AP Syllabus focus: ‘Strong acids have very weak conjugate bases stabilized by electronegativity, inductive effects, resonance, or a combination of these structural effects.’
Acid strength is best understood by focusing on what forms after proton loss. The more stable an acid’s conjugate base is, the more completely the acid donates in water.
Core principle: conjugate base stability controls acid strength
When an acid donates a proton, the products compete with the reactants in an equilibrium.
If the conjugate base is especially stable, it has little tendency to recombine with , so the equilibrium lies far to the right and the acid is strong.
Conjugate base and “weakness” of bases
A strong acid produces a conjugate base that is a very weak base (poor at accepting a proton). Weak basicity is typically the result of negative charge being stabilised by structural features in the conjugate base.
Conjugate base: the species formed when an acid donates a proton; it differs from the acid by one fewer and usually carries more negative charge.
This “stability of products” idea aligns with many trends students see: acids are stronger when the negative charge in the conjugate base is harder to concentrate at a single atom.
= acid dissociation constant (unitless)
= equilibrium concentration in
A larger corresponds to a more product-favoured dissociation, which generally means a more stabilised .
How structure stabilises conjugate bases (why acids get stronger)
The syllabus emphasises four closely related stabilisation ideas: electronegativity, inductive effects, resonance, or their combination. Each reduces the energetic “cost” of holding negative charge in the conjugate base.
1) Electronegativity: stabilising negative charge on the same atom type
If the negative charge in resides primarily on an atom, higher electronegativity stabilises that charge by attracting electron density more strongly. As a result, acids tend to be stronger when deprotonation places negative charge on more electronegative atoms (commonly O vs N vs C in typical AP contexts).
Key reasoning moves:
Identify where the negative charge is located in the conjugate base.
Ask whether that atom “likes” electron density (more electronegative = more stable anion).
More stable → weaker base → stronger acid.
2) Inductive effects: pulling electron density through sigma bonds
Electron-withdrawing groups can stabilise a conjugate base without being part of a resonance system. They work by drawing electron density through bonds, reducing charge build-up near the deprotonated site.
Inductive effect: stabilisation (or destabilisation) of charge caused by electron-withdrawing or electron-donating groups transmitting electron density through bonds.
Practical structural cues:
Electron-withdrawing substituents (often atoms/groups with high electronegativity) increase acidity by stabilising .
The effect is strongest when withdrawing groups are closer to the charged site and when multiple withdrawing groups are present.
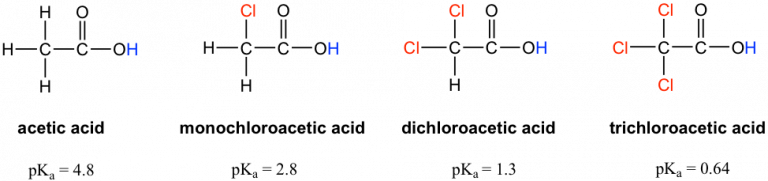
Inductive effects on acidity: acetic acid is compared with mono-, di-, and tri-chloroacetic acids, with values included to show the quantitative trend. Adding electron-withdrawing chlorine substituents increases polarization through bonds, stabilizing the conjugate base and increasing acid strength. Source
3) Resonance: delocalising charge over multiple atoms
If the conjugate base can be represented by multiple valid Lewis structures, the negative charge can be delocalised, lowering the energy of the anion and making it less basic.
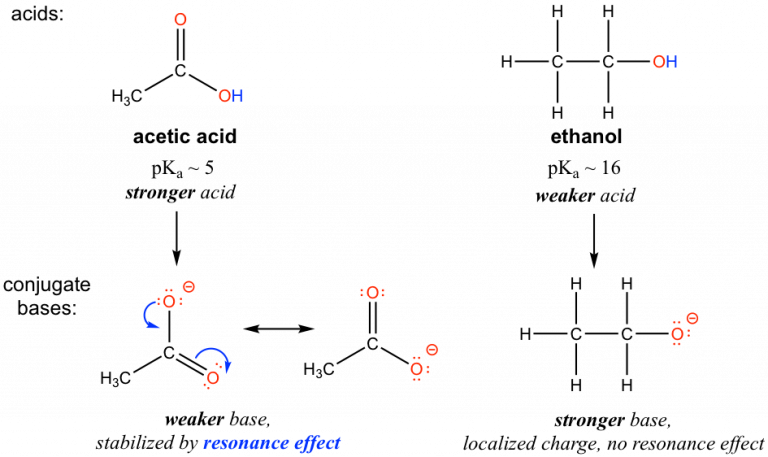
Resonance stabilization in carboxylates: the acetate conjugate base is shown with multiple resonance contributors that delocalize the negative charge over two oxygen atoms. This delocalization lowers the anion’s energy and reduces basicity, which shifts the dissociation equilibrium toward products and makes the parent acid stronger. Source
Resonance stabilisation: lowering of energy when electron density (often negative charge) is delocalised over two or more atoms across a conjugated system.
What to look for:
A deprotonation site adjacent to a bond or a functional group that can share electron density (common in many oxygen-containing acids).
More resonance forms that place negative charge on electronegative atoms generally mean greater stabilisation.
Greater stabilisation of strongly favours proton loss, increasing acid strength.
4) Combining effects: why some acids are extremely strong
In many strong acids, multiple stabilising factors reinforce each other:
An electronegative atom bears much of the negative charge.
Additional electronegative atoms/groups withdraw electron density by induction.
Resonance spreads the charge over several atoms.
This combined stabilisation produces an exceptionally weak conjugate base that has minimal tendency to reclaim a proton, which is the structural reason some acids behave as “strong” in water.
FAQ
Look for highly electronegative atoms (often O, N, halogens) attached near the charged site.
More and closer electron-withdrawing groups generally increase stabilisation.
Delocalisation spreads electron density over a larger volume and across multiple atoms.
This reduces charge concentration and lowers electrostatic energy.
Inductive effects are transmitted through $\sigma$ bonds.
They decrease rapidly with distance (more bonds between substituent and charge means weaker influence).
Yes—if resonance in the acid (reactant) is lost upon deprotonation, the conjugate base may be less stabilised than expected.
Compare stabilisation on both sides, not just the anion.
When electronegativity, induction, and resonance strongly stabilise the anion, proton recombination is unfavourable.
The equilibrium lies far to the right, giving “strong acid” behaviour in water.
Practice Questions
(2 marks) Explain, in terms of conjugate base stability, why a strong acid has a weak conjugate base.
States that strong acid dissociation is favoured when the conjugate base is stabilised / low energy. (1)
Links stabilised conjugate base to weak basicity (low tendency to accept / reform acid). (1)
(5 marks) A student compares two acids, and . The conjugate base is stabilised by resonance and an electron-withdrawing group, while has neither feature. Using structural arguments only (no calculations), justify which acid is stronger.
Identifies that acid strength increases with conjugate base stabilisation. (1)
Explains resonance delocalises negative charge, stabilising . (1)
Explains electron-withdrawing group exerts an inductive effect, further stabilising . (1)
Concludes is less basic than . (1)
Therefore is the stronger acid (more product-favoured dissociation). (1)

