselbalch equation, explain qualitatively why the pH increase is small compared with adding the same base to pure water.
Added is consumed by HA to form A− and (1)
Therefore decreases slightly and increases slightly (1)
The ratio changes only a little for a small addition (1)
From , a small ratio change gives a small change in the log term (1)
In pure water there is no conjugate pair to consume , so accumulates and pH rises much more (1)

