AP Syllabus focus: ‘Which protons participate in acid–base reactions, and their relative strengths, can be inferred from molecular structure.’
Recognising acidic protons from structure lets you predict which site will donate in water and compare acid strengths without memorising every compound. Focus on the atom bearing H, and how stable the conjugate base would be.
What it means for a proton to be “acidic”
An acidic proton is one that can be transferred in a Brønsted–Lowry acid–base reaction under typical aqueous conditions.
Acidic proton: a hydrogen atom in a species that can be removed as by a base, producing a conjugate base that is sufficiently stable to exist in solution.
Whether a proton is acidic is mainly a question of conjugate-base stability after deprotonation.
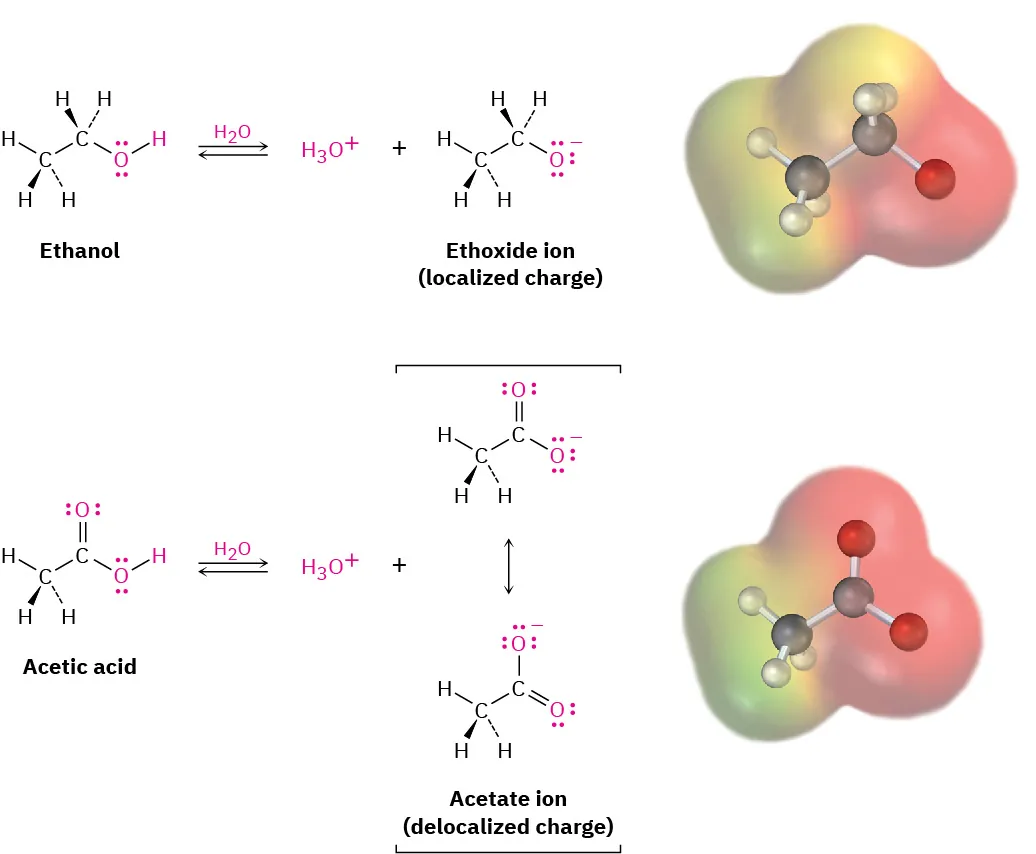
Comparison of deprotonation products showing an alkoxide ion (charge localized on one oxygen) versus a carboxylate ion (charge delocalized across two oxygens). The more delocalized conjugate base is lower in energy, so the corresponding acid is stronger in water. Source
Conjugate base: the species formed after an acid donates a proton; it has one fewer and typically carries a more negative (or less positive) charge than the acid.
A useful way to “see” acidity is to picture the deprotonation event.
