AP Syllabus focus: ‘Electronegative elements stabilize the conjugate base relative to the conjugate acid, increasing acid strength.’
Electronegativity trends help you predict which acids donate protons more readily by focusing on how well the atoms in a molecule can pull, hold, and stabilise electron density after deprotonation.
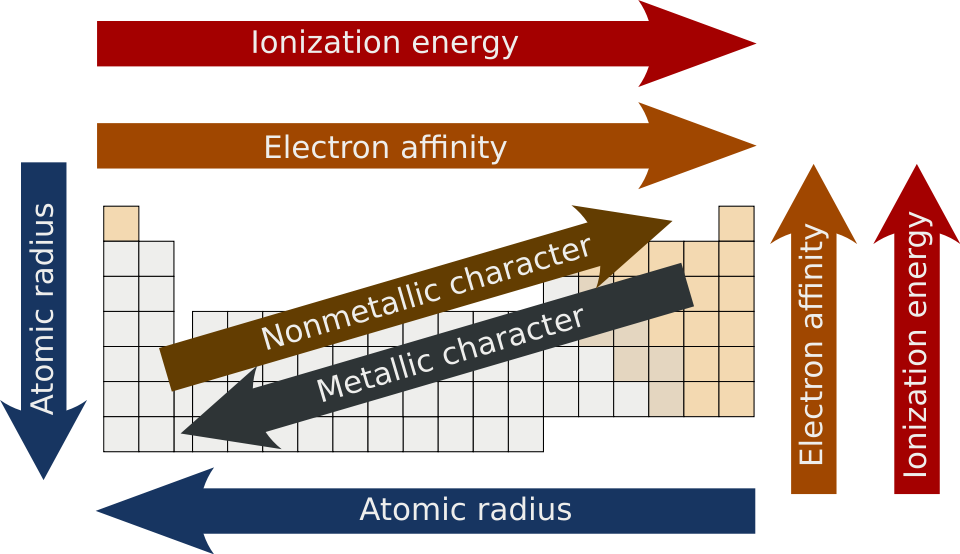
This periodic-trends diagram summarizes the directional increase of electronegativity on the periodic table (generally up and to the right). It helps justify why, within comparable structures, placing negative charge on a more electronegative atom tends to lower the conjugate base’s energy and increase acid strength. Source
Core idea: electronegativity stabilises the conjugate base
Acid strength increases when the conjugate base formed after deprotonation is lower in energy (more stable). A more electronegative atom can better accommodate negative charge, stabilising the conjugate base relative to the conjugate acid.
Electronegativity: A measure of an atom’s ability to attract shared electron density in a chemical bond.
In an acid–base reaction, stabilising the conjugate base shifts the equilibrium toward products, making proton donation more favourable.
What “stabilising the conjugate base” means in practice
When an acid loses H+:
Negative charge is left behind on (or near) the atom that was bonded to H.
A more electronegative atom:
Holds negative charge more tightly
Lowers the conjugate base’s potential energy
Makes the conjugate base less reactive (weaker base), which corresponds to a stronger acid
Binary acids (H–X): periodic electronegativity trend across a row
For binary acids of the form H–X within the same period (row) of the periodic table, acid strength generally increases as X becomes more electronegative. Two linked electronegativity-based ideas support this:
Bond polarity: As X becomes more electronegative, the H–X bond becomes more polar, making H more δ+ and easier to transfer as H+.
Conjugate-base stability: The conjugate base X− is stabilised when X is more electronegative.
How to apply the trend (qualitatively)
To compare acidity for H–X acids across the same row:
Identify X in each acid.
Compare electronegativity of X.
Predict: higher electronegativity of X → more stable X− → stronger acid.
This trend is most reliable when the acids are structurally similar and the comparison is made across the same period, where electronegativity changes substantially.
Oxyacids: electronegativity of the central atom
For oxyacids with the same number of oxygens and similar structures (e.g., HOX), the electronegativity of the central atom X is a key predictor of acid strength.
Why central-atom electronegativity matters
If X is more electronegative, it pulls electron density through the O–X bond:
This increases the O–H bond’s polarity, making the H more δ+.
