AP Syllabus focus: ‘A weak acid establishes equilibrium between HA and A− with Ka (and pKa = −log Ka); pH can be determined from the initial acid concentration and pKa.’
Weak acids only partially ionize in water, so their pH must be found by combining an equilibrium model with (or ). This page outlines the essential setup and solution logic.
Weak acid equilibrium model
A weak acid reacts reversibly with water, establishing an equilibrium mixture of undissociated acid and ions.
Weak acid: an acid that ionizes only partially in water, so equilibrium lies mostly toward reactants.
The standard reaction model (for a monoprotic acid) is:

General monoprotic weak-acid equilibrium in water, showing establishing equilibrium with and . This visual directly supports writing the equilibrium expression and setting up an ICE table for weak-acid pH calculations. Source
Water is a pure liquid and is not included in the equilibrium expression.
The acid dissociation constant,
Acid dissociation constant (): an equilibrium constant that quantifies the extent to which donates a proton to water to form and .
A larger means more products at equilibrium and therefore a lower pH (more acidic).
= acid dissociation constant (unitless)
= equilibrium hydronium concentration (M)
= equilibrium conjugate base concentration (M)
= equilibrium weak acid concentration (M)
Using to simplify acid strength comparisons
Because values often span many powers of ten, chemists commonly use .
: the negative base-10 logarithm of , used to express acid strength on a compressed scale.
A lower corresponds to a stronger acid (larger ).
= logarithmic acid-strength measure (unitless)
= acid dissociation constant (unitless)
Finding pH from initial concentration and
To determine pH, you connect the initial acid concentration (often called the formal concentration, ) to the equilibrium produced by dissociation.
Core setup: ICE approach (no numbers required)
Use an ICE (Initial–Change–Equilibrium) framework:
Initial: , ,
Change: decreases by ; increases by ; increases by
Equilibrium: , ,
Substitute these equilibrium expressions into the expression:
Common AP-level approximation
For many weak acids, ionization is small relative to the starting amount, so . Then:
, and represents
This approximation must be checked conceptually: it is most reliable for small and not-too-dilute .
Converting to pH
Once equilibrium is found (exactly or approximately), compute pH.
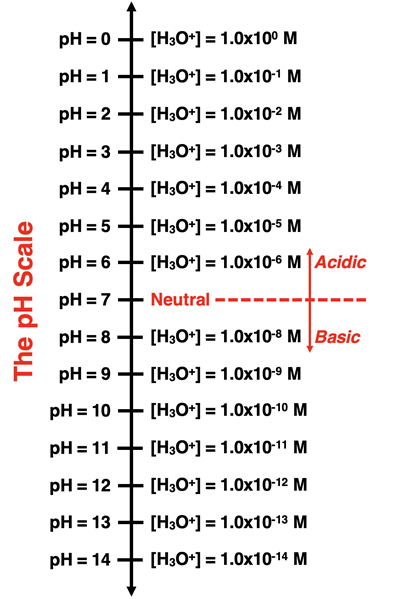
A labeled pH scale that pairs each integer pH value with the corresponding hydronium concentration, emphasizing the base-10 logarithmic nature of . It helps students interpret how a 1-unit change in pH corresponds to a tenfold change in . Source
= acidity scale value (unitless)
= equilibrium hydronium concentration (M)
Practical guidance for correct equilibrium reasoning
Choosing vs
If is given, convert to when you need to solve with algebra: .
If is given, you can proceed directly to the equilibrium setup.
What the equilibrium picture implies
Because most remains, is much smaller than for a weak acid.
The conjugate base forms in the same amount as hydronium for a monoprotic acid at equilibrium: (within the model’s assumptions).
When the approximation is not sufficient
If is not negligible compared with , you must treat exactly and solve the resulting equation rather than simplifying it. This is an equilibrium-math issue, not a different chemical model.
FAQ
Stoichiometry: each dissociation event produces one $A^-$ for each $H_3O^+$. This equality holds within the simplified model and ignores other sources/sinks of $H_3O^+$.
Compare magnitudes: smaller $K_a$ and larger $C_0$ favour small $x$. If you expect substantial dissociation, keep $C_0-x$ and solve exactly.
Initial (formal) concentration is what you prepare, $C_0$. Equilibrium concentrations are what remain after partial dissociation and must satisfy both mass balance and $K_a$.
At extremely low acid concentrations, water’s own contribution to $[H_3O^+]$ can become comparable. Then the simple assumption that all $H_3O^+$ comes from $HA$ is less accurate.
Log scales are easier to compare and interpret: a change of 1 unit in $pK_a$ corresponds to a tenfold change in $K_a$. It can also reduce calculator rounding when handling very small $K_a$ values.
Practice Questions
(2 marks) For the weak acid in water, write the expression for and identify which equilibrium concentration corresponds to in an ICE table using change .
1 mark:
1 mark: states (and/or , )
(5 marks) A solution of a monoprotic weak acid has . Calculate the pH, stating any approximation used.
1 mark: converts to
1 mark: sets up with
1 mark: uses approximation (explicitly stated)
1 mark: finds and identifies
1 mark: with correct pH value

