AP Syllabus focus: ‘Weak bases partially ionize in water to produce OH− and establish equilibrium characterized by Kb (and pKb = −log Kb); pH depends on initial base concentration and pKb.’
Weak bases create basic solutions by reacting incompletely with water, establishing an equilibrium that produces OH−. AP Chemistry problems focus on using or with initial concentration to determine pH.
Weak bases and their equilibrium with water
A weak base reacts with water but only partially forms products, so appreciable amounts of both reactants and products are present at equilibrium.
Weak base: a base that partially ionises in water, establishing an equilibrium rather than reacting to completion.
A common generic reaction is:
is the base; is its conjugate acid
Water is a reactant but is treated as a constant in the equilibrium expression
The base ionisation constant,
The equilibrium position is described by , which is specific to the base and temperature.
Base ionisation constant (): the equilibrium constant for a base reacting with water to form its conjugate acid and hydroxide ion.
= base ionisation constant (unitless)
= equilibrium concentration of conjugate acid,
= equilibrium concentration of hydroxide ion,
= equilibrium concentration of base,
Larger means the base produces more (lies further to products) and therefore gives a higher pH for the same initial concentration.
Using
Chemists often use to simplify comparisons and calculations involving logarithms.
: the negative base-10 logarithm of ; smaller corresponds to a stronger base.
Finding pH of a weak base (problem setup)
To find pH, you determine the equilibrium produced by the weak base, then convert to pH.
Core method (ICE + equilibrium)
Write the weak-base equilibrium:
Set up an ICE table (Initial, Change, Equilibrium) in
Initial: known; typically ,
Change: let be the amount that reacts; then , ,
Substitute equilibrium concentrations into the expression and solve for
Decide whether the small- approximation is valid:
If , then
If not valid, solve the resulting quadratic (or use a calculator)
Converting to pH
Once is known, compute pOH and then pH (at ).
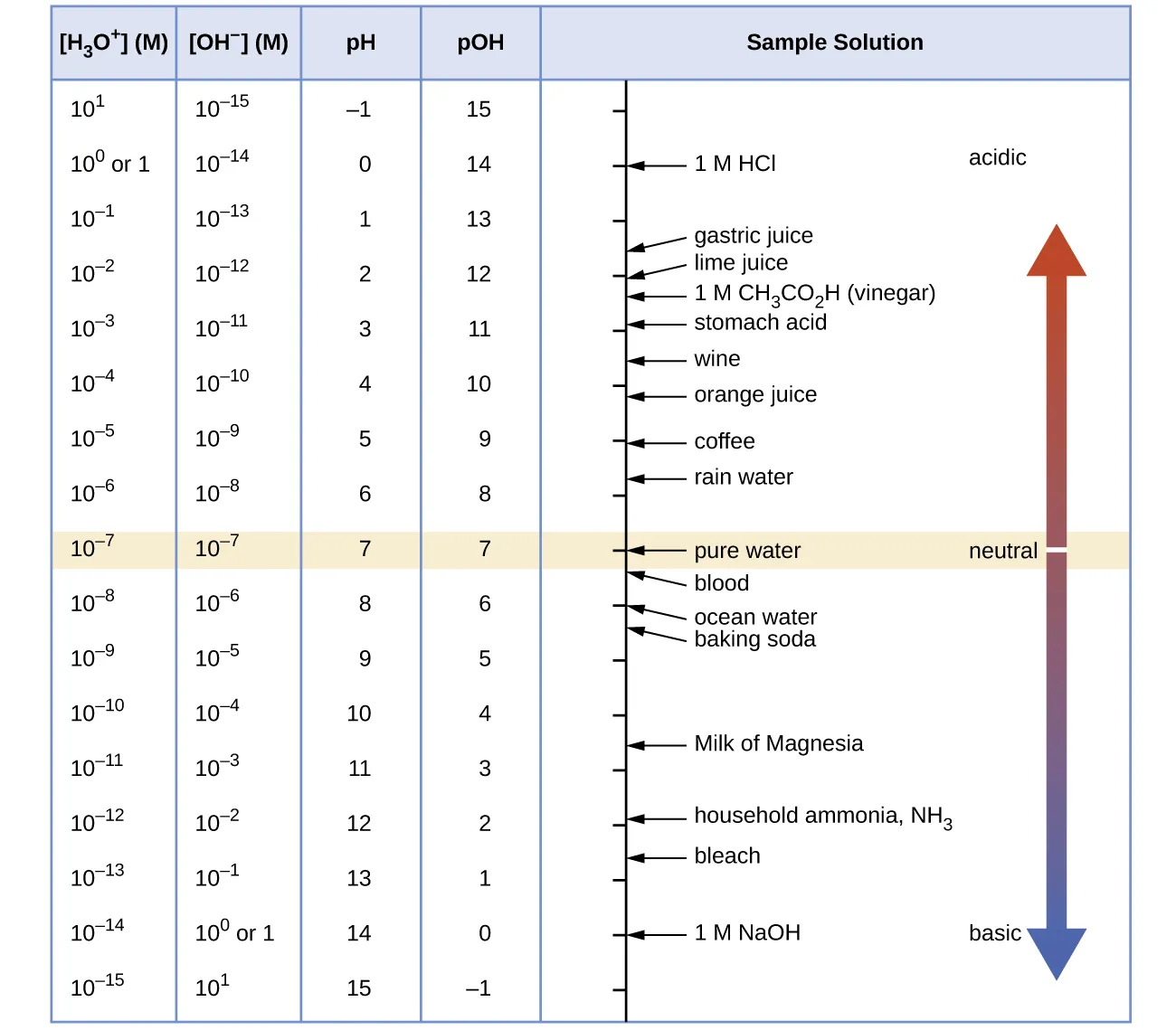