= acid (proton donor)
= base (proton acceptor)
= hydronium ion formed in water
= conjugate base formed after deprotonation
Structural checklist for identifying acidic protons
1) Where is the hydrogen attached?
Start by identifying the atom directly bonded to H.
H on electronegative atoms (O–H, N–H, S–H) is commonly acidic.
H on carbon (C–H) is usually not acidic unless special stabilisation is available.
2) Does deprotonation place negative charge on a stable atom?
After removing , ask: “Where does the electron density go?”
Negative charge on oxygen is generally more stabilised than on nitrogen, and both are far more stabilised than on carbon.
Positive species with N–H (e.g., ammonium-like groups) often have acidic N–H protons because deprotonation relieves positive charge.
3) Can the conjugate base delocalise charge?
Look for resonance possibilities once the proton is removed.
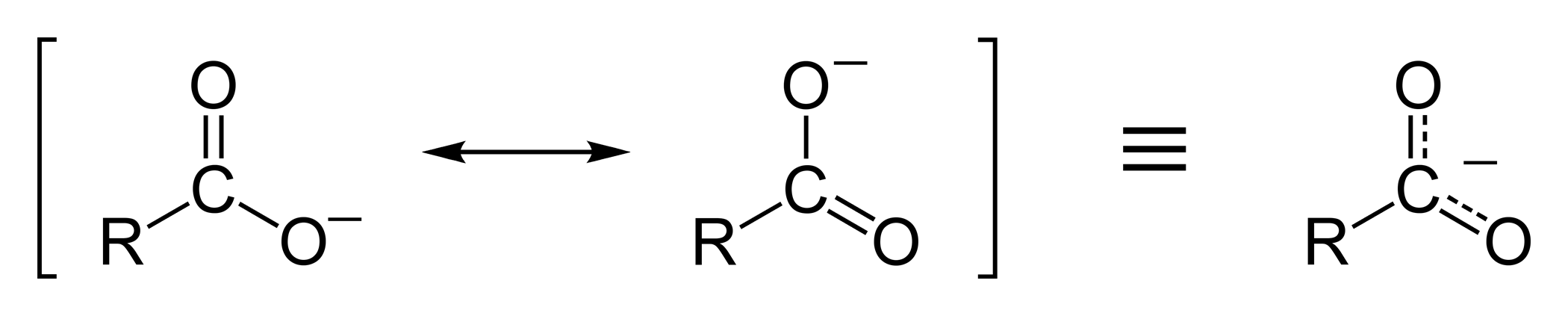
Resonance contributors for a carboxylate ion, illustrating that the negative charge is delocalized over two equivalent oxygen atoms. This delocalization stabilizes the conjugate base, which is why the O–H proton of a carboxylic acid is comparatively acidic. Source
If deprotonation creates an anion that can spread charge across multiple atoms (especially onto electronegative atoms), the proton is more acidic.
Common resonance-enabled sites:
Carboxylic acid O–H (anion delocalised over two oxygens)
Phenol O–H (anion delocalised into aromatic ring)
Sulfonic acid O–H (extensive delocalisation over multiple oxygens)
4) Are there electron-withdrawing groups nearby?
Nearby substituents can pull electron density away through the sigma framework (inductive effects), stabilising the conjugate base.
Electron-withdrawing groups (e.g., halogens, nitro, carbonyl-containing groups) near the acidic site increase acidity.
The effect is stronger when the group is:
closer to the acidic atom
present in greater number
more strongly withdrawing
5) Is the proton “activated” by bonding and orbital effects?
Some acidic protons appear on carbon when the resulting carbanion is unusually stabilised.
Terminal alkyne C–H can be acidic relative to other C–H bonds because the negative charge resides in an orbital with high character (carbon holds electron density closer to the nucleus).
Alpha C–H next to carbonyls (adjacent to ) can be acidic because deprotonation can lead to resonance-stabilised electron distribution (often discussed as enolate formation). For AP-level identification, it is usually enough to recognise “C–H next to carbonyl” as a comparatively acidic C–H site.