After deprotonation, the negative charge on the conjugate base is stabilised because electron density is drawn away toward X.
So, for a structurally comparable series of oxyacids:
More electronegative central atom → more stabilised conjugate base → stronger acid.
Substituent electronegativity: local electron withdrawal increases acidity
Electronegativity trends also explain why adding electronegative atoms (like F, Cl, or other strongly electron-withdrawing groups) near an acidic site often increases acidity.
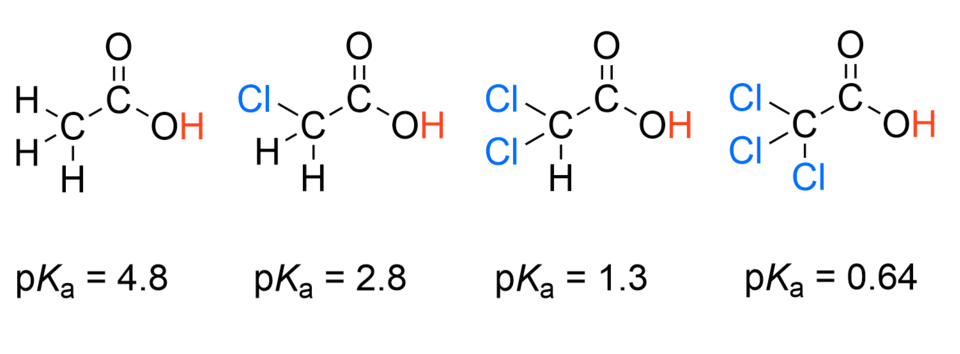
This figure lists values for acetic acid and the mono-, di-, and trichloroacetic acids, showing a strong acidity increase as more chlorines are substituted. The trend illustrates an inductive (electron-withdrawing) effect: electronegative substituents stabilize the conjugate base by pulling electron density through sigma bonds. Source
Interpreting electronegative substituents
Electronegative substituents pull electron density through σ bonds.
This decreases electron density near the site that will bear negative charge in the conjugate base.
Result: the conjugate base is stabilised, so the conjugate acid is stronger.
When comparing acids that differ only by substituents, prioritise:
Number of electronegative substituents
Proximity of the substituent to the acidic proton (closer usually has a larger effect)
Relative electronegativities of the substituent atoms
Common boundaries of the electronegativity rule (what to watch for)
Electronegativity is a powerful trend tool, but it is not the only factor controlling acid strength. Use it most confidently when:
The acids are closely related in structure
The comparison emphasises where the negative charge resides in the conjugate base
If different atoms, bonding environments, or charge distributions are involved, electronegativity still informs the prediction, but may not be the dominant factor.
FAQ
Across a period, electronegativity trends are often dominant.
Down a group, other factors can outweigh electronegativity, so predictions require extra caution.
It can better accommodate negative charge by attracting electron density towards itself.
That lowers the energy (reactivity) of the conjugate base.
Start by deprotonating at the H being discussed, then assign the formal negative charge to the atom that lost H.
Then assess whether electronegative atoms nearby can pull electron density.
No. The effect depends on:
how electronegative the substituent is
how many are present
how close they are to the acidic site
Because it assumes the same kind of conjugate base is formed and the negative charge is in a comparable position.
If charge distribution changes, electronegativity may not be the main factor.
Practice Questions
(2 marks) State whether HOCl or HOI is the stronger acid, and explain your choice in terms of electronegativity and conjugate-base stability.
Identifies HOCl as stronger (1)
Explains Cl is more electronegative than I, stabilising the conjugate base more (1)
(5 marks) Consider three acids with similar structures: HA, HB, and HC, where the atom bonded to H is A, B, or C from the same period, and electronegativity increases A < B < C. Predict the relative acid strengths and justify using conjugate-base stability and bond polarity.
Correct order: HC strongest, then HB, then HA (1)
Links higher electronegativity to more stable conjugate base (1)
States more stable conjugate base means stronger acid (1)
Links higher electronegativity to greater H–X bond polarity / H more δ+ (1)
Connects increased polarity to easier proton donation (1)