pH–pOH scale chart showing the inverse relationship between and across many orders of magnitude. It visually reinforces that each 1-unit change in pH corresponds to a tenfold change in concentration, and that at , pH and pOH are linked by . Source
= negative log of (unitless)
= base ionisation constant (unitless)
= negative log of hydroxide concentration (unitless)
= hydroxide concentration,
= negative log of hydronium concentration (unitless)
What “pH depends on initial base concentration and ” means
Higher initial generally increases equilibrium , raising pH
Smaller (larger ) shifts equilibrium toward , raising pH
Because ionisation is partial, is typically much smaller than
FAQ
It can fail when $K_b$ is relatively large and/or the initial base concentration is very small, so ionisation is not negligible compared with $[\mathrm{B}]_0$.
A quick check is whether the computed $x$ would exceed about 5% of $[\mathrm{B}]_0$.
In many weak-base solutions, the $\mathrm{OH^-}$ produced by base ionisation is far greater than $1.0\times10^{-7},\mathrm{mol,L^{-1}}$ from water.
If the base is extremely dilute or very weak, water’s contribution can become non-negligible.
No. Dilution decreases the initial base concentration, which usually lowers equilibrium $[\mathrm{OH^-}]$.
Although percent ionisation can increase upon dilution, the actual $[\mathrm{OH^-}]$ often decreases, so pH typically drops.
You treat each protonation step with its own equilibrium and constant (often stepwise $K_b$ values).
If only one $K_b$ (or $pK_b$) is provided, AP-style problems usually intend a single dominant equilibrium.
Liquid water’s concentration is effectively constant in dilute aqueous solutions, so it is incorporated into the constant.
This is why $K_b$ uses only aqueous species concentrations at equilibrium.
Practice Questions
(2 marks) A solution of has . Write the equilibrium expression for and state which species’ concentration you would solve for first to obtain pH.
Writes (1)
Identifies solving for (or from an ICE table) as the first step toward pH (1)
(5 marks) A weak base is prepared at concentration and has . Describe, with chemical equations and relationships, how to determine the pH of the solution. You may assume .
Writes equilibrium (1)
Uses to obtain (1)
Sets up equilibrium concentrations (e.g., ICE: , , ) (1)
Substitutes into and solves for (allow small- or quadratic) (1)
Converts to pH using and (1)