Common functional groups: which H is the acidic one?
Strongly acidic (easy to spot)
Carboxylic acids: the O–H proton is acidic; the –COOH hydrogen is the one that transfers.
Sulfonic acids: the O–H proton is acidic.
Moderately acidic
Phenols: the O–H proton is acidic due to aromatic resonance.
Ammonium (protonated amines): N–H protons are acidic because deprotonation gives a neutral amine.
Weakly acidic (still can act as acids with strong bases)
Alcohols: the O–H proton is acidic but much less so than carboxylic acids.
Thiols: the S–H proton can be acidic; compare using conjugate-base stability rather than assuming it matches alcohols.
Usually not acidic in water
Alkane C–H (typical hydrocarbon hydrogens): not appreciably acidic.
Aryl/vinylic C–H (on benzene rings or alkenes): generally not the acidic site for standard aqueous acid–base reasoning.
Prioritising when multiple acidic protons exist
When a molecule has more than one candidate proton, select the site whose conjugate base is most stabilised.
Prioritise O–H acidity over N–H over most C–H, unless a particular C–H is clearly resonance/inductively stabilised.
If comparing two O–H sites, the more acidic one is usually the one whose deprotonation places negative charge into a more resonance-stabilised and/or more electron-withdrawn environment.
In polyfunctional molecules, identify the most likely deprotonation site by checking:
charge relief (loss of positive charge)
resonance after deprotonation
proximity of electron-withdrawing groups
whether the anion ends up mainly on O vs N vs C
FAQ
In most neutral molecules, $O!-!H$ is more acidic than $N!-!H$ because the conjugate base places negative charge on oxygen, which stabilises it better.
Exception: if the nitrogen is positively charged (e.g., ammonium), $N!-!H$ can be quite acidic because deprotonation removes the positive charge.
A C–H can be acidic when the conjugate base is stabilised unusually well, for example by:
strong resonance delocalisation into adjacent $\pi$ systems
very strong electron-withdrawing substituents
orbital effects (greater $s$ character holding negative charge closer to the nucleus)
These factors can outweigh carbon’s usual tendency to destabilise negative charge.
Hydrogen bonding can influence acidity by stabilising either the acid or the conjugate base.
If hydrogen bonding stabilises the conjugate base more than the acid, acidity increases; if it stabilises the acid more, acidity decreases.
The direction depends on the specific structure and solvent interactions.
Compare how stabilised each resulting oxyanion would be.
Look for:
resonance possibilities after deprotonation (major advantage)
nearby electron-withdrawing groups that would stabilise the negative charge inductively
whether deprotonation would create or disrupt an especially stable arrangement (e.g., aromaticity in some specialised cases)
Not always.
Kinetic factors, solvent, steric hindrance, and the strength/bulk of the base can change which proton is removed fastest.
“Most acidic” is a thermodynamic idea (most stable conjugate base), while “removed first” can sometimes be controlled by conditions.
Practice Questions
Question 1 (2 marks) Circle the acidic proton(s) in each species and state the atom type (O, N, or C) to which each acidic proton is attached: a) b)
(1) a) Identifies the proton in ethanol as acidic; attached to O.
(1) b) Identifies an proton in as acidic; attached to N.
Question 2 (6 marks) For the molecule (glycolic acid), identify two different acidic protons. For each, justify which is more acidic using structural arguments about the conjugate base. Your answer must reference at least two of: electronegativity, resonance, inductive effects, charge.
(1) Identifies the carboxylic acid proton as acidic.
(1) Identifies the alcohol proton as acidic (though weaker).
(1) States the carboxylic acid proton is more acidic.
(1) Justifies using resonance: carboxylate conjugate base delocalises charge over two O atoms.
(1) Justifies using electronegativity/charge placement: negative charge resides on O (stabilised) in carboxylate.
(1) Additional valid justification: inductive withdrawal from the nearby O group stabilises the carboxylate (or notes the alkoxide lacks comparable resonance stabilisation).

